Abstract
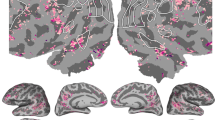
Neurobiological theories of knowledge processing are biased toward the language-dominant (usually the left) hemisphere. Does the right hemisphere critically contribute to knowledge processing? J.A. is a left-hemisphere language-dominant individual who suffered a lesion confined to the right mid- and anterior fusiform gyrus. Although her language abilities are intact, she showed a partial loss of knowledge of the visual attributes of biological and nonbiological entities. This was observed regardless of the task performed: object discrimination, oral feature generation, forced-choice naming-to-definition or free-hand drawing. Functional-associative and nonvisual sensory attributes were spared. The same region that was lesioned in J.A. was activated in a functional magnetic resonance imaging study in 27 volunteers who retrieved semantic associations between concepts, but only if the concepts were represented as pictures and not as words. Therefore, right fusiform gyrus critically contributes to the conscious recollection of visual attributes of familiar entities.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Plato . Phaedo <http://classics.mit.edu/Plato/phaedo.html> (1994).
Saffran, E.M. & Sholl, A. Clues to the functional and neural architecture of word meaning. in The Neurocognition of Language (eds. Brown, C.M. & Hagoort, P.) Ch. 8 241–272 (Oxford University Press, Oxford, 1999).
Caramazza, A. The organization of conceptual knowledge in the brain. in The New Cognitive Neurosciences (ed. Gazzaniga, M.) 1037–1046 (MIT Press, Cambridge, MA, 2000).
Sartori, G. & Job, R. The oyster with four legs: a neuropsychological study on the interaction between vision and semantic information. Cogn. Neuropsychol. 5, 105–132 (1988).
Luzzatti, C. & Davidoff, J. Impaired retrieval of object-colour knowledge with preserved colour naming. Neuropsychologia 32, 933–950 (1994).
Breedin, S.D., Saffran, E.M. & Coslett, H.B. Reversal of the concreteness effet in a patient with semantic dementia. Cogn. Neuropsychol. 11, 617–660 (1994).
Carbonnel, S., Charnallet, A., David, D. & Pellat, J. One or several semantic systems? Maybe none: evidence from a case study of modality and category-specific “semantic” impairment. Cortex 33, 391–417 (1997).
Forde, E.M.E., Francis, D., Riddoch, M.J., Rumiati, R.I. & Humphreys, G.W. On the links between visual knowledge and naming: a single case study of a patient with a category-specific impairment for living things. Cogn. Neuropsychol. 14, 403–458 (1997).
Miceli, G. et al. The dissociation of color from form and function knowledge. Nat. Neurosci. 4, 662–667 (2001).
Warrington, E. & Shallice, T. Category specific semantic impairments. Brain 107, 829–853 (1984).
Hillis, A.E. & Caramazza, A. Category-specific naming and comprehension impairment: a double dissociation. Brain 114, 2081–2094 (1991).
Hart, J. & Gordon, B. Neural subsystems for object knowledge. Nature 359, 60–64 (1992).
Farah, M. The neurological basis of mental imagery: a componential analysis. Cognition 18, 245–272 (1984).
Sergent, J. The neuropsychology of visual image generation: data, method, and theory. Brain Cogn. 13, 98–129 (1990).
Kosslyn, S.M. & Pomerantz, J.R. Imagery, propositions, and the form of internal representations. Cogn. Psychol. 9, 52–76 (1977).
Ishai, A., Ungerleider, L.G. & Haxby, J.V. Distributed neural systems for the generation of visual images. Neuron 28, 979–990 (2000).
Shallice, T. From Neuropsychology to Mental Structure (Cambridge University Press, Cambridge, UK, 1988).
Marr, D. Vision (Freeman, San Francisco, 1982).
Allport, D.A. Distributed memory, modular subsystems and dysphasia. in Current Perspectives in Dysphasia (eds. Newman, S.K. & Epstein, R.) 32–60 (Churchill Livingstone, Edinburgh, 1985).
Warrington, E.K. & McCarthy, R. Categories of knowledge: further fractionations and an attempted integration. Brain 110, 1273–1296 (1987).
Lauro-Grotto, R., Piccini, C. & Shallice, T. Modality-specific operations in semantic dementia. Cortex 33, 593–622 (1997).
Humphreys, G.W. & Forde, E.M.E. Hierarchies, similarity and interactivity in object recognition. Behav. Brain Sci. 24, 453–476 (2001).
Riddoch, J. & Humphreys, G. Visual object processing in optic aphasia: a case of semantic access agnosia. Cogn. Neuropsychol. 4, 131–185 (1987).
Hadjikhani, N. & de Gelder, B. Neural basis of prosopagnosia: an fMRI study. Hum. Brain Mapp. 16, 176–182 (2002).
Kourtzi, Z. & Kanwisher, N. Representation of perceived object shape by the human lateral occipital complex. Science 293, 1506–1509 (2001).
Hadjikhani, N., Liu, A.K., Dale, A.M., Cavanagh, P. & Tootell, R.B.H. Retinotopy and color sensitivity in human visual cortical area V8. Nat. Neurosci. 1, 235–241 (1998).
