Abstract
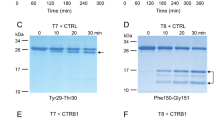
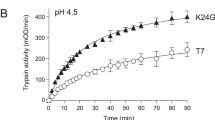
Hereditary pancreatitis (HP) is a rare, early-onset genetic disorder characterized by epigastric pain and often more serious complications. We now report that an Arg–His substitution at residue 117 of the cationic trypsinogen gene is associated with the HP phenotype. This mutation was observed in all HP affected individuals and obligate carriers from five kindreds, but not in individuals who married into the families nor in 140 unrelated individuals. X-ray crystal structure analysis, molecular modelling, and protein digest data indicate that the Arg 117 residue is a trypsin-sensitive site. Cleavage at this site is probably part of a fail-safe mechanism by which trypsin, which is activated within the pancreas, may be inactivated; loss of this cleavage site would permit autodigestion resulting in pancreatitis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Perrault, J. Hereditary pancreatitis. Gastroenterol. Clin. North Am. 23, 743–752 (1994).
Madraso-de la Garza, J., Hill, I. & Lebenthal, E. Hereditary pancreatitis. in The Pancreas: Biology, Pathobiology, and Disease. 2nd ed. (ed. Go, V.) 1095–1101 (New York: Raven, 1993).
Whitcomb, D.C. et al. A gene for hereditary pancreatitis maps to chromosome 7q35. Gastroenterology 110, 1975–1980 (1996).
Bodic, L.L. et al. The hereditary pancreatitis gene maps to long arm of chromosome 7. Hum. Mol. Genet. 5, 549–554 (1996).
Comfort, M. & Steinberg, A. Pedigree of a family with hereditary chronic relapsing pancreatitis. Gastroenterology 21, 54 (1952).
Chiara, H. Ueber selbstverdauung des menschlichen pankreas. Zeitschrift fur heilkunde 17, 69–96 (1896).
Steer, M.L. & Meldolesi, J. The cell biology of experimental pancreatitis. N. Engl. J. Med. 316, 144–50 (1987).
Stewart, E.A., Craik, C.S., Hake, L. & Bowcock, A.M. Human carboxypeptidase A identifies a BgLII RFLP and maps to 7q31–qter. Am. J. Hum. Genet 46, 795–800, (1990).
Rommens, J.M. et al. Identification and regional localization of DNA markers on chromosome 7 for the cloning of the cystic fibrosis gene. Am. J. Hum. Genet. 43, 645–663 (1988).
Rommens, J.M. et al. Identification of the cystic fibrosis gene: chromosome walking and jumping. Science 245, 1059–1065 (1989).
Martise, T.C., Perlin, M. & Chakravarti, A. Automated construction of genetic linkage maps using an expert system (MultiMap): a human genome linkage map. Nature Genet. 6, 384–390 (1994).
Rowen, L., Koop, B.F. & Hood, L. The Complete 685-Kilobase DNA Sequence of the Human βT Cell Receptor Locus. Science (in the press).
Emi, M. et al. Cloning, characterization and nucleotide sequences of two cDNAs encoding human pancreatic trypsinogens. Gene 41, 305–310 (1986).
Tani, T., Kawashima, I., Mita, K. & Takiguchi, Y. Nucleotide sequence of the human pancreatic tryspinogen III cDNA. Nucl. Acids Res. 18, 1631 (1990).
Weingand, U., Corbach, S., Minn, A., Kang, J. & Muller-Hill, B. Cloning of the cDNA encoding human brain trypsinogen and characterization of its product. Gene 136, 167–175 (1993).
McElroy, R. & Christiansen, P.A. Hereditary pancreatitis in a kinship association with portal vein thrombosis. Am. J. Med. 52, 228–241 (1972).
Cucchiara, S., Staiano, A., Minella, R., Uomo, G. & Cipolletta, L., Hereditary Pancreatitis: Report of the second kindred in Italy. J. Ped. Gastro. Nutr. 3, 422–425 (1990).
Bernstein, F.C. et al. The Protein Data Bank: a computer-based archival file for macromolecular structures. J. Mol. Biol. 112, 535–542 (1977).
