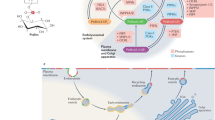
Abstract
Membrane rafts are thought to be sphingolipid- and cholesterol-dependent lateral assemblies involved in diverse cellular functions. Their biological roles and even their existence, however, remain controversial. Using an original fluorescence correlation spectroscopy strategy that recently enabled us to identify nanoscale membrane organizations in live cells, we report here that highly dynamic nanodomains exist in both the outer and inner leaflets of the plasma membrane. Through specific inhibition of biosynthesis, we show that sphingolipids and cholesterol are essential and act in concert for formation of nanodomains, thus corroborating their raft nature. Moreover, we find that nanodomains play a crucial role in triggering the phosphatidylinositol-3 kinase/Akt signaling pathway, by facilitating Akt recruitment and activation upon phosphatidylinositol-3,4,5-triphosphate accumulation in the plasma membrane. Thus, through direct monitoring and controlled alterations of rafts in living cells, we demonstrate that rafts are critically involved in the activation of a signaling axis that is essential for cell physiology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Simons, K. & Ikonen, E. Functional rafts in cell membranes. Nature 387, 569–572 (1997).
Brown, D.A. & London, E. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 14, 111–136 (1998).
Edidin, M. The state of lipid rafts: from model membranes to cells. Annu. Rev. Biophys. Biomol. Struct. 32, 257–283 (2003).
Hancock, J.F. Lipid rafts: contentious only from simplistic standpoints. Nat. Rev. Mol. Cell Biol. 7, 456–462 (2006).
Marguet, D., Lenne, P.F., Rigneault, H. & He, H.T. Dynamics in the plasma membrane: how to combine fluidity and order. EMBO J. 25, 3446–3457 (2006).
Jacobson, K., Mouritsen, O.G. & Anderson, R.G. Lipid rafts: at a crossroad between cell biology and physics. Nat. Cell Biol. 9, 7–14 (2007).
Simons, K. & Toomre, D. Lipid rafts and signal transduction. Nat. Rev. Mol. Cell Biol. 1, 31–39 (2000).
Munro, S. Lipid rafts: elusive or illusive? Cell 115, 377–388 (2003).
Pralle, A., Keller, P., Florin, E.L., Simons, K. & Horber, J.K. Sphingolipid-cholesterol rafts diffuse as small entities in the plasma membrane of mammalian cells. J. Cell Biol. 148, 997–1008 (2000).
Niv, H., Gutman, O., Kloog, Y. & Henis, Y.I. Activated K-Ras and H-Ras display different interactions with saturable nonraft sites at the surface of live cells. J. Cell Biol. 157, 865–872 (2002).
Zacharias, D.A., Violin, J.D., Newton, A.C. & Tsien, R.Y. Partitioning of lipid-modified monomeric GFPs into membrane microdomains of live cells. Science 296, 913–916 (2002).
Prior, I.A., Muncke, C., Parton, R.G. & Hancock, J.F. Direct visualization of Ras proteins in spatially distinct cell surface microdomains. J. Cell Biol. 160, 165–170 (2003).
Gaus, K. et al. Visualizing lipid structure and raft domains in living cells with two-photon microscopy. Proc. Natl. Acad. Sci. USA 100, 15554–15559 (2003).
Sharma, P. et al. Nanoscale organization of multiple GPI-anchored proteins in living cell membranes. Cell 116, 577–589 (2004).
Ianoul, A. et al. Imaging nanometer domains of beta-adrenergic receptor complexes on the surface of cardiac myocytes. Nat. Chem. Biol. 1, 196–202 (2005).
Wawrezinieck, L., Rigneault, H., Marguet, D. & Lenne, P.F. Fluorescence correlation spectroscopy diffusion laws to probe the submicron cell membrane organization. Biophys. J. 89, 4029–4042 (2005).
Lenne, P.F. et al. Dynamic molecular confinement in the plasma membrane by microdomains and the cytoskeleton meshwork. EMBO J. 25, 3245–3256 (2006).
Hanada, M., Feng, J. & Hemmings, B.A. Structure, regulation and function of PKB/AKT-a major therapeutic target. Biochim. Biophys. Acta 1697, 3–16 (2004).
Scheid, M.P. & Woodgett, J.R. Unravelling the activation mechanisms of protein kinase B/Akt. FEBS Lett. 546, 108–112 (2003).
Sarbassov, D.D., Guertin, D.A., Ali, S.M. & Sabatini, D.M. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307, 1098–1101 (2005).
Zhuang, L., Lin, J., Lu, M.L., Solomon, K.R. & Freeman, M.R. Cholesterol-rich lipid rafts mediate akt-regulated survival in prostate cancer cells. Cancer Res. 62, 2227–2231 (2002).
