Abstract
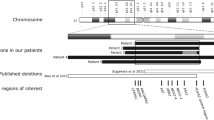
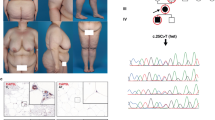
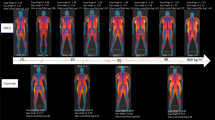
Congenital generalized lipodystrophy, or Berardinelli–Seip syndrome (BSCL), is a rare autosomal recessive disease characterized by a near-absence of adipose tissue from birth or early infancy and severe insulin resistance1,2,3,4. Other clinical and biological features include acanthosis nigricans, hyperandrogenism, muscular hypertrophy, hepatomegaly, altered glucose tolerance or diabetes mellitus, and hypertriglyceridemia. A locus (BSCL1) has been mapped to 9q34 with evidence of heterogeneity5. Here, we report a genome screen of nine BSCL families from two geographical clusters (in Lebanon and Norway). We identified a new disease locus, designated BSCL2, within the 2.5-Mb interval flanked by markers D11S4076 and D11S480 on chromosome 11q13. Analysis of 20 additional families of various ethnic origins led to the identification of 11 families in which the disease cosegregates with the 11q13 locus; the remaining families provide confirmation of linkage to 9q34. Sequence analysis of genes located in the 11q13 interval disclosed mutations in a gene homologous to the murine guanine nucleotide-binding protein (G protein), γ3-linked gene6 (Gng3lg) in all BSCL2-linked families. BSCL2 is most highly expressed in brain and testis and encodes a protein (which we have called seipin) of unknown function. Most of the variants are null mutations and probably result in a severe disruption of the protein. These findings are of general importance for understanding the molecular mechanisms underlying regulation of body fat distribution and insulin resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Berardinelli, W. An undiagnosed endocrinometabolic syndrome: report of two cases. J. Clin. Endocrinol. Metab. 14, 193–204 (1954).
Seip, M. Lipoatrophy and gigantism with associated endocrine manifestations: a new diencephalic syndrome? Acta Paediatr. Scand. 48, 555–574 (1959).
Moller, D.E. & O'Rahilly, S. Syndromes of severe insulin resistance: clinical and pathophysiological features. in Insulin resistance (ed. Moller, D.E.) 49–81 (Wiley and Sons, New York, 1993).
Seip, M. & Trygstad, O. Generalized lipodystrophy, congenital and acquired (lipoatrophy). Acta Paediatr. Suppl. 413, 2–28 (1996).
Garg, A. et al. A gene for congenital generalized lipodystrophy maps to human chromosome 9q34. J. Clin. Endocrinol. Metab. 84, 3390–3394 (1999).
Downes, G.B., Copeland, N.G., Jenkins, N.A. & Gautam, N. Structure and mapping of the G protein gamma3 subunit gene and a divergently transcribed novel gene, gng3lg. Genomics 53, 220–230 (1998).
Lander, E.S. & Botstein, D. Homozygosity mapping: a way to map human recessive traits with the DNA of inbred children. Science 236, 1567–1570 (1987).
Dib, C. et al. A comprehensive genetic map of the human genome based on 5,264 microsatellites. Nature 380, 152–154 (1996).
Gedde-Dahl, T. Jr. et al. Genetics of the Berardinelli-Seip syndrome (congenital generalized lipodystrophy) in Norway: epidemiology and gene mapping. Berardinelli- Seip Study Group. Acta Paediatr. Suppl. 413, 52–58 (1996).
Yu, W. et al. Large-scale concatenation cDNA sequencing. Genome Res. 7, 353–358 (1997).
Inoue, S., Sano, H. & Ohta, M. Growth suppression of Escherichia coli by induction of expression of mammalian genes with transmembrane or ATPase domains. Biochem. Biophys. Res. Commun. 268, 553–561 (2000).
Adams, M.D. et al. The genome sequence of Drosophila melanogaster. Science 287, 2185–2195 (2000).
Lawrence, R.D. Lipodystrophy and hepatomegaly with diabetes, lipemia and other metabolic disturbances. Lancet 1, 724–741 (1946).
Corbin, A., Upton, G.V., Mabry, C.C. & Hollingsworth, D.R. Diencephalic involvement in generalized lipodystrophy: rationale and treatment with the neuroleptic agent, pimozide. Acta Endocrinol. (Copenh.) 77, 209–220 (1974).
Mabry, C.C. & Hollingsworth, D.R. Failure of hypophysectomy in generalized lipodystrophy. J. Pediatr. 81, 990–992 (1972).
Mabry, C.C., Hollingsworth, D.R., Upton, G.V. & Corbin, A. Pituitary-hypothalamic dysfunction in generalized lipodystrophy. J. Pediatr. 82, 625–633 (1973).
