Abstract
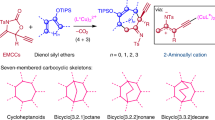
With the intention of improving synthetic efficiency, organic chemists have turned to bioinspired organocascade or domino processes that generate multiple bonds and stereocentres in a single operation. However, despite the great importance of substituted cyclopentanes, given their prevalence in complex natural products and pharmaceutical agents, the rapid, enantioselective assembly of these carbocycles lags behind cyclohexanes. Here, we describe a Michael–aldol-β-lactonization organocascade process for the synthesis of complex cyclopentanes utilizing chiral α,β-unsaturated acylammonium intermediates, readily generated by activation of commodity unsaturated acid chlorides with chiral isothiourea catalysts. This efficient methodology enables the construction of two C–C bonds, one C–O bond, two rings and up to three contiguous stereogenic centres delivering complex cyclopentanes with high levels of relative and absolute stereocontrol. Our results suggest that α,β-unsaturated acylammonium intermediates have broad utility for the design of organocascade and multicomponent processes, with the latter demonstrated by a Michael–Michael–aldol-β-lactonization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Pellissier, H. Stereocontrolled domino reactions. Chem. Rev. 113, 442–524 (2013).
Grondal, C., Jeanty, M. & Enders, D. Organocatalytic cascade reactions as a new tool in total synthesis. Nature Chem. 2, 167–178 (2010).
Jones, S. B., Simmons, B., Mastracchio, A. & MacMillan, D. W. C. Collective synthesis of natural products by means of organocascade catalysis. Nature 475, 183–188 (2011).
Enders, D., Huttl, M. R., Grondal, C. & Raabe, G. Control of four stereocentres in a triple cascade organocatalytic reaction. Nature 441, 861–863 (2006).
Gawley, R. E. The Robinson annelation and related reactions. Synthesis 777–794 (1976).
Bui, T. & Barbas, C. F. III A proline-catalyzed asymmetric Robinson annulation reaction. Tetrahedron Lett. 41, 6951–6954 (2000).
Sakakura, A., Ukai, A. & Ishihara, K. Enantioselective halocyclization of polyprenoids induced by nucleophilic phosphoramidites. Nature 445, 900–903 (2007).
Nicolaou, K. C., Synder, S. A., Montagnon, T. & Vassilikogiannakis, G. The Diels–Alder reaction in total synthesis. Angew. Chem. Int. Ed. 41, 1668–1698 (2002).
Jorg, H. The Pauson–Khand reaction in the synthesis of pharmacologically active compounds. Curr. Org. Chem. 14, 1139–1152 (2010).
Trost, B. M. & Chan, D. M. T. New conjunctive reagents. 2-Acetoxymethyl-3-allyltrimethylsilane for methylenecyclopentane annulations catalyzed by palladium(0). J. Am. Chem. Soc. 101, 6429–6432 (1979).
Streit, U. & Bochet, C. G. The arene–alkene photocycloaddition. Beilstein J. Org. Chem. 7, 525–542 (2011).
Vaidya, T., Eisenberg, R. & Frontier, A. J. Catalytic Nazarov cyclization: the state of the art. Chem. Catal. Chem. 3, 1531–1548 (2011).
Heasley, B. Stereocontrolled preparation of fully substituted cyclopentanes: relevance to total synthesis. Eur. J. Org. Chem. 1447–1489 (2009).
Moyano, A. & Ramon Rios, R. Asymmetric organocatalytic cyclization and cycloaddition reactions. Chem. Rev. 111, 4703–4832 (2011).
Tan, B., Candeias, N. R. & Barbas, C. F. III Construction of bispirooxindoles containing three quaternary stereocentres in a cascade using a single multifunctional organocatalyst. Nature Chem. 3, 473–477 (2011).
Remes, M. & Vesely, J. Highly enantioselective organocatalytic formation of functionalized cyclopentane derivatives via tandem conjugate addition/α-alkylation of enals. Eur. J. Org. Chem. 3747–3752 (2012).
Albertshofer, K., Tan, B. & Barbas, C. F. III Assembly of spirooxindole derivatives containing four consecutive stereocenters via organocatalytic Michael–Henry cascade reactions. Org. Lett. 14, 1834–1837 (2012).
Barrero, A. F., Quilez del Moral, J. F., Herrador, M. M., Rodriguez, H. & Morales, M. C. P. Cyclopentane sesquiterpenes from fungi: occurrence–bioactivity, biosynthesis and chemical synthesis. Curr. Org. Chem. 18, 1164–1181 (2009).
Cortez, G. S., Tennyson, R. & Romo, D. Intramolecular nucleophile catalyzed aldol-lactonization (NCAL) reactions: catalytic, asymmetric synthesis of bicyclic β-lactones. J. Am. Chem. Soc. 123, 7945–7946 (2001).
Leverett, C. A., Purohit, V. C. & Romo, D. Enantioselective, organocatalyzed intramolecular aldol-lactonizations with ketoacids leading to bicyclic and tricyclic β-lactones and topology morphing transformations. Angew. Chem. Int. Ed. 122, 9669–9673 (2010).
