Abstract
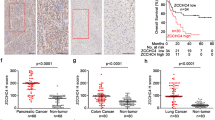
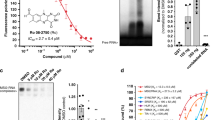
The identification of the cellular targets of small molecules with anticancer activity is crucial to their further development as drug candidates. Here, we present the application of a large-scale RNA interference–based short hairpin RNA (shRNA) barcode screen to gain insight in the mechanism of action of nutlin-3 (1). Nutlin-3 is a small-molecule inhibitor of MDM2, which can activate the p53 pathway. Nutlin-3 shows strong antitumor effects in mice, with surprisingly few side effects on normal tissues1. Aside from p53, we here identify 53BP1 as a critical mediator of nutlin-3–induced cytotoxicity. 53BP1 is part of a signaling network induced by DNA damage that is frequently activated in cancer but not in healthy tissues2. Our results suggest that nutlin-3's tumor specificity may result from its ability to turn a cancer cell–specific property (activated DNA damage signaling3) into a weakness that can be exploited therapeutically.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Vassilev, L.T. et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 303, 844–848 (2004).
DiTullio, R.A., Jr. et al. 53BP1 functions in an ATM-dependent checkpoint pathway that is constitutively activated in human cancer. Nat. Cell Biol. 4, 998–1002 (2002).
Gorgoulis, V.G. et al. Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 434, 907–913 (2005).
Lane, D.P. & Fischer, P.M. Turning the key on p53. Nature 427, 789–790 (2004).
Vogelstein, B., Lane, D. & Levine, A.J. Surfing the p53 network. Nature 408, 307–310 (2000).
Kubbutat, M.H., Jones, S.N. & Vousden, K.H. Regulation of p53 stability by Mdm2. Nature 387, 299–303 (1997).
Kussie, P.H. et al. Structure of the MDM2 oncoprotein bound to the p53 tumor suppressor transactivation domain. Science 274, 948–953 (1996).
Zheng, X.S., Chan, T.F. & Zhou, H.H. Genetic and genomic approaches to identify and study the targets of bioactive small molecules. Chem. Biol. 11, 609–618 (2004).
Harris, M.P. et al. Adenovirus-mediated p53 gene transfer inhibits growth of human tumor cells expressing mutant p53 protein. Cancer Gene Ther. 3, 121–130 (1996).
Berns, K. et al. A large-scale RNAi screen in human cells identifies new components of the p53 pathway. Nature 428, 431–437 (2004).
Brummelkamp, T.R. & Bernards, R. New tools for functional mammalian cancer genetics. Nat. Rev. Cancer 3, 781–789 (2003).
Paddison, P.J. et al. A resource for large-scale RNA-interference-based screens in mammals. Nature 428, 427–431 (2004).
Westbrook, T.F. et al. A genetic screen for candidate tumor suppressors identifies REST. Cell 121, 837–848 (2005).
Janicke, R.U., Sprengart, M.L., Wati, M.R. & Porter, A.G. Caspase-3 is required for DNA fragmentation and morphological changes associated with apoptosis. J. Biol. Chem. 273, 9357–9360 (1998).
Brummelkamp, T.R. et al. Functional identification of cancer-relevant genes through large-scale RNA interference screens in mammalian cells. Cold Spring Harb. Symp. Quant. Biol. 69, 439–445 (2004).
Moumen, A., Masterson, P., O'Connor, M.J. & Jackson, S.P. hnRNP K: an HDM2 target and transcriptional coactivator of p53 in response to DNA damage. Cell 123, 1065–1078 (2005).
Wu, M.X. Roles of the stress-induced gene IEX-1 in regulation of cell death and oncogenesis. Apoptosis 8, 11–18 (2003).
Wong, J., Li, P.X. & Klamut, H.J. A novel p53 transcriptional repressor element (p53TRE) and the asymmetrical contribution of two p53 binding sites modulate the response of the placental transforming growth factor-beta promoter to p53. J. Biol. Chem. 277, 26699–26707 (2002).
Reinholz, M.M. et al. Differential gene expression of TGF beta inducible early gene (TIEG), Smad7, Smad2 and Bard1 in normal and malignant breast tissue. Breast Cancer Res. Treat. 86, 75–88 (2004).
Wang, B., Matsuoka, S., Carpenter, P.B. & Elledge, S.J. 53BP1, a mediator of the DNA damage checkpoint. Science 298, 1435–1438 (2002).
Bartkova, J. et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434, 864–870 (2005).
Rappold, I., Iwabuchi, K., Date, T. & Chen, J. Tumor suppressor p53 binding protein 1 (53BP1) is involved in DNA damage-signaling pathways. J. Cell Biol. 153, 613–620 (2001).
Sedelnikova, O.A. et al. Senescing human cells and ageing mice accumulate DNA lesions with unrepairable double-strand breaks. Nat. Cell Biol. 6, 168–170 (2004).
Blasina, A., Price, B.D., Turenne, G.A. & McGowan, C.H. Caffeine inhibits the checkpoint kinase ATM. Curr. Biol. 9, 1135–1138 (1999).
Zhou, B.B. et al. Caffeine abolishes the mammalian G(2)/M DNA damage checkpoint by inhibiting ataxia-telangiectasia-mutated kinase activity. J. Biol. Chem. 275, 10342–10348 (2000).
Sarkaria, J.N. et al. Inhibition of ATM and ATR kinase activities by the radiosensitizing agent, caffeine. Cancer Res. 59, 4375–4382 (1999).
Lengauer, C., Kinzler, K.W. & Vogelstein, B. Genetic instabilities in human cancers. Nature 396, 643–649 (1998).
Mendrysa, S.M. et al. mdm2 is critical for inhibition of p53 during lymphopoiesis and the response to ionizing irradiation. Mol. Cell. Biol. 23, 462–472 (2003).
Heetebrij, R.J. et al. Platinum(II)-based coordination compounds as nucleic acid labeling reagents: synthesis, reactivity, and applications in hybridization assays. ChemBioChem 4, 573–583 (2003).
Quackenbush, J. Microarray data normalization and transformation. Nat. Genet. 32 (Suppl.), 496–501 (2002).
Acknowledgements
We thank the Netherlands Cancer Institute microarray facility group for assistance; K. Berns, S. Nijman, H. Ovaa and members of the Bernards and Beijersbergen laboratories for help and discussions; L. Oomen for help with confocal microscopy; and R. Kortlever for critically reading the manuscript. This work was supported by grants from the Dutch Cancer Society (KWF) and the European Union integrated project INTACT.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Brummelkamp, T., Fabius, A., Mullenders, J. et al. An shRNA barcode screen provides insight into cancer cell vulnerability to MDM2 inhibitors. Nat Chem Biol 2, 202–206 (2006). https://doi.org/10.1038/nchembio774
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio774
This article is cited by
-
High-throughput synthetic rescue for exhaustive characterization of suppressor mutations in human genes
Cellular and Molecular Life Sciences (2020)
-
A quantitative shRNA screen identifies ATP1A1 as a gene that regulates cytotoxicity by aurilide B
Scientific Reports (2017)
-
An unbiased in vivo functional genomics screening approach in mice identifies novel tumor cell-based regulators of immune rejection
Cancer Immunology, Immunotherapy (2017)
-
Co-targeting of FAK and MDM2 triggers additive anti-proliferative effects in mesothelioma via a coordinated reactivation of p53
British Journal of Cancer (2016)
-
The lethal response to Cdk1 inhibition depends on sister chromatid alignment errors generated by KIF4 and isoform 1 of PRC1
Scientific Reports (2015)