Abstract
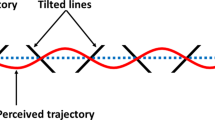
Eye movements markedly change the pattern of retinal stimulation. To maintain stable vision, the brain possesses a variety of mechanisms that compensate for the retinal consequences of eye movements. However, eye movements may also be important for resolving the ambiguities often posed by visual inputs, because motor commands contain additional spatial information that is necessarily absent from retinal signals. To test this possibility, we used a perceptually ambiguous stimulus composed of four line segments, consistent with a shape whose vertices were occluded. In a passive condition, subjects fixated a spot while the shape translated along a certain trajectory. In several active conditions, the spot, occluder and shape translated such that when subjects tracked the spot, they experienced the same retinal stimulus as during fixation. We found that eye movements significantly promoted perceptual coherence compared to fixation. These results indicate that eye movement information constrains the perceptual interpretation of visual inputs.
This is a preview of subscription content, access via your institution
Access options








Similar content being viewed by others
References
Bridgeman, B., Hendry, D. & Stark, L. Failure to detect displacement of the visual world during saccadic eye movements. Vision Res. 15, 719–722 (1975).
Grimes, J. On the failure to detect changes in scenes across saccades. in Perception 5 (Vancouver Studies in Cognitive Science) (ed. Akins, K.) 89–109 (Oxford Univ. Press, New York, 1996).
Henderson, J.M. & Hollingworth, A. Global transsaccadic change blindness during scene perception. Psychol. Sci. 14, 493–497 (2003).
McConkie, G.W. & Currie, C.B. Visual stability across saccades while viewing complex pictures. J. Exp. Psychol. Hum. Percept. Perform. 22, 563–581 (1996).
Burr, D.C., Morrone, M.C. & Ross, J. Selective suppression of the magnocellular visual pathway during saccadic eye movements. Nature 371, 511–513 (1994).
Irwin, D.E. & Brockmole, J.R. Suppressing where but not what: the effect of saccades on dorsal- and ventral-stream visual processing. Psychol. Sci. 15, 467–473 (2004).
Castet, E. & Masson, G.S. Motion perception during saccadic eye movements. Nat. Neurosci. 3, 177–183 (2000).
Currie, C.B., McConkie, G.W., Carlson-Radvansky, L.A. & Irwin, D.E. The role of the saccade target object in the perception of a visually stable world. Percept. Psychophys. 62, 673–683 (2000).
O'Regan, J.K. & Noe, A. A sensorimotor account of vision and visual consciousness. Behav. Brain Sci. 24, 939–1011 (2001).
Nakamura, K. & Colby, C.L. Updating of the visual representation in monkey striate and extrastriate cortex during saccades. Proc. Natl. Acad. Sci. USA 99, 4026–4031 (2002).
Duhamel, J.R., Colby, C.L. & Goldberg, M.E. The updating of the representation of visual space in parietal cortex by intended eye movements. Science 255, 90–92 (1992).
Walker, M.F., Fitzgibbon, E.J. & Goldberg, M.E. Neurons in the monkey superior colliculus predict the visual result of impending saccadic eye movements. J. Neurophysiol. 73, 1988–2003 (1995).
DeSouza, J.F.X., Dukelow, S.P. & Vilis, T. Eye position signals modulate early dorsal and ventral visual areas. Cereb. Cortex 12, 991–997 (2002).
Sylvester, R., Haynes, J.-D. & Rees, G. Saccades differentially modulate human LGN and V1 responses in the presence and absence of visual stimulation. Curr. Biol. 15, 37–41 (2005).
Krauzlis, R.J. & Stone, L.S. Tracking with the mind's eye. Trends Neurosci. 22, 544–550 (1999).
Steinbach, M.J. Pursuing the perceptual rather than the retinal stimulus. Vision Res. 16, 1371–1376 (1976).
Beutter, B.R. & Stone, L.S. Motion coherence affects human perception and pursuit similarly. Vis. Neurosci. 17, 139–153 (2000).
Lorenceau, J. & Shiffrar, M. The influence of terminators on motion integration across space. Vision Res. 32, 263–273 (1992).
Sommer, M.A. & Wurtz, R.H. What the brain stem tells the frontal cortex. I. Oculomotor signals sent from superior colliculus to frontal eye field via mediodorsal thalamus. J. Neurophysiol. 91, 1381–1402 (2004).
Sommer, M.A. & Wurtz, R.H. What the brain stem tells the frontal cortex. II. Role of the SC-MD-FEF pathway in corollary discharge. J. Neurophysiol. 91, 1403–1423 (2004).
Lorenceau, J. & Alais, D. Form constraints in motion binding. Nat. Neurosci. 4, 745–751 (2001).
Tadin, D., Lappin, J.S., Blake, R. & Grossman, E.D. What constitutes an efficient reference frame for vision? Nat. Neurosci. 5, 1010–1015 (2002).
McDermott, J., Weiss, Y. & Adelson, E.H. Beyond junctions: nonlocal form constraints on motion interpretation. Perception 30, 905–923 (2001).
McDermott, J. & Adelson, E.H. The geometry of the occluding contour and its effect on motion interpretation. J. Vis. 4, 944–954 (2004).