Liu, G., Shirley, M. E. & Romo, D. A diastereoselective, nucleophile-promoted aldol-lactonization of ketoacids leading to bicyclic-β-lactones. J. Org. Chem. 77, 2496–2500 (2012).
France, S., Guerin, D. J., Miller, S. J. & Lectka, T. Nucleophile chiral amines as catalysts in asymmetric synthesis. Chem. Rev. 103, 2985–3012 (2003).
Fu, G. C. Asymmetric catalysis with ‘planar-chiral’ derivatives of 4-(dimethylamino)pyridine. Acc. Chem. Res. 37, 542–547 (2004).
Birman, V. B., Uffman, E. W., Jiang, H., Li, X. & Kilbane, C. J. 2,3-Dihydroimidazo[1,2-a]pyridines: a new class of enantioselective acyl transfer catalysts and their use in kinetic resolution of alcohols. J. Am. Chem. Soc. 126, 12226–12227 (2004).
Kobayashi, M. & Okamoto, S. Unexpected reactivity of annulated 3H-benzothiazol-2-ylideneamines as acyl transfer catalyst. Tetrahedron Lett. 47, 4347–4350 (2006).
Gaunt, M. J. & Johansson, C. C. C. Recent developments in the use of catalytic asymmetric ammonium enolates in chemical synthesis. Chem. Rev. 107, 5596–5605 (2007).
Wynberg, H. & Staring, E. G. Asymmetric synthesis of (S)- and (R)-malic acid from ketene chloral. J. Am. Chem. Soc. 104, 166–168 (1982).
Nelson, S. C., Zhu, C. & Shen, X. Catalytic asymmetric acyl halide–aldehyde cyclocondensation reactions of substituted ketenes J. Am. Chem. Soc. 126, 14–15 (2004).
Lectka, T. et al. Catalytic, asymmetric α-chlorination of acid halides. J. Am. Chem. Soc. 126, 4245–4255 (2004).
Morrill, L. C., Lebl, T., Slawin, A. M. A. & Smith, A. D. Catalytic asymmetric α-amination of carboxylic acids using isothioureas. Chem. Sci. 3, 2088–2093 (2012).
Abraham, C. J., Paull, D. H., Scerba, M. T., Grebinski, J. W. & Leckta, T. Catalytic, enantioselective bifunctional inverse electron demand hetero-Diels–Alder reactions of ketene enolates and o-benzoquinone diimides. J. Am. Chem. Soc. 128, 13370–13371 (2006).
Bekele, T. et al. Catalytic, enantioselective [4+2] cycloadditions of ketene enolates and o-quinones: efficient entry to chiral, α-oxygenated carboxylic acid derivatives. J. Am. Chem. Soc. 128, 1810–1811 (2006).
Wolfer, J., Bekele, T., Abraham, C. J., Dogo-Isonagie, C. & Lectka, T. Catalytic, asymmetric synthesis of 1,4-benzoxazinones: a remarkably enantioselective route to α-amino acid derivatives from o-benzoquinone imides. Angew. Chem. Int. Ed. 45, 7398–7400 (2006).
Belmessieri, D., Morrill, L. C., Simal, C., Slawin, A. M. Z. & Smith, A. D. Organocatalytic functionalization of carboxylic acids: isothiourea-catalyzed asymmetric intra- and intermolecular Michael addition-lactonizations. J. Am. Chem. Soc. 133, 2714–2720 (2011).
Simal, C., Lebl, T., Slawin, A. M. Z. & Smith, A. D. Dihydropyridones: catalytic asymmetric synthesis, N- to C-sulfonyl transfer, and derivatizations. Angew. Chem. Int. Ed. 51, 3653–3657 (2012).
Tiseni, P. S. & Peters, R. Catalytic asymmetric formation of δ-lactones by [4+2] cycloaddition of zwitterionic dienolates generated from α,β-unsaturated acid chlorides. Angew. Chem. Int. Ed. 46, 5325–5328 (2007).
Bappert, E., Muller, P. & Fu, G. C. Asymmetric [3+2] annulations catalyzed by a planar-chiral derivative of DMAP. Chem. Commun. 42, 2604–2606 (2006).
Robinson, E., Fallan, C., Simal, C., Slawin, A. & Smith, A. D. Anhydrides as α,β-unsaturated acyl ammonium precursors: isothiourea-promoted catalytic asymmetric annulation processes. Chem. Sci. 4, 2193–2200 (2013).
Böttcher, T. & Sieber, S. A. β-Lactams and β-lactones as activity-based probes in chemical biology. Med. Chem. Commun. 3, 408–417 (2012).
Yang, X. & Birman, V. B. Homobenzotetramisole-catalyzed kinetic resolution of α-aryl-, α-aryloxy-, and α-arylthioalkanoic acids. Adv. Synth. Catal. 351, 2301–2304 (2009).
Taylor, J. E., Bull, S. D. & Williams, J. M. J. Amidines, isothioureas, and guanidines as nucleophilic catalysts. Chem. Soc. Rev. 41, 2109–2121 (2012).
