Abstract
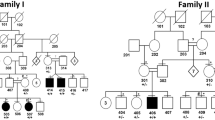
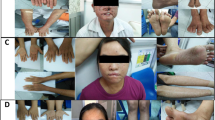
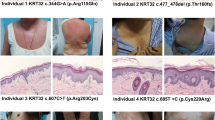
White sponge nevus (WSN) is a benign autosomal dominant disorder which affects non-cornifying stratified squamous epithelia (MIM 193900) (ref. 1). Phenotypically it presents as white ‘spongy’ plaques (oral leukokeratoses), most commonly in the mouth but also reported in the esophagus and anogenital mucosa2. Histologically, the plaques show evidence of hyperproliferation, acanthosis and tonofilament aggregation3–5. These types of pathogenic changes are characteristic of many of the epidermal keratin disorders6. Keratins are expressed in pairs by epithelial cells in a tissue and cell specific manner. The major differentiation specific keratins of the buccal mucosa, nasal, esophageal and anogenital epithelia are K4 and K13 (ref. 7). The tissue distribution and nature of the lesions in patients affected by WSN suggested that mutations in K4 and/or K13 might be responsible for this disorder. We have now confirmed this hypothesis and report here a three base-pair (bp) deletion in the helix initiation peptide of K4 in affected members from two families with this condition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
McKusick, V.A. OMIM (On-line Mendelian Inheritance in Man) (Johns Hopkins University, Baltimore, 1995).
Jorgenson, R.J. & Levin, S. White sponge nevus. Arch. Dematol. 117, 73–76 (1981).
Aloi, F.G. & Molinero, A. White sponge nevus with epidermolytic changes. Dermatologica 177, 323–326 (1988).
McGinnis, J.P. & Turner, J.E. Ultrastructure of the white sponge nevus. Oral Surg. 40, 644–651 (1975).
Frithiof, L & Banoczy, J White sponge nevus (leukoedema exfoliativum mucosae oris): ultrastructural observations. Oral Surg. 41, 607–622 (1976).
McLean, W.H.I. & Lane, E.B. Intermediate filaments in disease. Curr. Opin. cell. Biol. 7, 118–125 (1995).
van Muijen, G.N.P. et al. Cell type heterogeneity of cytokeratin expression in complex epithelia and carcinomas as demonstrated by monoclonal antibodies specific for cytokeratins nos 4 and 13. Exp. Cell Res. 162, 97–113 (1986).
Hatzfeld, M. & Weber, K. Modulation of keratin intermediate filament assembly by single amino acid exchanges in the consensus sequence at the C-terminal end of the rod domain. J. Cell Sci. 99, 351–362 (1991).
Letai, A. et al. Disease severity correlates with position of keratin point mutations in patients with epidermolysis bullosa simplex. Proc. natn. Acad. Sci. U.S.A. 90, 3197–3201 (1993).
Leube, R.E. et al. Molecular characterization and expression of the stratification-related cytokeratins 4 and 15. J. Cell Biol. 106, 1249–1261 (1988).
Mischke, D., Wachter, E., Hochstrasser, K., Wild, A.G. & Schulz, P. The N-, but not the C-terminal domains of human keratins 13 and 15 are closely related. Nucl. Acids Res. 17, 7984 (1989).
Cooper, D.N. & Krawczak, M. Hum. Gene Mutat. (BIOS Scientific Publishers Ltd., Oxford, 1993).
Bowden, P.E. et al. Mutation of a type II keratin gene (K6a) in pachyonychia congenita. Nature Genet. 10, 363–365 (1995).
Yoon, S.-J., LeBlanc-Straceski, J., Ward, D., Krauter, K. & Kucherlapati, R. Organization of the human keratin type II gene cluster at 12q13. Genomics 24, 502–508 (1994).
McLean, W.H.I., Sultan, N., Parfitt, E. & Lane, E.B. Polymorphisms in the keratin 8 gene detected by PCR. Hum. molec. Genet. 3, 1031 (1994).
Gyapay, G. et al The 1993–94 Généthon human linkage map. Nature Genet. 7, 246–339 (1994).
McLean, W.H.I. et al. Keratin 16 and keratin 17 mutations cause pachyonychia congenita. Nature Genet. 9, 273–278 (1995).
McDonagh, A.J., Gawkrodger, D.J. & Walker, A.E. White sponge nevus successfully treated with topical tetracycline. Clin. exp. Dermatol. 15, 152–153 (1990).
Lane, E.B. Keratin diseases. Curr. Op. Genet. Dev. 4, 412–418 (1994).
Lane, E.B. et al. A mutation in the conserved helix termination peptide of keratin 5 in hereditary skin blistering. Nature 356, 244–246 (1992).
Richard, G. et al. Keratin 13 point mutation underlies the hereditary mucosal epithelia disorder white sponge nevus. Nature Genet. 11, 453–455 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rugg, E., McLean, W., Allison, W. et al. A mutation in the mucosal keratin K4 is associated with oral white sponge nevus. Nat Genet 11, 450–452 (1995). https://doi.org/10.1038/ng1295-450
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1295-450
This article is cited by
-
Expression profiling of white sponge nevus by RNA sequencing revealed pathological pathways
Orphanet Journal of Rare Diseases (2015)
-
Genome-wide analysis of the mouse lung transcriptome reveals novel molecular gene interaction networks and cell-specific expression signatures
Respiratory Research (2011)