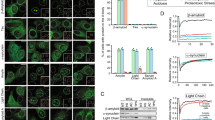
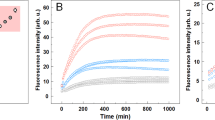
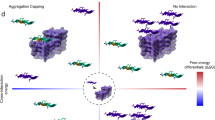
Abstract
Many amyloid inhibitors resemble molecules that form chemical aggregates, which are known to inhibit many proteins. Eight known chemical aggregators inhibited amyloid formation of the yeast and mouse prion proteins Sup35 and recMoPrP in a manner characteristic of colloidal inhibition. Similarly, three known anti-amyloid molecules inhibited β-lactamase in a detergent-dependent manner, which suggests that they too form colloidal aggregates. The colloids localized to preformed fibers and prevented new fiber formation in electron micrographs. They also blocked infection of yeast cells with Sup35 prions, which suggests that colloidal inhibition may be relevant in more biological milieus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Nelson, R. et al. Nature 435, 773–778 (2005).
Blanchard, B.J. et al. Proc. Natl. Acad. Sci. USA 101, 14326–14332 (2004).
Lorenzo, A. & Yankner, B.A. Proc. Natl. Acad. Sci. USA 91, 12243–12247 (1994).
Ritchie, C.W. et al. Arch. Neurol. 60, 1685–1691 (2003).
Yang, F. et al. J. Biol. Chem. 280, 5892–5901 (2005).
Zhu, M. et al. J. Biol. Chem. 279, 26846–26857 (2004).
Heiser, V. et al. Proc. Natl. Acad. Sci. USA 97, 6739–6744 (2000).
McGovern, S.L., Caselli, E., Grigorieff, N. & Shoichet, B.K. J. Med. Chem. 45, 1712–1722 (2002).
McGovern, S.L., Helfand, B.T., Feng, B. & Shoichet, B.K. J. Med. Chem. 46, 4265–4272 (2003).
True, H.L. & Lindquist, S.L. Nature 407, 477–483 (2000).
Abid, K. & Soto, C. Cell. Mol. Life Sci. 63, 2342–2351 (2006).
Coan, K.E.D. & Shoichet, B.K. Mol. Biosyst. 3, 208–213 (2007).
Feng, B.Y., Shelat, A., Doman, T.N., Guy, R.K. & Shoichet, B.K. Nat. Chem. Biol. 1, 146–148 (2005).
Tanaka, M. & Weissman, J.S. Methods Enzymol. 412, 185–200 (2006).
Acknowledgements
This work was supported by grants from the National Institutes of Health (GM71630 to B.K.S. and AG02132, AG10770 and AG021601 to S.B.P.) and by a gift from the G. Harold and Leila Y. Mathers Charitable Foundation (to S.B.P.). B.Y.F. is supported by a Kozloff research fellowship and by a University of California San Francisco School of Pharmacy fellowship. B.H.T. is supported by an Achievement Rewards for College Scientists foundation fellowship and by the Howard Hughes Medical Institute. D.W.C. is supported by a fellowship from the Jane Coffin Childs Memorial Fund for Medical Research. S.R.C. is supported by a predoctoral fellowship from the Burroughs Wellcome Fund and by the Howard Hughes Medical Institute. We thank V. Thomas, M. Diamond and B. Frost for thoughtful discussion and K. Coan for reading this manuscript.
Author information
Authors and Affiliations
Contributions
B.Y.F. performed the experiments unless otherwise noted, and wrote the manuscript with B.K.S. B.H.T. helped perform the Sup35 infectivity assay. H.W. performed the electron microscopy studies. D.W.C. performed the recMoPrP polymerization experiments. S.R.C. helped perform the Sup35 polymerization experiments. B.C.H.M., S.B.P. and J.W. provided guidance and helped edit the manuscript.
Corresponding author
Ethics declarations
Competing interests
B.C.H.M. and S.B.P. declare a financial interest in Inpro Biotechnology, Inc. All other authors declare no competing interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4, Supplementary Tables 1–3 and Supplementary Methods (PDF 2658 kb)
Rights and permissions
About this article
Cite this article
Feng, B., Toyama, B., Wille, H. et al. Small-molecule aggregates inhibit amyloid polymerization. Nat Chem Biol 4, 197–199 (2008). https://doi.org/10.1038/nchembio.65
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.65
This article is cited by
-
Tuning the rate of aggregation of hIAPP into amyloid using small-molecule modulators of assembly
Nature Communications (2022)
-
Ion mobility spectrometry combined with multivariate statistical analysis: revealing the effects of a drug candidate for Alzheimer’s disease on Aβ1-40 peptide early assembly
Analytical and Bioanalytical Chemistry (2019)
-
Novel Small Molecules Targeting the Intrinsically Disordered Structural Ensemble of α-Synuclein Protect Against Diverse α-Synuclein Mediated Dysfunctions
Scientific Reports (2019)
-
Differential inhibition of metabolite amyloid formation by generic fibrillation-modifying polyphenols
Communications Chemistry (2018)
-
Stabilizing Off-pathway Oligomers by Polyphenol Nanoassemblies for IAPP Aggregation Inhibition
Scientific Reports (2016)