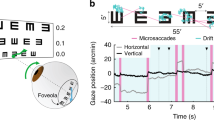
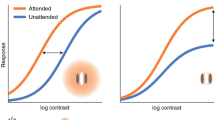
Abstract
Efficient control of attentional resources and high-acuity vision are both fundamental for survival. Shifts in visual attention are known to covertly enhance processing at locations away from the center of gaze, where visual resolution is low. It is unknown, however, whether selective spatial attention operates where the observer is already looking—that is, within the high-acuity foveola, the small yet disproportionally important rod-free region of the retina. Using new methods for precisely controlling retinal stimulation, here we show that covert attention flexibly improves and speeds up both detection and discrimination at loci only a fraction of a degree apart within the foveola. These findings reveal a surprisingly precise control of attention and its involvement in fine spatial vision. They show that the commonly studied covert shifts of attention away from the fovea are the expression of a global mechanism that exerts its action across the entire visual field.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Carrasco, M. Visual attention: the past 25 years. Vision Res. 51, 1484–1525 (2011).
Reynolds, J.H. & Chelazzi, L. Attentional modulation of visual processing. Annu. Rev. Neurosci. 27, 611–647 (2004).
Carrasco, M. Covert attention increases contrast sensitivity: Psychophysical, neurophysiological and neuroimaging studies. Prog. Brain Res. 154, 33–70 (2006).
Anton-Erxleben, K. & Carrasco, M. Attentional enhancement of spatial resolution: linking behavioural and neurophysiological evidence. Nat. Rev. Neurosci. 14, 188–200 (2013).
Kowler, E., Anderson, E., Dosher, B. & Blaser, E. The role of attention in the programming of saccades. Vision Res. 35, 1897–1916 (1995).
Schall, J.D. On the role of frontal eye field in guiding attention and saccades. Vision Res. 44, 1453–1467 (2004).
Li, H.H., Barbot, A. & Carrasco, M. Saccade preparation reshapes sensory tuning. Curr. Biol. 26, 1564–1570 (2016).
Egeth, H.E. & Yantis, S. Visual attention: control, representation, and time course. Annu. Rev. Psychol. 48, 269–297 (1997).
Krauzlis, R.J., Lovejoy, L.P. & Zénon, A. Superior colliculus and visual spatial attention. Annu. Rev. Neurosci. 36, 165–182 (2013).
Curcio, C.A., Sloan, K.R., Kalina, R.E. & Hendrickson, A.E. Human photoreceptor topography. J. Comp. Neurol. 292, 497–523 (1990).
Cavanagh, P., Hunt, A.R., Afraz, A. & Rolfs, M. Visual stability based on remapping of attention pointers. Trends Cogn. Sci. 14, 147–153 (2010).
Poletti, M. & Rucci, M. A compact field guide to the study of microsaccades: challenges and functions. Vision Res. 118, 83–97 (2016).
Kowler, E. Eye movements: the past 25 years. Vision Res. 51, 1457–1483 (2011).
Rucci, M. & Poletti, M. Control and functions of fixational eye movements. Annu. Rev. Vis. Sci. 1, 499–518 (2015).
Eriksen, C.W. & Hoffman, J.E. Temporal and spatial characteristics of selective encoding from visual displays. Percept. Psychophys. 12, 2-B, 201–204 (1972).
Eriksen, C.W. & St James, J.D. Visual attention within and around the field of focal attention: a zoom lens model. Percept. Psychophys. 40, 225–240 (1986).
Nakayama, K. & Mackeben, M. Sustained and transient components of focal visual attention. Vision Res. 29, 1631–1647 (1989).
Intriligator, J. & Cavanagh, P. The spatial resolution of visual attention. Cognit. Psychol. 43, 171–216 (2001).
Barbot, A. & Carrasco, M. Attention modifies spatial resolution according to task demands. Psychol. Sci. 28, 285–296 (2017).
Poletti, M., Listorti, C. & Rucci, M. Microscopic eye movements compensate for nonhomogeneous vision within the fovea. Curr. Biol. 23, 1691–1695 (2013).
