Abstract
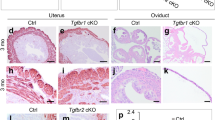
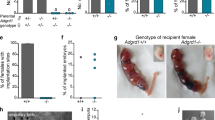
Elimination of the developing female reproductive tract in male fetuses is an essential step in mammalian sexual differentiation1. In males, the fetal testis produces the transforming growth factor β (TGF-β) family member anti-Müllerian hormone (Amh, also known as Müllerian-inhibiting substance (Mis)), which causes regression of the Müllerian ducts, the primordia of the oviducts, uterus and upper vagina2. Amh induces regression by binding to a specific type II receptor (Amhr2) expressed in the mesenchyme surrounding the ductal epithelium3,4,5,6,7. Mutations in AMH or AMHR2 in humans and mice disrupt signaling, producing male pseudohermaphrodites that possess oviducts and uteri8,9,10. The type I receptor and Smad proteins that are required in vivo for Müllerian duct regression have not yet been identified11,12,13,14. Here we show that targeted disruption of the widely expressed type I bone morphogenetic protein (BMP) receptor Bmpr1a (also known as Alk3) in the mesenchymal cells of the Müllerian ducts leads to retention of oviducts and uteri in males. These results identify Bmpr1a as a type I receptor for Amh-induced regression of Müllerian ducts. Because Bmpr1a is evolutionarily conserved, these findings indicate that a component of the BMP signaling pathway has been co-opted during evolution for male sexual development in amniotes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Josso, N. et al. Anti-Müllerian hormone: the Jost factor. Recent Prog. Horm. Res. 48, 1–59 (1993).
Jost, A. Problems of fetal endocrinology: the gonadal and hypophyseal hormones. Recent Prog. Horm. Res. 8, 379–418 (1953).
Baarends, W.M. et al. A novel member of the transmembrane serine/threonine kinase receptor family is specifically expressed in the gonads and in mesenchymal cells adjacent to the Müllerian duct. Development 120, 189–197 (1994).
di Clemente, N. et al. Cloning, expression, and alternative splicing of the receptor for anti-Müllerian hormone. Mol. Endocrinol. 8, 1006–1020 (1994).
Teixeira, J. et al. Developmental expression of a candidate Müllerian inhibiting substance type II receptor. Endocrinology 137, 160–165 (1996).
Roberts, L.M., Hirokawa, Y., Nachtigal, M.W. & Ingraham, H.A. Paracrine-mediated apoptosis in reproductive tract development. Dev. Biol. 208, 110–122 (1999).
Allard, S. et al. Molecular mechanisms of hormone-mediated Müllerian duct regression: involvement of β-catenin. Development 127, 3349–3360 (2000).
Behringer, R.R., Finegold, M.J. & Cate, R.L. Müllerian-inhibiting substance function during mammalian sexual development. Cell 79, 415–425 (1994).
Mishina, Y. et al. Genetic analysis of the Müllerian-inhibiting substance signal transduction pathway in mammalian sexual differentiation. Genes Dev. 10, 2577–2587 (1996).
Belville, C., Josso, N. & Picard, J.Y. Persistence of Müllerian derivatives in males. Am. J. Med. Genet. 89, 218–223 (1999).
Massagué, J., Blain, S.W. & Lo, R.S. TGF-β signaling in growth control, cancer, and heritable disorders. Cell 103, 295–309 (2000).
Gouédard, L. et al. Engagement of bone morphogenetic protein type IB receptor and Smad1 signaling by anti-Müllerian hormone and its type II receptor. J. Biol. Chem. 275, 27973–27978 (2000).
Clarke, T.R. et al. Müllerian inhibiting substance signaling uses a bone morphogenetic protein (BMP)-like pathway mediated by ALK2 and induces SMAD6 expression. Mol. Endocrinol. 15, 946–959 (2001).
Visser, J.A. et al. The serine/threonine transmembrane receptor ALK2 mediates Müllerian inhibiting substance signaling. Mol. Endocrinol. 15, 936–945 (2001).
Mishina, Y., Suzuki, A., Ueno, N. & Behringer, R.R. Bmpr encodes a type I bone morphogenetic protein receptor that is essential for gastrulation during mouse embryogenesis. Genes Dev. 9, 3027–3037 (1995).
Gu, Z. et al. The type I activin receptor ActRIB is required for egg cylinder organization and gastrulation in the mouse. Genes Dev. 12, 844–857 (1998).
