Abstract
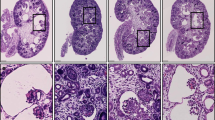
Defects in the structure or function of the primary cilium, an antennae-like structure whose functional integrity has been linked to the suppression of uncontrolled kidney epithelial cell proliferation, are a common feature of genetic disorders characterized by kidney cysts1,2. However, the mechanisms by which primary cilia are maintained remain poorly defined. von Hippel-Lindau (VHL) disease is characterized by the development of premalignant renal cysts and arises because of functional inactivation of the VHL tumour suppressor gene product, pVHL3,4. Here, we show that pVHL and glycogen synthase kinase (GSK)3β are key components of an interlinked signalling pathway that maintains the primary cilium. Although inactivation of either pVHL or GSK3β alone did not affect cilia maintenance, their combined inactivation leads to loss of cilia. In VHL patients, GSK3β is subjected to inhibitory phosphorylation in renal cysts, but not in early VHL mutant lesions, and these cysts exhibit reduced frequencies of primary cilia. We propose that pVHL and GSK3β function together in a ciliary-maintenance signalling network, disruption of which enhances the vulnerability of cells to lose their cilia, thereby promoting cyst formation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Davenport, J. R. & Yoder, B. K. An incredible decade for the primary cilium: a look at a once-forgotten organelle. Am. J. Physiol. Renal Physiol. 289, F1159–F1169 (2005).
Eley, L., Yates, L. M. & Goodship, J. A. Cilia and disease. Curr. Opin. Genet. Dev. 15, 308–314 (2005).
Choyke, P. L. et al. The natural history of renal lesions in von Hippel-Lindau disease: a serial CT study in 28 patients. Am. J. Roentgenol. 159, 1229–1234 (1992).
Solomon, D. & Schwartz, A. Renal pathology in von Hippel-Lindau disease. Human Pathol. 19, 1072–1079 (1988).
Maxwell, P. H. et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 399, 271–275 (1999).
Hergovich, A., Lisztwan, J., Barry, R., Ballschmieter, P. & Krek, W. Regulation of microtubule stability by the von Hippel-Lindau tumour suppressor protein pVHL. Nature Cell Biol. 5, 64–70 (2003).
Lolkema, M. P. et al. The von Hippel-Lindau tumor suppressor protein influences microtubule dynamics at the cell periphery. Exp. Cell Res. 301, 139–146 (2004).
Praetorius, H. A., Praetorius, J., Nielsen, S., Frokiaer, J. & Spring, K. R. β1-integrins in the primary cilium of MDCK cells potentiate fibronectin-induced Ca2+ signaling. Am. J. Physiol. Renal. Physiol. 287, F969–F978 (2004).
Schneider, L. et al. PDGFRα signaling is regulated through the primary cilium in fibroblasts. Curr. Biol. 15, 1861–1866 (2005).
Praetorius, H. A. & Spring, K. R. Bending the MDCK cell primary cilium increases intracellular calcium. J. Membr. Biol. 184, 71–79 (2001).
Praetorius, H. A. & Spring, K. R. Removal of the MDCK cell primary cilium abolishes flow sensing. J. Membr. Biol. 191, 69–76 (2003).
Alieva, I. B., Gorgidze, L. A., Komarova, Y. A., Chernobelskaya, O. A. & Vorobjev, I. A. Experimental model for studying the primary cilia in tissue culture cells. Membr. Cell Biol. 12, 895–905 (1999).
Haase, V. H., Glickman, J. N., Socolovsky, M. & Jaenisch, R. Vascular tumors in livers with targeted inactivation of the von Hippel-Lindau tumor suppressor. Proc. Natl Acad. Sci. USA 98, 1583–1588 (2001).
Dickins, R. A. et al. Probing tumor phenotypes using stable and regulated synthetic microRNA precursors. Nature Genet. 37, 1289–1295 (2005).
Esteban, M. A., Harten, S. K., Tran, M. G. & Maxwell, P. H. Formation of primary cilia in the renal epithelium is regulated by the von Hippel-Lindau tumor suppressor protein. J. Am. Soc. Nephrol. 17, 1801–1806 (2006).
Lutz, M. S. & Burk, R. D. Primary cilium formation requires von hippel-lindau gene function in renal-derived cells. Cancer Res. 66, 6903–6907 (2006).
Schermer, B. et al. The von Hippel-Lindau tumor suppressor protein controls ciliogenesis by orienting microtubule growth. J. Cell Biol. 175, 547–554 (2006).
Zhou, F. Q. & Snider, W. D. Cell biology. GSK-3β and microtubule assembly in axons. Science 308, 211–214 (2005).
Wilson, N. F. & Lefebvre, P. A. Regulation of flagellar assembly by glycogen synthase kinase 3 in Chlamydomonas reinhardtii. Eukaryot. Cell 3, 1307–1319 (2004).
Hergovich, A. et al. Priming-dependent phosphorylation and regulation of the tumor suppressor pVHL by glycogen synthase kinase 3. Mol. Cell Biol. 26, 5784–5796 (2006).
