Abstract
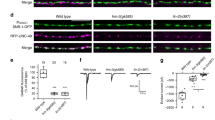
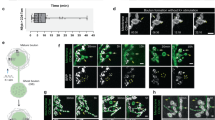
At the Drosophila melanogaster larval neuromuscular junction (NMJ), a motor neuron releases glutamate from 30–100 boutons onto the muscle it innervates. How transmission strength is distributed among the boutons of the NMJ is unknown. To address this, we created synapcam, a version of the Ca2+ reporter Cameleon. Synapcam localizes to the postsynaptic terminal and selectively reports Ca2+ influx through glutamate receptors (GluRs) with single-impulse and single-bouton resolution. GluR-based Ca2+ signals were uniform within a given connection (that is, a given bouton/postsynaptic terminal pair) but differed considerably among connections of an NMJ. A steep gradient of transmission strength was observed along axonal branches, from weak proximal connections to strong distal ones. Presynaptic imaging showed a matching axonal gradient, with higher Ca2+ influx and exocytosis at distal boutons. The results suggest that transmission strength is mainly determined presynaptically at the level of individual boutons, possibly by one or more factors existing in a gradient.
*Note: In the version of this article initially published online, the second author’s name was misspelled. The correct spelling should be Dierk F Reiff. The error has been corrected in the HTML version of the article. This correction has been appended to the PDF and print versions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
04 September 2005
Erratum PDF added and note added to XML
References
Hua, J.Y. & Smith, S.J. Neural activity and the dynamics of central nervous system development. Nat. Neurosci. 7, 327–332 (2004).
Constantine-Paton, M. & Cline, H.T. LTP and activity-dependent synaptogenesis: the more alike they are, the more different they become. Curr. Opin. Neurobiol. 8, 139–148 (1998).
Turrigiano, G.G. & Nelson, S.B. Hebb and homeostasis in neuronal plasticity. Curr. Opin. Neurobiol. 10, 358–364 (2000).
Liu, G. Presynaptic control of quantal size: kinetic mechanisms and implications for synaptic transmission and plasticity. Curr. Opin. Neurobiol. 13, 324–331 (2003).
Oertner, T.G. Functional imaging of single synapses in brain slices. Exp. Physiol. 87, 733–736 (2002).
Fetcho, J.R. & O'Malley, D.M. Imaging neuronal networks in behaving animals. Curr. Opin. Neurobiol. 7, 832–838 (1997).
Murthy, V.N. Optical detection of synaptic vesicle exocytosis and endocytosis. Curr. Opin. Neurobiol. 9, 314–320 (1999).
Koester, H.J. & Johnston, D. Target cell-dependent normalization of transmitter release at neocortical synapses. Science 308, 863–866 (2005).
Murthy, V.N., Sejnowski, T.J. & Stevens, C.F. Heterogeneous release properties of visualized individual hippocampal synapses. Neuron 18, 599–612 (1997).
Koester, H.J. & Sakmann, B. Calcium dynamics associated with action potentials in single nerve terminals of pyramidal cells in layer 2/3 of the young rat neocortex. J. Physiol. (Lond.) 529, 625–646 (2000).
Zito, K., Fetter, R.D., Goodman, C.S. & Isacoff, E.Y. Synaptic clustering of Fasciclin II and Shaker: Essential targeting sequences and role of Dlg. Neuron 19, 1007–1016 (1997).
Tejedor, F.J. et al. Essential role for dlg in synaptic clustering of shaker K+ channels in vivo. J. Neurosci. 17, 152–159 (1997).
Sorra, K.E. & Harris, K.M. Occurrence and three-dimensional structure of multiple synapses between individual radiatum axons and their target pyramidal cells in hippocampal area CA1. J. Neurosci. 13, 3736–3748 (1993).
Atwood, H.L., Govind, C.K. & Wu, C.-F. Differential ultrastructure of synaptic terminals on ventral longitudinal abdominal muscles in Drosophila larvae. J. Neurobiol. 24, 1008–1024 (1993).
Schuster, C.M., Davis, G.W., Fetter, R.D. & Goodman, C.S. Genetic dissection of structural and functional components of synaptic plasticity. I. Fasciclin II controls synaptic stabilization and growth. Neuron 17, 641–654 (1996).
Zito, K., Parnas, D., Fetter, R.D., Isacoff, E.Y. & Goodman, C.S. Watching a synapse grow: Noninvasive confocal imaging of synaptic growth in Drosophila. Neuron 22, 719–729 (1999).