Wexler, M., Panerai, F., Lamouret, I. & Droulez, J. Self-motion and the perception of stationary objects. Nature 409, 85–88 (2001).
Hildreth, E.C. & Koch, C. The analysis of visual motion: from computational theory to neuronal mechanisms. Annu. Rev. Neurosci. 10, 477–533 (1987).
Shimojo, S., Silverman, G.H. & Nakayama, K. Occlusion and the solution to the aperture problem for motion. Vision Res. 29, 619–626 (1989).
Lorenceau, J., Shiffrar, M., Walls, N. & Castet, E. Different motion sensitive units are involved in recovering the direction of moving lines. Vision Res. 33, 1207–1217 (1993).
Mingolla, E., Todd, J.T. & Norman, J.F. The perception of globally coherent motion. Vision Res. 32, 1015–1031 (1992).
Rubin, N. & Hochstein, S. Isolating the effect of one-dimensional motion signals on the perceived direction of moving two-dimensional objects. Vision Res. 33, 1385–1396 (1993).
Ferrera, V.P. & Wilson, H.R. Perceived direction of moving two-dimensional patterns. Vision Res. 30, 273–287 (1990).
Lorenceau, J. Veridical perception of global motion from disparate component motions. Vision Res. 38, 1605–1610 (1998).
Ball, K. & Sekuler, R. Direction-specific improvement in motion discrimination. Vision Res. 27, 953–965 (1987).
Krukowski, A.E. & Stone, L.S. Expansion of direction space around the cardinal axes revealed by smooth pursuit eye movements. Neuron 45, 315–323 (2005).
Heeley, D.W. & Buchanan-Smith, H.M. Directional acuity for drifting plaids. Vision Res. 32, 97–104 (1992).
Gros, B.L., Blake, R. & Hiris, E. Anisotropies in visual motion perception: a fresh look. J. Opt. Soc. Am. A Opt. Image Sci. Vis. 15, 2003–2011 (1998).
Burr, D. Eye movements: keeping vision stable. Curr. Biol. 14, R195–R197 (2004).
Thier, P., Haarmeier, T., Chakraborty, S., Lindner, A. & Tikhonov, A. Cortical substrates of perceptual stability during eye movements. Neuroimage 14, S33–S39 (2001).
Khayat, P.S., Spekreijse, H. & Roelfsema, P.R. Correlates of transsaccadic integration in the primary visual cortex of the monkey. Proc. Natl. Acad. Sci. USA 101, 12712–12717 (2004).
Bedell, H.E., Chung, S.T.L. & Patel, S.S. Attenuation of perceived motion smear during vergence and pursuit tracking. Vision Res. 44, 895–902 (2004).
Souman, J.L., Hooge, I.T. & Wertheim, A.H. Perceived motion direction during smooth pursuit eye movements. Exp. Brain Res. 164, 376–386 (2005).
Souman, J.L., Hooge, I.T. & Wertheim, A.H. Vertical object motion during horizontal ocular pursuit: compensation for eye movements increases with presentation duration. Vision Res. 45, 845–853 (2005).
Li, H.-C.O., Brenner, E., Cornelissen, F.W. & Kim, E.-S. Systematic distortion of perceived 2D shape during smooth pursuit eye movements. Vision Res. 42, 2569–2575 (2002).
Brainard, D.H. The psychophysics toolbox. Spat. Vis. 10, 433–436 (1997).
Pelli, D.G. The VideoToolbox software for visual psychophysics: transforming numbers into movies. Spat. Vis. 10, 437–442 (1997).
Barlow, H.B. Eye movements during fixation. J. Physiol. (Lond.) 116, 290–306 (1952).
Wichmann, F.A. & Hill, N.J. The psychometric function: I. Fitting, sampling, and goodness of fit. Percept. Psychophys. 63, 1293–1313 (2001).
Wichmann, F.A. & Hill, N.J. The psychometric function: II. Bootstrap-based confidence intervals and sampling. Percept. Psychophys. 63, 1314–1329 (2001).
Acknowledgements
Supported by the Natural Sciences and Engineering Research Council of Canada, the Sloan-Swartz Center for Theoretical Neurobiology and the US National Institutes of Health (grant EY12212).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Exploring differential effects of eye movement direction relative to retinal motion direction. (PDF 221 kb)
Supplementary Fig. 2
Subjective experience of coherence was highest for orthogonal tracking. (PDF 140 kb)
Supplementary Fig. 3
Exploring differential effects of eye movement direction relative to retinal motion direction with a different shape. (PDF 181 kb)
Supplementary Fig. 4
Subjective experience of coherence was highest for horizontal tracking with the diamond stimulus of Supplementary Figure 3. (PDF 106 kb)
Rights and permissions
About this article
Cite this article
Hafed, Z., Krauzlis, R. Ongoing eye movements constrain visual perception. Nat Neurosci 9, 1449–1457 (2006). https://doi.org/10.1038/nn1782
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1782
This article is cited by
-
Action and attentional load can influence aperture effects on motion perception
Experimental Brain Research (2011)
-
Enactivism and the Unity of Perception and Action
Topoi (2009)