Blanchette, M. A. et. al. Horner–Wadsworth–Emmons reaction: use of lithium chloride and an amine for base-sensitive compounds. Tetrahedron Lett. 25, 2183–2186 (1984).
Birman, V. B. & Li, X. Homobenzotetramisole: an effective catalyst for kinetic resolution of aryl-cycloalkanols. Org. Lett. 6, 1115–1118 (2008).
Tsuji, H. et al. Palladium-catalyzed decarboxylation–allylation of allylic esters of α-substituted β-keto carboxylic, malonic, cyanoacetic, and nitroacetic acid. J. Org. Chem. 52, 2988–2995 (1997).
Lang, A. Gibberellins: structure and mechanism. Annu. Rev. Plant Physiol. 21, 537–570 (1970).
Wilds, A. L., Harnik, M., Shimizu, R. Z. & Tyner, D. A. Methods for total synthesis of steroids. XVII. Δ14–16-Keto steroid approach to ring d. H. Introduction of 17-caboxy group. Synthesis of 14α,17β and 14β,17α isomers of rac-estra-5(10),6,8-triene-17-carboxylic acid. J. Am. Chem. Soc. 88, 799–804 (1965).
Minami, I., Nisar, M., Yuhara, M., Shimizu, I. & Tsuji, M. New methods for the syntheses of α,β-unsaturated ketones, aldehydes, and nitriles by the palladium-catalyzed reactions of allyl β-oxo esters, allyl 1-alkenyl carbonates, and allyl α-cyano esters. Synthesis 992–999 (1987).
Candish, L. & Lupton, D. W. N-Heterocyclic carbene-catalyzed Ireland–Coates Claisen rearrangement: synthesis of functionalized β-lactones. J. Am. Chem. Soc. 135, 58–61 (2013).
Birman, V. B., Li, X. & Han, Z. Nonaromatic amidine derivatives as acylation catalysts. Org. Lett. 9, 37–40 (2007).
Seayad, J. & List, B. in Multicomponent Reactions (eds Zhu, J. & Bienaymé, H.) 277–299 (Wiley-VCH, 2005).
Wang, Y., Tennyson, R. & Romo, D. β-Lactones: intermediates for natural product total synthesis and new transformations. Heterocycles 64, 605–658 (2004).
Ranieri, B., Robles, O. & Romo, D. Concise synthesis of the isothiourea organocatalysts homobenzotetramisole and derivatives. J. Org. Chem. 78, 6291–6296 (2013).
Vellalath, S., Van, K. N. & Romo, D. Direct catalytic asymmetric synthesis of N-heterocycles from commodity acid chlorides by employing α,β-unsaturated acylammonium salts. Angew. Chem. Int. Ed. http://dx.doi.org/10.1002/anie.201306050 (2013).
Acknowledgements
This work was supported by the National Science Foundation (CHE- 1112397) and the Robert A. Welch Foundation (A-1280), together with partial support from the National Institutes of Health (GM 069874). The authors thank N. Harvey for synthesis of HBTM and assistance with early studies of the NCMAL. N. Bhuvanesh (Center for X-ray Analysis, TAMU) and W. K. Russell (Laboratory for Biological Mass Spectrometry, TAMU) secured X-ray crystal structure and mass data, respectively.
Author information
Authors and Affiliations
Contributions
G.L. initiated the studies of the α,β-unsaturated acylammonium intermediate from acid chlorides. D.R., G.L. and M.E.S. were involved in the design of experiments for exploration of the NCMAL. D.R. and K.N.V. conceived and developed the three-component NCMAL process. G.L., M.E.S., K.N.V. and R.L.M. performed the experiments. D.R., G.L. and M.E.S. composed the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 6254 kb)
Supplementary information
Crystallographic data for compound (+)-S16 (CIF 16 kb)
Supplementary information
Crystallographic data for compound (±)-14k (CIF 14 kb)
Supplementary information
Crystallographic data for compound (±)-14m (CIF 15 kb)
Supplementary information
Crystallographic data for compound (+)-14p (CIF 15 kb)
Supplementary information
Crystallographic data for compound (±)-14q (CIF 15 kb)
Supplementary information
Crystallographic data for compound (±)-14t (CIF 14 kb)
Supplementary information
Crystallographic data for compound (±)-14v (CIF 13 kb)
Supplementary information
Crystallographic data for compound (±)-14x (CIF 12 kb)
Supplementary information
Crystallographic data for compound (-)-24 (CIF 27 kb)
Rights and permissions
About this article
Cite this article
Liu, G., Shirley, M., Van, K. et al. Rapid assembly of complex cyclopentanes employing chiral, α,β-unsaturated acylammonium intermediates. Nature Chem 5, 1049–1057 (2013). https://doi.org/10.1038/nchem.1788
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1788
This article is cited by
-
Dynamic stereoselective annulation via aldol-oxa-cyclization cascade reaction to afford spirooxindole pyran polycycles
Communications Chemistry (2019)