Gu, Z. et al. The type I serine/threonine kinase receptor ActRIA (Alk2) is required for gastrulation of the mouse embryo. Development 126, 2551–2561 (1999).
Mishina, Y., Crombie, R., Bradley, A. & Behringer, R.R. Multiple roles for activin-like kinase-2 signaling during mouse embryogenesis. Dev. Biol. 213, 314–326 (1999).
Oh, S.P. et al. Activin receptor-like kinase 1 modulates transforming growth factor-β 1 signaling in the regulation of angiogenesis. Proc. Natl Acad. Sci. USA 97, 2626–2631 (2000).
Yi, S.E., Daluiski, A., Pederson, R., Rosen, V. & Lyons, K.M. The type I BMP receptor BMPRIB is required for chondrogenesis in the mouse limb. Development 127, 621–630 (2000).
Yi, S.E. et al. The type I BMP receptor BmprIB is essential for female reproductive function. Proc. Natl Acad. Sci. USA 98, 7994–7999 (2001).
Durlinger, A.L. et al. Control of primordial follicle recruitment by anti-Müllerian hormone in the mouse ovary. Endocrinology 140, 5789–5796 (1999).
Nagy, A. Cre recombinase: the universal reagent for genome tailoring. Genesis 26, 99–109 (2000).
Arango, N.A., Lovell-Badge, R. & Behringer, R.R. Targeted mutagenesis of the endogenous mouse Mis gene promoter: in vivo definition of genetic pathways of vertebrate sexual development. Cell 99, 409–419 (1999).
Soriano, P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat. Genet. 21, 70–71 (1999).
Mishina, Y., Hanks, M.C., Miura, S., Tallquist, M.D. & Behringer, R.R. Generation of Bmpr/Alk3 conditional knockout mice. Genesis 32, 69–72 (2002).
Ahn, K., Mishina, Y., Hanks, M.C., Behringer, R.R. & Crenshaw, E.B. 3rd. BMPR-IA signaling is required for the formation of the apical ectodermal ridge and dorsal-ventral patterning of the limb. Development 128, 4449–4461 (2001).
Gaussin, V. et al. Endocardial cushion and myocardial defects after cardiac myocyte-specific conditional deletion of the bone morphogenetic protein receptor ALK3. Proc. Natl Acad. Sci. USA 99, 2878–2883 (2002).
Newfeld, S.J., Wisotzkey, R.G. & Kumar, S. Molecular evolution of a developmental pathway: phylogenetic analyses of transforming growth factor-b family ligands, receptors and Smad signal transducers. Genetics 152, 783–795 (1999).
Albrecht, U. et al. Platelet-activating factor acetylhydrolase expression and activity suggest a link between neuronal migration and platelet-activating factor. Dev. Biol. 180, 579–593 (1996).
Acknowledgements
We thank S. Pfaff for the IRES–cre plasmid, L. Li for help in the construction of the cre targeting vector, A. Bradley for embryonic stem and STO feeder cells, J.M. Deng for assistance with tissue culture, P. Soriano for R26R mice and Y. Wang for help with histology. This work was supported by grants from the US National Institutes of Health and the Barnts Family to R.R.B. S.P.J. is supported by a fellowship from the Lalor Foundation. DNA sequencing and veterinary resources were supported by the US National Institutes of Health Cancer Center Support (Core) Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Jamin, S., Arango, N., Mishina, Y. et al. Requirement of Bmpr1a for Müllerian duct regression during male sexual development. Nat Genet 32, 408–410 (2002). https://doi.org/10.1038/ng1003
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1003
This article is cited by
-
A re-appraisal of mesenchymal-epithelial transition (MET) in endometrial epithelial remodeling
Cell and Tissue Research (2023)
-
Insights into in vivo follicle formation: a review of in vitro systems
Histochemistry and Cell Biology (2022)
-
New insights into anti-Müllerian hormone role in the hypothalamic–pituitary–gonadal axis and neuroendocrine development
Cellular and Molecular Life Sciences (2021)
-
A Comprehensive Overview of Common Polymorphic Variants in Genes Related to Polycystic Ovary Syndrome
Reproductive Sciences (2021)
-
Overactivated sonic hedgehog signaling aggravates intrauterine adhesion via inhibiting autophagy in endometrial stromal cells
Cell Death & Disease (2020)