McCabe, B.D. et al. The BMP homolog Gbb provides a retrograde signal that regulates synaptic growth at the Drosophila neuromuscular junction. Neuron 39, 241–254 (2003).
Aberle, H. et al. wishful thinking encodes a BMP type II receptor that regulates synaptic growth in Drosophila. Neuron 33, 545–558 (2002).
Marques, G. et al. The Drosophila BMP type II receptor Wishful Thinking regulates neuromuscular synapse morphology and function. Neuron 33, 529–543 (2002).
Haghighi, A.P. et al. Retrograde control of synaptic transmission by postsynaptic CaMKII at the Drosophila neuromuscular junction. Neuron 39, 255–267 (2003).
Chang, H., Ciani, S. & Kidokoro, Y. Ion permeation properties of the glutamate receptor channel in cultured embryonic Drosophila myotubes. J. Physiol. (Lond.) 476, 1–16 (1994).
Miyawaki, A. et al. Fluorescent indicators for Ca2+ based on green fluorescent proteins and calmodulin. Nature 388, 882–887 (1997).
Miyawaki, A., Griesbeck, O., Heim, R. & Tsien, R.Y. Dynamic and quantitative Ca2+ measurements using improved cameleons. Proc. Natl. Acad. Sci. USA 96, 2135–2140 (1999).
Chiba, A., Snow, P., Keshishian, H. & Hotta, Y. Fasciclin III as a synaptic target recognition molecule in Drosophila. Nature 374, 166–168 (1995).
Johansen, J., Halpern, M.E., Johansen, K.M. & Keshishian, H. Stereotypic morphology of glutamatergic synapses on identified muscle cells of Drosophila larvae. J. Neurosci. 9, 710–725 (1989).
Jia, X.X., Gorczyca, M. & Budnik, V. Ultrastructure of neuromuscular junctions in Drosophila: comparison of wild type and mutants with increased excitability. J. Neurobiol. 24, 1025–1044 (1993).
Treiman, M., Caspersen, C. & Christensen, S.B. A tool coming of age: thapsigargin as an inhibitor of sarcoendoplasmic reticulum Ca2+-ATPases. Trends Pharmacol. Sci. 19, 131–135 (1998).
Sullivan, K.M.C., Scott, K., Zuker, C.S. & Rubin, G.M. The ryanodine receptor is essential for larval development in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 97, 5942–5947 (2000).
Davis, G.W. & Goodman, C.S. Synapse-specific control of synaptic efficacy at the terminals of a single neuron. Nature 392, 82–86 (1998).
Petersen, S.A., Fetter, R.D., Noordemeer, J.N., Goodman, C.S. & DiAntonio, A. Genetic analysis of glutamate receptors in Drosophila reveals a retrograde signal regulating presynaptic transmitter release. Neuron 19, 1237–1248 (1997).
Marrus, S.B., Portman, S.L., Allen, M.J., Moffat, K.G. & DiAntonio, A. Differential localization of glutamate receptor subunits at the Drosophila neuromuscular junction. J. Neurosci. 24, 1406–1415 (2004).
Qin, G. et al. Four different subunits are essential for expressing the synaptic glutamate receptor at neuromuscular junctions of Drosophila. J. Neurosci. 25, 3209–3218 (2005).
DiAntonio, A., Petersen, S.A., Heckmann, M. & Goodman, C.S. Glutamate receptor expression regulates quantal size and quantal content at the Drosophila neuromuscular junction. J. Neurosci. 19, 3023–3032 (1999).
Wucherpfennig, T., Wilsch-Brauninger, M. & Gonzalez-Gaitan, M. Role of Drosophila Rab5 during endosomal trafficking at the synapse and evoked neurotransmitter release. J. Cell Biol. 161, 609–624 (2003).
Griesbeck, O., Baird, G.S., Campbell, R.E., Zacharias, D.A. & Tsien, R.Y. Reducing the environmental sensitivity of yellow fluorescent protein. Mechanism and applications. J. Biol. Chem. 276, 29188–29194 (2001).
Reiff, D.F. et al. In vivo performance of genetically encoded indicators of neural activity in flies. J. Neurosci. 25, 4766–4778 (2005).
Miesenbock, G., De Angelis, D.A. & Rothman, J.E. Visualizing secretion and synaptic transmission with pH-sensitive green fluorescent proteins. Nature 394, 192–195 (1998).
Atwood, H.L. Variation in physiological properties of crustacean motor synapses. Nature 215, 57–58 (1967).