Galli, J. et al. Genetic analysis of non-insulin dependent diabetes mellitus in the GK rat. Nature Genet. 12, 31–37 (1996).
Gauguier, D. et al. Chromosomal mapping of genetic loci associated with non-insulin dependent diabetes in the GK rat. Nature Genet. 12, 38–43 (1996).
Watanabe, T.K. et al. Genetic dissection of “OLETF,” a rat model for non-insulin-dependent diabetes mellitus: quantitative trait locus analysis of (OLETF x BN) x OLETF. Genomics 58, 233–239 (1999).
Vigouroux, C. et al. Lamin A/C gene: sex-determined expression of mutations in Dunnigan-type familial partial lipodystrophy and absence of coding mutations in congenital and acquired generalized lipoatrophy. Diabetes 49, 1958–1962 (2000).
van der Vorm, E.R. et al. Patients with lipodystrophic diabetes mellitus of the Seip-Berardinelli type, express normal insulin receptors. Diabetologia 36, 172–174 (1993).
Krook, A. et al. Molecular scanning of the insulin receptor gene in syndromes of insulin resistance. Diabetes 43, 357–368 (1994).
Vigouroux, C. et al. Genetic exclusion of 14 candidate genes in lipoatropic diabetes using linkage analysis in 10 consanguineous families. J. Clin. Endocrinol. Metab. 82, 3438–3444 (1997).
Huseman, C., Johanson, A., Varma, M. & Blizzard, R.M. Congenital lipodystrophy: An endocrine study in three siblings. I. Disorders of carbohydrate metabolism. J. Pediatr. 93, 221–226 (1978).
Panz, V.R., Raal, F.J., O'Rahilly, S., Kedda, M.A. & Joffe, B.I. Insulin receptor substrate-1 gene variants in lipoatrophic diabetes mellitus and non-insulin-dependent diabetes mellitus: a study of South African black and white subjects. Hum. Genet. 101, 118–119 (1997).
Van Maldergem, L. et al. Syndrome of lipoatrophic diabetes, vitamin D resistant rickets, and persistent Mullerian ducts in a Turkish boy born to consanguineous parents. Am. J. Med. Genet. 64, 506–513 (1996).
Sambrook, J., Fritsch, E.F. & Maniatis, T. Molecular cloning: a laboratory manual., 9.17–9.19 (Cold Spring Harbor Laboratory Press, New York, 1989).
Stewart, E.A. et al. An STS-based radiation hybrid map of the human genome. Genome Res. 7, 422–433 (1997).
Sobel, E. & Lange, K. Descent graphs in pedigree analysis: applications to haplotyping, location scores, and marker-sharing statistics. Am. J. Hum. Genet. 58, 1323–1337 (1996).
Ewing, B., Hillier, L., Wendl, M.C. & Green, P. Base-calling of automated sequencer traces using phred. I. Accuracy assessment. Genome Res. 8, 175–185 (1998).
Acknowledgements
We thank the patients and their families for their participation; all the contributors for the families' collections, including the Medical Genetic Unit in Beyrouth; A. Lemainque, A. Faure, V. La Villa, S. Pavek, F. Cavallin and L. Bodson for genotyping; R. Schaaning Jörgensen and A. Gjesti for their technical assistance; P. Balladur, M. Chawatt and their collaborators for adipose tissues; B. Surin for quantification of RNA expression; D. Recan and her colleagues for immortalization of lymphocytes; Y. Chrétien, N. Chignard, L. Grivot, B. Jacquin and C. Lestringuez for computer assistance; and B. Guilmart for critical reading of the manuscript. This work was supported by grants from Aide aux Jeunes Diabétiques, Anders Jahres Fond, Assistance Public-Hôpitaux de Paris, the European Commission (contract BMH4-CT 97-4841), INSERM, Medinnova and the Ministère de la Recherche. This article is dedicated to the memory of Martin Seip (1921–2001).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Magré, J., Delépine, M., Khallouf, E. et al. Identification of the gene altered in Berardinelli–Seip congenital lipodystrophy on chromosome 11q13. Nat Genet 28, 365–370 (2001). https://doi.org/10.1038/ng585
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng585
This article is cited by
-
Rab18 maintains homeostasis of subcutaneous adipose tissue to prevent obesity-induced metabolic disorders
Science China Life Sciences (2024)
-
Beyond energy provider: multifunction of lipid droplets in embryonic development
Biological Research (2023)
-
Clinical genetics of Charcot–Marie–Tooth disease
Journal of Human Genetics (2023)
-
Lipid droplet biogenesis and functions in health and disease
Nature Reviews Endocrinology (2023)
-
The metabolic effects of APOL1 in humans
Pflügers Archiv - European Journal of Physiology (2023)