Abstract
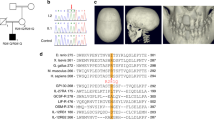
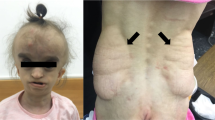
Interferon regulatory factor 6 (IRF6) belongs to a family of nine transcription factors that share a highly conserved helix–turn–helix DNA-binding domain and a less conserved protein-binding domain. Most IRFs regulate the expression of interferon-α and -β after viral infection1, but the function of IRF6 is unknown. The gene encoding IRF6 is located in the critical region for the Van der Woude syndrome (VWS; OMIM 119300) locus at chromosome 1q32–q41 (refs 2,3). The disorder is an autosomal dominant form of cleft lip and palate with lip pits4, and is the most common syndromic form of cleft lip or palate. Popliteal pterygium syndrome (PPS; OMIM 119500) is a disorder with a similar orofacial phenotype that also includes skin and genital anomalies5. Phenotypic overlap6 and linkage data7 suggest that these two disorders are allelic. We found a nonsense mutation in IRF6 in the affected twin of a pair of monozygotic twins who were discordant for VWS. Subsequently, we identified mutations in IRF6 in 45 additional unrelated families affected with VWS and distinct mutations in 13 families affected with PPS. Expression analyses showed high levels of Irf6 mRNA along the medial edge of the fusing palate, tooth buds, hair follicles, genitalia and skin. Our observations demonstrate that haploinsufficiency of IRF6 disrupts orofacial development and are consistent with dominant-negative mutations disturbing development of the skin and genitalia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Taniguchi, T., Ogasawara, K., Takaoka, A. & Tanaka, N. IRF family of transcription factors as regulators of host defense. Annu. Rev. Immunol. 19, 623–655 (2001).
Murray, J.C. et al. Linkage of an autosomal dominant clefting syndrome (Van der Woude) to loci on chromosome 1q. Am. J. Hum. Genet. 46, 486–491 (1990).
Schutte, B.C. et al. A preliminary gene map for the Van der Woude syndrome critical region derived from 900 kb of genomic sequence at 1q32-q41. Genome Res. 10, 81–94 (2000).
Van der Woude, A. Fistula labii inferioris congenita and its association with cleft lip and palate. Am. J. Hum. Genet. 6, 244–256 (1954).
Gorlin, R.J., Sedano, H.O. & Cervenka, J. Popliteal pterygium syndrome. A syndrome comprising cleft lip-palate, popliteal and intercrural pterygia, digital and genital anomalies. Pediatrics 41, 503–509 (1968).
Bixler, D., Poland, C. & Nance, W.E. Phenotypic variation in the popliteal pterygium syndrome. Clin. Genet. 4, 220–228 (1973).
Lees, M.M., Winter, R.M., Malcolm, S., Saal, H.M. & Chitty, L. Popliteal pterygium syndrome: a clinical study of three families and report of linkage to the Van der Woude syndrome locus on 1q32. J. Med. Genet. 36, 888–892 (1999).
Sachidanandam, R. et al. A map of human genome sequence variation containing 1.42 million single nucleotide polymorphisms. Nature 409, 928–933 (2001).
Fraser, F.C. Thoughts on the etiology of clefts of the palate and lip. Acta Genetica 5, 358–369 (1955).
Bocian, M. & Walker, A.P. Lip pits and deletion 1q32–q41. Am. J. Med. Genet. 26, 437–443 (1987).
Sander, A., Schmelzle, R. & Murray, J. Evidence for a microdeletion in 1q32–41 involving the gene responsible for Van der Woude syndrome. Hum. Mol. Genet. 3, 575–578 (1994).
Schutte, B.C. et al. Microdeletions at chromosome bands 1q32-q41 as a cause of Van der Woude syndrome. Am. J. Med. Genet. 84, 145–150 (1999).
Eroshkin, A. & Mushegian, A. Conserved transactivation domain shared by interferon regulatory factors and Smad morphogens. J. Mol. Med. 77, 403–405 (1999).
Mamane, Y. et al. Interferon regulatory factors: the next generation. Gene 237, 1–14 (1999).
Au, W.C., Yeow, W.S. & Pitha, P.M. Analysis of functional domains of interferon regulatory factor 7 and its association with IRF-3. Virology 280, 273–282 (2001).
Escalante, C.R., Yie, J., Thanos, D. & Aggarwal, A.K. Structure of IRF-1 with bound DNA reveals determinants of interferon regulation. Nature 391, 103–106 (1998).
Lin, R., Heylbroeck, C., Genin, P., Pitha, P.M. & Hiscott, J. Essential role of interferon regulatory factor 3 in direct activation of RANTES chemokine transcription. Mol. Cell Biol. 19, 959–966 (1999).
van den Boogaard, M.J., Dorland, M., Beemer, F.A. & van Amstel, H.K. MSX1 mutation is associated with orofacial clefting and tooth agenesis in humans. Nature Genet. 24, 342–343 (2000).