Lavidis, N.A. & Bennett, M.R. Probabilistic secretion of quanta from visualized sympathetic nerve varicosities in mouse vas deferens. J. Physiol. (Lond.) 454, 9–26 (1992).
Bennett, M.R., Jones, P. & Lavidis, N.A. The probability of quantal secretion along visualized terminal branches at amphibian (Bufo marinus) neuromuscular synapses. J. Physiol. (Lond.) 379, 257–274 (1986).
Bittner, G.D. Differentiation of nerve terminals in the crayfish opener muscle and its functional significance. J. Gen. Physiol. 51, 731–758 (1968).
Cooper, R.L., Harrington, C.C., Marin, L. & Atwood, H.L. Quantal release at visualized terminals of a crayfish motor axon: intraterminal and regional differences. J. Comp. Neurol. 375, 583–600 (1996).
Robitaille, R. & Tremblay, J.P. Non-uniform responses to Ca2+ along the frog neuromuscular junction: effects on the probability of spontaneous and evoked transmitter release. Neuroscience 40, 571–585 (1991).
Davis, G.W., DiAntonio, A., Petersen, S.A. & Goodman, C.S. Postsynaptic PKA controls quantal size and reveals a retrograde signal that regulates presynaptic transmitter release in Drosophila. Neuron 20, 305–315 (1998).
Reiff, D.F., Thiel, P.R. & Schuster, C.M. Differential regulation of active zone density during long-term strengthening of Drosophila neuromuscular junctions. J. Neurosci. 22, 9399–9409 (2002).
Quigley, P.A., Msghina, M., Govind, C.K. & Atwood, H.L. Visible evidence for differences in synaptic effectiveness with activity-dependent vesicular uptake and release of FM1–43. J. Neurophysiol. 81, 356–370 (1999).
Roos, J., Hummel, T., Ng, N., Klambt, C. & Davis, G.W. Drosophila Futsch regulates synaptic microtubule organization and is necessary for synaptic growth. Neuron 26, 371–382 (2000).
Ruiz-Canada, C. et al. New synaptic bouton formation is disrupted by misregulation of microtubule stability in aPKC mutants. Neuron 42, 567–580 (2004).
Stewart, B.A., Atwood, H.L., Renger, J.J., Wang, J. & Wu, C.-F. Improved stability of Drosophila larval neuromuscular preparations in haemolymph-like physiological solutions. J. Comp. Physiol. A 175, 179–191 (1994).
Acknowledgements
We thank D. Raymond for developing the Bouton Project software; K. Zito for initial cloning and transfection of synapcam3.1 and M.-M. Poo, R. Zucker and M. Neff for comments on the manuscript. This work was funded by a US National Institutes of Health grant (E.Y.I. and C.S.G.), the Max-Planck-Society (D.F.R. and A.B.) and the Howard Hughes Medical Institute (G.G. and C.S.G.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
The relative increase in ΔFRET or decrease in current after two stimuli is smaller for postsynaptic terminals and NMJs with higher transmission. (PDF 267 kb)
Supplementary Fig. 2
GluR identity and distribution cannot explain the proximal-distal gradient of transmission strength. (PDF 4397 kb)
Supplementary Fig. 3
Differences between boutons in the number of active zone patches or the total quantity of an active zone marker cannot explain the proximal-distal gradient of transmission strength. (PDF 340 kb)
Supplementary Fig. 4
Possible origins for a presynaptic gradient of transmission strength. (PDF 553 kb)
Supplementary Table 1
Recording conditions and synapcam3.1 expression do not affect the physiological properties of the NMJ. (PDF 77 kb)
Rights and permissions
About this article
Cite this article
Guerrero, G., Reiff, D., Agarwal, G. et al. Heterogeneity in synaptic transmission along a Drosophila larval motor axon. Nat Neurosci 8, 1188–1196 (2005). https://doi.org/10.1038/nn1526
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1526
This article is cited by
-
Retinal bipolar cells: elementary building blocks of vision
Nature Reviews Neuroscience (2014)
-
Quantitative super-resolution imaging of Bruchpilot distinguishes active zone states
Nature Communications (2014)
-
Analysis of various physiological salines for heart rate, CNS function, and synaptic transmission at neuromuscular junctions in Drosophila melanogaster larvae
Journal of Comparative Physiology A (2014)
-
Ultrasensitive fluorescent proteins for imaging neuronal activity
Nature (2013)
-
Optical quantal analysis of synaptic transmission in wild-type and rab3-mutant Drosophila motor axons
Nature Neuroscience (2011)