Santini, F., Redner, G., Iovin, R. & Rucci, M. EyeRIS: a general-purpose system for eye-movement-contingent display control. Behav. Res. Methods 39, 350–364 (2007).
Holmqvist, K. et al. Eye Tracking: A Comprehensive Guide to Methods and Measures (Oxford Univ. Press, 2011).
Poletti, M., Aytekin, M. & Rucci, M. Head-eye coordination at a microscopic scale. Curr. Biol. 25, 3253–3259 (2015).
Aytekin, M., Victor, J.D. & Rucci, M. The visual input to the retina during natural head-free fixation. J. Neurosci. 34, 12701–12715 (2014).
Ko, H.-K., Poletti, M. & Rucci, M. Microsaccades precisely relocate gaze in a high visual acuity task. Nat. Neurosci. 13, 1549–1553 (2010).
Virsu, V. & Rovamo, J. Visual resolution, contrast sensitivity, and the cortical magnification factor. Exp. Brain Res. 37, 475–494 (1979).
Carrasco, M., McElree, B., Denisova, K. & Giordano, A.M. Speed of visual processing increases with eccentricity. Nat. Neurosci. 6, 699–700 (2003).
Wyman, D. & Steinman, R.M. Latency characteristics of small saccades. Vision Res. 13, 2173–2175 (1973).
Sinha, R. et al. Cellular and circuit mechanisms shaping the perceptual properties of the primate fovea. Cell 168, 413–426 (2017).
Azzopardi, P., Jones, K.E. & Cowey, A. Uneven mapping of magnocellular and parvocellular projections from the lateral geniculate nucleus to the striate cortex in the macaque monkey. Vision Res. 39, 2179–2189 (1999).
Malpeli, J.G., Lee, D. & Baker, F.H. Laminar and retinotopic organization of the macaque lateral geniculate nucleus: magnocellular and parvocellular magnification functions. J. Comp. Neurol. 375, 363–377 (1996).
Goffart, L., Hafed, Z.M. & Krauzlis, R.J. Visual fixation as equilibrium: evidence from superior colliculus inactivation. J. Neurosci. 32, 10627–10636 (2012).
He, S., Cavanagh, P. & Intriligator, J. Attentional resolution. Trends Cogn. Sci. 1, 115–121 (1997).
Yeshurun, Y. & Rashal, E. Precueing attention to the target location diminishes crowding and reduces the critical distance. J. Vis. 10, 16 (2010).
Levi, D.M., Klein, S.A. & Aitsebaomo, A.P. Vernier acuity, crowding and cortical magnification. Vision Res. 25, 963–977 (1985).
Rucci, M., Iovin, R., Poletti, M. & Santini, F. Miniature eye movements enhance fine spatial detail. Nature 447, 851–854 (2007).
Womelsdorf, T., Anton-Erxleben, K. & Treue, S. Receptive field shift and shrinkage in macaque middle temporal area through attentional gain modulation. J. Neurosci. 28, 8934–8944 (2008).
Fries, P., Reynolds, J.H., Rorie, A.E. & Desimone, R. Modulation of oscillatory neuronal synchronization by selective visual attention. Science 291, 1560–1563 (2001).
Cohen, M.R. & Maunsell, J.H.R. Attention improves performance primarily by reducing interneuronal correlations. Nat. Neurosci. 12, 1594–1600 (2009).
Mitchell, J.F., Sundberg, K.A. & Reynolds, J.H. Spatial attention decorrelates intrinsic activity fluctuations in macaque area V4. Neuron 63, 879–888 (2009).
Azzopardi, P. & Cowey, A. Preferential representation of the fovea in the primary visual cortex. Nature 361, 719–721 (1993).
Cherici, C., Kuang, X., Poletti, M. & Rucci, M. Precision of sustained fixation in trained and untrained observers. J. Vis. 12, 1–16 (2012).