Riddoch, M.J. & Humphreys, G.W. Birmingham Object Recognition Battery (Lawrence Erlbaum Associates, Hove, UK, 1993).
Warrington, E.K. & James, M. Visual Object and Space Perception Battery (Thames Valley Test Company, Bury St. Edmunds, UK, 1991).
Farah, M. Visual Agnosia (MIT Press, Cambridge, Massachusetts, 2004).
Ruts, W. et al. Dutch norm data for 13 semantic categories and 338 exemplars. Behav. Res. Methods Instrum. Comput. 36, 506–515 (2004).
Crawford, J.R. & Garthwaite, P.H. Testing for suspected impairments and dissociations in single-case studies in neuropsychology: evaluation of alternatives using Monte Carlo simulations and revised tests for dissociations. Neuropsychology 19, 318–331 (2005).
Powell, J. & Davidoff, J. Selective impairments of object knowledge in a case of acquired color blindness. Memory 3, 435–461 (1995).
Faglioni, P. & Botti, C. How to differentiate retrieval from storage deficit: a stochastic approach to semantic memory modeling. Cortex 29, 501–518 (1993).
Laiacona, M. & Capitani, E. A case of prevailing deficit of nonliving categories or a case of prevailing sparing of living categories. Cogn. Neuropsychol. 18, 39–70 (2001).
Vandenberghe, R., Price, C.J., Wise, R., Josephs, O. & Frackowiak, R.S.J. Functional anatomy of a common semantic system for words and pictures. Nature 383, 254–256 (1996).
Chao, L.L., Haxby, J.V. & Martin, A. Attribute-based neural substrates in temporal cortex for perceiving and knowing about objects. Nat. Neurosci. 2, 913–919 (1999).
Barsalou, L.W. Perceptual symbol systems. Behav. Brain Sci. 22, 577–609 (1999).
Pylyshyn, Z. Seeing and Visualizing: It is Not What You Think (MIT Press, Cambridge, Massachusetts, 2003).
Jackendoff, R. On beyond zebra: the relation of linguistic and visual information. Cognition 26, 89–114 (1987).
Paivio, A. Dual coding theory: retrospect and current status. Can. J. Psychol. 45, 255–287 (1991).
Howard, D. & Patterson, K. Pyramids and Palm Trees: a Test of Semantic Access from Pictures and Words (Thames Valley Publishing, Bury St. Edmunds, UK, 1992).
McKenna, P. & Warrington, E.K. The neuropsychology of semantic memory. in Handbook of Neuropsychology Vol. 2 (eds. Boller, F. & Grafman, J.) Ch. 15 355–382 (Elsevier Science, Amsterdam, 2000).
McCarthy, R. & Warrington, E. Evidence for modality-specific meaning systems in the brain. Nature 334, 428–430 (1988).
Warrington, E. & McCarthy, R. Multiple meaning systems in the brain: a case for visual semantics. Neuropsychologia 32, 1465–1473 (1994).
Crawford, J.R. & Howell, D.C. Comparing an individual's test score against norms derived from small samples. Clin. Neuropsychol. 12, 482–486 (1998).
Snodgrass, J. & Vanderwart, M. A standardized set of 260 pictures: norms for name agreement, image agreement, familiarity and visual complexity. J. Exp. Psychol. [Hum. Learn.] 6, 174–215 (1980).
Vandenbulcke, M., Peeters, R., Van Hecke, P. & Vandenberghe, R. Anterior temporal laterality in primary progressive aphasia shifts to the right. Ann. Neurol. 58, 362–370 (2005).
Friston, K. et al. Statistical parametric maps in functional imaging: a general approach. Hum. Brain Mapp. 2, 189–210 (1995).
Denys, K. et al. The processing of visual shape in the cerebral cortex of human and non-human primates: an fMRI study. J. Neurosci. 24, 2551–2565 (2004).
Vanduffel, W. et al. Extracting 3D from motion: differences in human and monkey intraparietal cortex. Science 298, 413–415 (2002).
Acknowledgements
We thank M.M. Mesulam, G. Orban, G. Storms and R. Vogels for comments on earlier versions of the manuscript. Supported by Fund for Scientific Research (Flanders) (G0277.05), KU Leuven (OT/04/41) and the Medical Foundation Queen Elisabeth. R.V. is a clinical investigator of the Fund for Scientific Research (Flanders).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1
Neuropsychological profile in JA and MV. (PDF 63 kb)
Rights and permissions
About this article
Cite this article
Vandenbulcke, M., Peeters, R., Fannes, K. et al. Knowledge of visual attributes in the right hemisphere. Nat Neurosci 9, 964–970 (2006). https://doi.org/10.1038/nn1721
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1721
This article is cited by
-
Functional imaging analyses reveal prototype and exemplar representations in a perceptual single-category task
Communications Biology (2022)
-
Right Hemisphere Recruitment During Language Processing in Frontotemporal Lobar Degeneration and Alzheimer’s Disease
Journal of Molecular Neuroscience (2011)