Adam, R.M. et al. Cholesterol sensitivity of endogenous and myristoylated Akt. Cancer Res. 67, 6238–6246 (2007).
Lee, K.Y., D'Acquisto, F., Hayden, M.S., Shim, J.H. & Ghosh, S. PDK1 nucleates T cell receptor-induced signaling complex for NF-kappaB activation. Science 308, 114–118 (2005).
Hill, M.M., Feng, J. & Hemmings, B.A. Identification of a plasma membrane Raft-associated PKB Ser473 kinase activity that is distinct from ILK and PDK1. Curr. Biol. 12, 1251–1255 (2002).
Lucero, H., Gae, D. & Taccioli, G.E. Novel localization of the DNA-PK complex in lipid rafts: a putative role in the signal transduction pathway of the ionizing radiation response. J. Biol. Chem. 278, 22136–22143 (2003).
Pages, F. et al. Binding of phosphatidylinositol-3-OH kinase to CD28 is required for T-cell signalling. Nature 369, 327–329 (1994).
Wenger, J. et al. Diffusion analysis within single nanometric apertures reveals the ultrafine cell membrane organization. Biophys. J. 92, 913–919 (2007).
Miyake, Y., Kozutsumi, Y., Nakamura, S., Fujita, T. & Kawasaki, T. Serine palmitoyltransferase is the primary target of a sphingosine-like immunosuppressant, ISP-1/myriocin. Biochem. Biophys. Res. Commun. 211, 396–403 (1995).
Bergstrom, J.D. et al. Zaragozic acids: a family of fungal metabolites that are picomolar competitive inhibitors of squalene synthase. Proc. Natl. Acad. Sci. USA 90, 80–84 (1993).
Freeburn, R.W. et al. Evidence that SHIP-1 contributes to phosphatidylinositol 3,4,5-trisphosphate metabolism in T lymphocytes and can regulate novel phosphoinositide 3-kinase effectors. J. Immunol. 169, 5441–5450 (2002).
Berg, L.J., Finkelstein, L.D., Lucas, J.A. & Schwartzberg, P.L. Tec family kinases in T lymphocyte development and function. Annu. Rev. Immunol. 23, 549–600 (2005).
Lippincott-Schwartz, J. & Patterson, G.H. Development and use of fluorescent protein markers in living cells. Science 300, 87–91 (2003).
Oancea, E., Teruel, M.N., Quest, A.F. & Meyer, T. Green fluorescent protein (GFP)-tagged cysteine-rich domains from protein kinase C as fluorescent indicators for diacylglycerol signaling in living cells. J. Cell Biol. 140, 485–498 (1998).
Varnai, P. et al. Selective cellular effects of overexpressed pleckstrin-homology domains that recognize PtdIns(3,4,5)P3 suggest their interaction with protein binding partners. J. Cell Sci. 118, 4879–4888 (2005).
Kiessling, V., Crane, J.M. & Tamm, L.K. Transbilayer effects of raft-like lipid domains in asymmetric planar bilayers measured by single molecule tracking. Biophys. J. 91, 3313–3326 (2006).
Kwik, J. et al. Membrane cholesterol, lateral mobility, and the phosphatidylinositol 4,5-bisphosphate-dependent organization of cell actin. Proc. Natl. Acad. Sci. USA 100, 13964–13969 (2003).
Kabouridis, P.S., Janzen, J., Magee, A.L. & Ley, S.C. Cholesterol depletion disrupts lipid rafts and modulates the activity of multiple signaling pathways in T lymphocytes. Eur. J. Immunol. 30, 954–963 (2000).
Magee, A.I., Adler, J. & Parmryd, I. Cold-induced coalescence of T-cell plasma membrane microdomains activates signalling pathways. J. Cell Sci. 118, 3141–3151 (2005).
Tong, J. et al. Role of GAP-43 in sequestering phosphatidylinositol 4,5-bisphosphate to Raft bilayers. Biophys. J. 94, 125–133 (2008).
Gokhale, N.A., Abraham, A., Digman, M.A., Gratton, E. & Cho, W. Phosphoinositide specificity of and mechanism of lipid domain formation by annexin A2-p11 heterotetramer. J. Biol. Chem. 280, 42831–42840 (2005).
Epand, R.M., Vuong, P., Yip, C.M., Maekawa, S. & Epand, R.F. Cholesterol-dependent partitioning of PtdIns(4,5)P2 into membrane domains by the N-terminal fragment of NAP-22 (neuronal axonal myristoylated membrane protein of 22 kDa). Biochem. J. 379, 527–532 (2004).
Golebiewska, U. et al. Membrane-bound basic peptides sequester multivalent (PIP2), but not monovalent (PS), acidic lipids. Biophys. J. 91, 588–599 (2006).
Datta, K. et al. AH/PH domain-mediated interaction between Akt molecules and its potential role in Akt regulation. Mol. Cell. Biol. 15, 2304–2310 (1995).