Lisi, M., Cavanagh, P. & Zorzi, M. Spatial constancy of attention across eye movements is mediated by the presence of visual objects. Atten. Percept. Psychophys. 77, 1159–1169 (2015).
Yuval-Greenberg, S., Merriam, E.P. & Heeger, D.J. Spontaneous microsaccades reflect shifts in covert attention. J. Neurosci. 34, 13693–13700 (2014).
Hafed, Z.M. Alteration of visual perception prior to microsaccades. Neuron 77, 775–786 (2013).
Crane, H.D. & Steele, C.M. Generation-V dual-Purkinje-image eyetracker. Appl. Opt. 24, 527–537 (1985).
Putnam, N.M. et al. The locus of fixation and the foveal cone mosaic. J. Vis. 5, 632–639 (2005).
Li, K.Y., Tiruveedhula, P. & Roorda, A. Intersubject variability of foveal cone photoreceptor density in relation to eye length. Invest. Ophthalmol. Vis. Sci. 51, 6858–6867 (2010).
Anderson, A.J. & Vingrys, A.J. Small samples: does size matter? Invest. Ophthalmol. Vis. Sci. 42, 1411–1413 (2001).
Acknowledgements
This work was supported by National Science Foundation grants BCS-1534932 (M.P.) and 1420212 (M.R.), and National Institutes of Health grants R01-EY18363 (M.R.), R01-EY019693 (M.C) and R01-EY016200 (M.C). We thank M. Landy, S. Ling, E. Niebur, M. Spering, J. Victor, A. White and Y. Yeshurun for comments and R. Ezzo for helping with data collection.
Author information
Authors and Affiliations
Contributions
M.P. conceived the study and collected and analyzed the data. The three authors contributed to the design of the experiments, the interpretation of experimental data, and the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Velocity of eye movements.
Data represent averages across observers (n=5) for the cue-target intervals of Experiment 1 (parafovea; Fig. 1) and 2 (foveola; Fig. 2). In Experiment 2, stimuli were stabilized on the retina. (A) Average polar histograms of ocular drift velocity. (B) Mean instantaneous speed of ocular drift. Similar speed values were measured in the two experiments (p=0.63; two-tailed paired t-test). Error bars represent s.e.m. Time t=0 marks cue onset.
Supplementary Figure 2 Comparison between manual and oculomotor reaction times with stimuli in the parafovea and in the foveola.
The two sets of data on the left (Manual) refer to the neutral trials of the detection experiments of Fig. 1 (parafovea; red) and Fig. 2 (foveola; blue). In these experiments, the observer reported the appearance of the target by pressing a button. For comparison, data points on the right (Saccade) show oculomotor reaction times in the neutral trials of similar experiments, but in which the target was reported by performing a saccade toward its location rather than by pressing a button. Reaction times are measured relative to target’s offset. Error bars are s.e.m. (n=4).
Supplementary Figure 3 Fine attentional control during normal retinal image motion (drift-only trials).
Means ± s.e.m. for two individual subjects who were run extensively in Experiment 4 to collect sufficient numbers of trials without microsaccades (percentages of trials with microsaccades: 34%). Both subjects continued to exhibit significant differences between valid and invalid trials in both sensitivity (p<0.05, two-tailed z-test) and reaction times (p<0.05, ranksum test).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 (PDF 686 kb)
Rights and permissions
About this article
Cite this article
Poletti, M., Rucci, M. & Carrasco, M. Selective attention within the foveola. Nat Neurosci 20, 1413–1417 (2017). https://doi.org/10.1038/nn.4622
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4622
This article is cited by
-
Visual perceptual learning modulates microsaccade rate and directionality
Scientific Reports (2023)
-
Binocular summation is affected by crowding and tagging
Scientific Reports (2021)
-
Different computations underlie overt presaccadic and covert spatial attention
Nature Human Behaviour (2021)
-
Oculomotor freezing reflects tactile temporal expectation and aids tactile perception
Nature Communications (2020)
-
Accuracy and precision of small saccades
Scientific Reports (2020)