Cross, D. A., Alessi, D. R., Cohen, P., Andjelkovich, M. & Hemmings, B. A. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 378, 785–789 (1995).
Wei, W., Jin, J., Schlisio, S., Harper, J. W. & Kaelin, W. G., Jr. The v-Jun point mutation allows c-Jun to escape GSK3-dependent recognition and destruction by the Fbw7 ubiquitin ligase. Cancer Cell 8, 25–33 (2005).
Hoeflich, K. P. et al. Requirement for glycogen synthase kinase-3β in cell survival and NF-κB activation. Nature 406, 86–90 (2000).
Knauth, K., Bex, C., Jemth, P. & Buchberger, A. Renal cell carcinoma risk in type 2 von Hippel-Lindau disease correlates with defects in pVHL stability and HIF-1α interactions. Oncogene 25, 370–377 (2006).
Frame, S. & Cohen, P. GSK3 takes centre stage more than 20 years after its discovery. Biochem J. 359, 1–16 (2001).
Mandriota, S. J. et al. HIF activation identifies early lesions in VHL kidneys: evidence for site-specific tumor suppressor function in the nephron. Cancer Cell 1, 459–468 (2002).
Lin, F. et al. Kidney-specific inactivation of the KIF3A subunit of kinesin-II inhibits renal ciliogenesis and produces polycystic kidney disease. Proc. Natl Acad. Sci. USA 100, 5286–5291 (2003).
Pazour, G. J. et al. Chlamydomonas IFT88 and its mouse homologue, polycystic kidney disease gene tg737, are required for assembly of cilia and flagella. J. Cell Biol. 151, 709–718 (2000).
Rankin, E. B., Tomaszewski, J. E. & Haase, V. H. Renal cyst development in mice with conditional inactivation of the von Hippel-Lindau tumor suppressor. Cancer Res 66, 2576–2583 (2006).
Brauch, H. et al. Von Hippel-Lindau (VHL) disease with pheochromocytoma in the Black Forest region of Germany: evidence for a founder effect. Hum. Genet. 95, 551–556 (1995).
Chen, F. et al. Germline mutations in the von Hippel-Lindau disease tumor suppressor gene: correlations with phenotype. Hum. Mutat. 5, 66–75 (1995).
Klein, P. S. & Melton, D. A. A molecular mechanism for the effect of lithium on development. Proc. Natl Acad. Sci. USA 93, 8455–8459 (1996).
Pastorekova, S., Zavadova, Z., Kostal, M., Babusikova, O. & Zavada, J. A novel quasi-viral agent, MaTu, is a two-component system. Virology 187, 620–626 (1992).
Lisztwan, J., Imbert, G., Wirbelauer, C., Gstaiger, M. & Krek, W. The von Hippel-Lindau tumor suppressor protein is a component of an E3 ubiquitin-protein ligase activity. Genes Dev. 13, 1822–1833 (1999).
Acknowledgements
We thank all members of our laboratory for helpful discussions. The authors are grateful to A. Berns, I. Verma, D. Trono, S. Lowe, J. Woodgett, P. Maxwell and J. Zavada for their generous gifts of reagents, to M. Hitz for assistance with mouse breeding and maintenance, and to the Light Microscopy Centre ETH Zurich, in particular G. Csucs and J. Kusch, for help with microscopy. This work was supported by grants from the Dr. Josef Steiner Foundation and Swiss National Foundation (W.K) and from the Serono Foundation for the Advancement of Medical Science (I.J.F).
Author information
Authors and Affiliations
Contributions
Project conception, planning, data analysis and manuscript preparation were performed by C.R.T., I.J.F. and W.K. C.R.T. and I.J.F. contributed equally to experimental work. C.R.H. contributed to Supplementary Information, Fig. S4c, d. M.M. and M.H. contributed VHL patient material.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures S1, S2, S3, S4, S5 and S6 (PDF 783 kb)
Rights and permissions
About this article
Cite this article
Thoma, C., Frew, I., Hoerner, C. et al. pVHL and GSK3β are components of a primary cilium-maintenance signalling network. Nat Cell Biol 9, 588–595 (2007). https://doi.org/10.1038/ncb1579
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1579
This article is cited by
-
Clear cell renal cell carcinoma with cystic component similar to multilocular cystic renal neoplasm of low malignant potential: a rare pattern of cyst-dependent progression from multilocular cystic renal neoplasm of low malignant potential
Diagnostic Pathology (2023)
-
VHL suppresses RAPTOR and inhibits mTORC1 signaling in clear cell renal cell carcinoma
Scientific Reports (2021)
-
ARP-T1-associated Bazex–Dupré–Christol syndrome is an inherited basal cell cancer with ciliary defects characteristic of ciliopathies
Communications Biology (2021)
-
Therapeutically actionable signaling node to rescue AURKA driven loss of primary cilia in VHL-deficient cells
Scientific Reports (2021)
-
HIF-1α and HIF-2α differently regulate tumour development and inflammation of clear cell renal cell carcinoma in mice
Nature Communications (2020)