Celli, J. et al. Heterozygous germline mutations in the p53 homolog p63 are the cause of EEC syndrome. Cell 99, 143–153 (1999).
McGrath, J.A. et al. Hay-Wells syndrome is caused by heterozygous missense mutations in the SAM domain of p63. Hum. Mol. Genet. 10, 221–229 (2001).
Machin, G.A. Some causes of genotypic and phenotypic discordance in monozygotic twin pairs. Am. J. Med. Genet. 61, 216–228 (1996).
Brivanlou, A.H. & Darnell, J.E., Jr. Signal transduction and the control of gene expression. Science 295, 813–818 (2002).
Fitzpatrick, D.R., Denhez, F., Kondaiah, P. & Akhurst, R.J. Differential expression of TGF β isoforms in murine palatogenesis. Development 109, 585–595 (1990).
Proetzel, G. et al. Transforming growth factor-β3 is required for secondary palate fusion. Nature Genet. 11, 409–414 (1995).
Sanford, L.P. et al. TGFβ2 knockout mice have multiple developmental defects that are non-overlapping with other TGFβ knockout phenotypes. Development 124, 2659–2670 (1997).
Matzuk, M.M. et al. Functional analysis of activins during mammalian development. Nature 374, 354–356 (1995).
Kaartinen, V. et al. Abnormal lung development and cleft palate in mice lacking TGF-β 3 indicates defects of epithelial–mesenchymal interaction. Nature Genet. 11, 415–421 (1995).
Dixon, J., Hovanes, K., Shiang, R. & Dixon, M.J. Sequence analysis, identification of evolutionary conserved motifs and expression analysis of murine tcof1 provide further evidence for a potential function for the gene and its human homologue, TCOF1. Hum. Mol. Genet. 6, 727–737 (1997).
Nieto, M.A., Patel, K. & Wilkinson, D.G. In situ hybridization analysis of chick embryos in whole mount and tissue sections. Methods Cell Biol. 51, 219–235 (1996).
Acknowledgements
We thank our many clinical colleagues and their patients for contributing samples for this study (N. Akarsu, M. Aldred, Z. Ali-Khan, W.P. Allen, L. Bartoshesky, B. Bernhard, E. Bijlsma, E. Breslau-Siderius, C. Brewer, L. Brueton, B. Burton, J. Canady, A. Chakravarti, K. Chen, J. Clayton-Smith, M. Cunningham, A. David, B.B.A. de Vries, F.R. Desposito, K. Devriendt, R. Falk, J.-P. Fryns, R.J.M. Gardner, M. Golahi, J. Graham, M. Greenstein, M. Hannibal, E. Hauselman, R. Hennekam, G. Hoganson, L. Holmes, J. Hoogeboom, E. Hoyme, S. Kirkpatrick, J. Klein, T.C. Matise, L. Meisner, Z. Miedzybrodzka, J. Mulliken, A. Newlin, R. Pauli, W. Reardon, S. Roberts, H. Saal, A. Schinzel, J. Siegel-Bartelt, D. Sternen, V. Sybert, D. Tiziani, M.-P. Vazquez, L. Williamson-Kruse, F. Wilt, C. Yardin and K. Yoshiura). We appreciate the advice of K. Buetow, J. Dixon and C. Baldock; technical assistance from S. Hoper, M. Malik, J. Allaman, C. Hamm, N. Rorick, C. Nishimura, B. Ludwig, M. Fang, P. Hemerson, A. Westphalen and S. Lilly; administrative support from K. Krahn, D. Benton and L. Muilenburg; and sharing of unpublished results by P. Jezewski, A. Grossman and T.W. Mak. This work was supported by grants from the US National Institutes of Health and by grants to M.J.D. from Wellcome Trust, Action Research, Biotechnology and Biological Sciences Research Council, The European Union and the Fundação Lucentis (R.L.L.F.L. & D.M.F.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Kondo, S., Schutte, B., Richardson, R. et al. Mutations in IRF6 cause Van der Woude and popliteal pterygium syndromes. Nat Genet 32, 285–289 (2002). https://doi.org/10.1038/ng985
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng985
This article is cited by
-
Association of single nucleotide polymorphisms in the IRF6 gene with nonsyndromic cleft lip with or without cleft palate in Kinh Vietnamese patients
Molecular Biology Reports (2023)
-
Novel de novo missense mutation in the interferon regulatory factor 6 gene in an Italian infant with IRF6-related disorder
Italian Journal of Pediatrics (2022)
-
Genetic markers for non-syndromic orofacial clefts in populations of European ancestry: a meta-analysis
Scientific Reports (2022)
-
The genetic factors contributing to the risk of cleft lip-cleft palate and their clinical utility
Oral and Maxillofacial Surgery (2022)
-
FACEts of mechanical regulation in the morphogenesis of craniofacial structures
International Journal of Oral Science (2021)