Calleja, V. et al. Intramolecular and intermolecular interactions of protein kinase B define its activation in vivo. PLoS Biol. 5, e95 (2007).
Feng, J., Park, J., Cron, P., Hess, D. & Hemmings, B.A. Identification of a PKB/Akt hydrophobic motif Ser-473 kinase as DNA-dependent protein kinase. J. Biol. Chem. 279, 41189–41196 (2004).
Jones, K.A., Jiang, X., Yamamoto, Y. & Yeung, R.S. Tuberin is a component of lipid rafts and mediates caveolin-1 localization: role of TSC2 in post-Golgi transport. Exp. Cell Res. 295, 512–524 (2004).
Gaus, K. et al. Condensation of the plasma membrane at the site of T lymphocyte activation. J. Cell Biol. 171, 121–131 (2005).
Costello, P.S., Gallagher, M. & Cantrell, D.A. Sustained and dynamic inositol lipid metabolism inside and outside the immunological synapse. Nat. Immunol. 3, 1082–1089 (2002).
Harriague, J. & Bismuth, G. Imaging antigen-induced PI3K activation in T cells. Nat. Immunol. 3, 1090–1096 (2002).
Axelrod, D., Koppel, D.E., Schlessinger, J., Elson, E. & Webb, W.W. Mobility measurement by analysis of fluorescence photobleaching recovery kinetics. Biophys. J. 16, 1055–1069 (1976).
Acknowledgements
This research project was supported by institutional grants from Institut National de la Santé et de la Recherche Médicale and Centre National de la Recherche Scientifique, and by specific grants from Agence Nationale de la Recherche, Association pour la Recherche sur le Cancer, European regional development fund, Fondation pour la Recherche Médicale, Institut National du Cancer (PL06-026, PL96-009 and PF108-05), Ligue Nationale Française contre le Cancer, Ministère de l'Éducation Nationale, de la Recherche et de la Technologie and Centre National de la Recherche Scientifique. Part of the work was supported by IFR30 Lipidomics of the Functional Exploration Platform of Toulouse Genopole. R.L. and F.C. were awarded fellowships from the Ministère de l'Éducation Nationale, de la Recherche et de la Technologie. We thank M. Fallet (PICsL imaging core facility) and F. Garçon for technical assistance, P. Chavrier (Institut Curie), J. Downward (Cancer Research UK London Research Institute), H. Lelouard (Centre d'Immunologie de Marseille-Luminy), S. Méresse (Centre d'Immunologie de Marseille-Luminy) and T. Meyer (Stanford University Medical School) for reagents, Y. Xia for chemical structure drawing, K. Simons, G. van Meer and C. Zhu for discussion, P. Golstein, J.P. Gorvel, A.-O. Hueber, L. Leserman, A. Pamidi and T. Reisine for critical reading of the manuscript and E. Witty (AngloScribe) for editing the English.
Author information
Authors and Affiliations
Contributions
R.L., X.-J.G., F.C., Y.H., B.P., D.M. and H.-T.H. conceived and designed the experiments. R.L., X.-J.G., F.C., Y.H., O.H., A.-M.B. and S.M.S. performed the experiments. R.L., X.-J.G., F.C., Y.H., B.P., D.M. and H.-T.H. analyzed the data. P.-F.L., H.R., D.O., G.B., J.A.N. and B.P. contributed reagents, materials and analysis tools. D.M. and H.-T.H. wrote the paper with contributions from R.L., X.-J.G., F.C., Y.H. and B.P.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5, Supplementary Tables 1 and 2, Supplementary Data, Supplementary Discussion and Supplementary Methods (PDF 2140 kb)
Rights and permissions
About this article
Cite this article
Lasserre, R., Guo, XJ., Conchonaud, F. et al. Raft nanodomains contribute to Akt/PKB plasma membrane recruitment and activation. Nat Chem Biol 4, 538–547 (2008). https://doi.org/10.1038/nchembio.103
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.103
This article is cited by
-
Cholesterol modulates type I/II TGF-β receptor complexes and alters the balance between Smad and Akt signaling in hepatocytes
Communications Biology (2024)
-
PI3K signalling at the intersection of cardio-oncology networks: cardiac safety in the era of AI
Cellular and Molecular Life Sciences (2022)
-
Do ABC transporters regulate plasma membrane organization?
Cellular & Molecular Biology Letters (2020)
-
CD90 serves as differential modulator of subcutaneous and visceral adipose-derived stem cells by regulating AKT activation that influences adipose tissue and metabolic homeostasis
Stem Cell Research & Therapy (2019)
-
A Complex Interplay of Anionic Phospholipid Binding Regulates 3′-Phosphoinositide-Dependent-Kinase-1 Homodimer Activation
Scientific Reports (2019)