Bolognesi, M. et al. Three-dimensional structure of the complex between pancreatic secretory trypsin inhibitor (Kazal type) and trypsinogen at 1. 8 Å resolution. Structure solution, crystallographic refinement and preliminary structural interpretation. J. Mol. Biol. 162, 839–868 (1982).
Huber, R., Bode, W., Deisenhofer, J. & Schwager, P. Trypsinogen complex with pancreatic trypsin inhibitor and Ile-Val. NCBI generated Kinemage File, Molecular Modeling Data Base Accesion ♯3116
Higaki, J.N. & Light, A. The identification of neotrypsinogens in samples of bovine trypsinogen. Anal. Biochem. 148, 111–120 (1985).
Rovery, M. Limited proteolyses in pancreatic chymotrypsinogens and trypsinogens. Biochemie 70, 1131–1135 (1988).
Higaki, J.N. & Light, A. Independent refolding of domains in the pancreatic serine proteinases. J. Biol. Chem. 261, 10606–10609 (1986).
Rinderknecht, H., Renner, I.G., Abramson, S.B. & Carmack, C. Mesotrypsin: a new inhibitor-resistant protease from a zymogen in human pancreatic tissue and fluid. Gastroenterology 86, 681–692 (1984).
Rinderknecht, H. Activation of Pancreatic Zymogens: Normal Activation, Premature Intrapancreatic Activation, Protective Mechanisms Against Inappropriate Activation. Dig. Dis. Sci. 31, 314–321 (1986).
Rinderknecht, H., Adham, N.F., Renner, I.G. & Carmack, C. A possible self-destruct mechanism preventing pancreatic autodigestion. Int. J. Pancreatol. 3, 33–34 (1988).
Rinderknecht, H. Pancreatic secretory enzymes. in The Pancreas: Biology, Pathobiology, and Disease 2nd edn. (eds Go, V.L. et al.) 219–251 (Raven Press, New York, 1993).
Sweet, R.M., Wright, H.T., Janin, J., Chothia, C.H. & Blow, D.M. Crystal structure of the complex of porcine trypsin with soybean trypsin inhibitor (Kunitz) at 2. 6-Å resolution. Biochemistry 13, 4212–4228 (1974).
Laskowski, M. Jr. & Ikunoshin, K. Protein inhibitors of proteinases. Annu. Rev. Biochem. 49, 593–626 (1980).
Yamamoto, T. et al. Molecular cloning and nucleotide sequence of human pancreatic secretory trypsin inhibitor (PSTI) cDNA. Biochem. Biophys. Res. Commun. 132, 605–612 (1985).
Dassell, B. & Kay, J. Zymogens of proteolytic enzymes. Science 180, 1022–1027 (1973).
Bieger, W. & Scheele, G. Two-dimensional isoelectric focusing/sodium dodecylsulfate gel electrophoresis of protein mixtures containing active or potentially active proteases. Analysis of human exocrine pancreatic proteins. Anal. Biochem. 109, 222–230 (1980).
Figarella, C., Amouric, M. & Guy-Crotee, O. Proteolysis of human trypsinogen. I. Pathogenetic implications in chronic pancreatitis. Biochem. Biophys. Res. Commun. 118, 154–161 (1984).
Gorry, M.C. et al. Crouzon Syndrom: Mutations in wo spliceforms of FGFR2 and a common point mutation shared with Jackson-Weiss Syndrome. Hum. Mol. Genet. 4, 1387–1390 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Whitcomb, D., Gorry, M., Preston, R. et al. Hereditary pancreatitis is caused by a mutation in the cationic trypsinogen gene. Nat Genet 14, 141–145 (1996). https://doi.org/10.1038/ng1096-141
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1096-141
This article is cited by
-
Fibronectin fragments generated by pancreatic trypsin act as endogenous inhibitors of pancreatic tumor growth
Journal of Experimental & Clinical Cancer Research (2023)
-
Expanding ACMG variant classification guidelines into a general framework
Human Genomics (2022)
-
Trypsinogen (PRSS1 and PRSS2) gene dosage correlates with pancreatitis risk across genetic and transgenic studies: a systematic review and re-analysis
Human Genetics (2022)
-
Pathogenese der akuten Pankreatitis
Wiener klinisches Magazin (2022)
-
Diagnostik und klinisches Management prämaligner Erkrankungen des Pankreas
Der Internist (2022)