Abstract
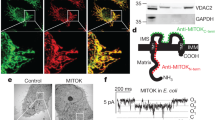
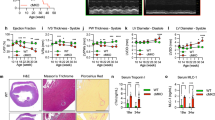
Mitochondria are critically involved in necrotic cell death induced by Ca2+ overload, hypoxia and oxidative damage. The mitochondrial permeability transition (MPT) pore — a protein complex that spans both the outer and inner mitochondrial membranes — is considered the mediator of this event and has been hypothesized to minimally consist of the voltage-dependent anion channel (Vdac) in the outer membrane, the adenine-nucleotide translocase (Ant) in the inner membrane and cyclophilin-D in the matrix1,2,3. Here, we report the effects of deletion of the three mammalian Vdac genes on mitochondrial-dependent cell death. Mitochondria from Vdac1-, Vdac3-, and Vdac1–Vdac3-null mice exhibited a Ca2+- and oxidative stress-induced MPT that was indistinguishable from wild-type mitochondria. Similarly, Ca2+- and oxidative-stress-induced MPT and cell death was unaltered, or even exacerbated, in fibroblasts lacking Vdac1, Vdac2, Vdac3, Vdac1–Vdac3 and Vdac1–Vdac2–Vdac3. Wild-type and Vdac-deficient mitochondria and cells also exhibited equivalent cytochrome c release, caspase cleavage and cell death in response to the pro-death Bcl-2 family members Bax and Bid. These results indicate that Vdacs are dispensable for both MPT and Bcl-2 family member-driven cell death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Zamzami, N. & Kroemer G. The mitochondrion in apoptosis: how Pandora's box opens. Nature Rev. Mol. Cell. Biol. 2, 67–71 (2001).
Crompton, M., Barksby, E., Johnson, N. & Capano, M. Mitochondrial intermembrane junctional complexes and their involvement in cell death. Biochimie 84, 143–152 (2002).
Halestrap, A. P. Calcium, mitochondria and reperfusion injury: a pore way to die. Biochem. Soc. Trans. 34, 232–237 (2006).
Rostovtseva, T. K., Tan, W. & Colombini, M. On the role of VDAC in apoptosis: fact and fiction. J. Bioenerg. Biomembr. 37, 129–142 (2005).
Blachly-Dyson. E. & Forte, M. VDAC channels. IUBMB Life 52, 113–118 (2001).
Wu, S., Sampson, M. J., Decker, W. K. & Craigen, W. J. Each mammalian mitochondrial outer membrane porin protein is dispensable: effects on cellular respiration. Biochim. Biophys. Acta. 1452, 68–78 (1999).
Anflous, K., Armstrong, D. D. & Craigen, W. J. Altered mitochondrial sensitivity for ADP and maintenance of creatine-stimulated respiration in oxidative striated muscles from VDAC1-deficient mice. J. Biol. Chem. 276, 1954–1960 (2001).
Sampson, M. J. et al. Immotile sperm and infertility in mice lacking mitochondrial voltage-dependent anion channel type 3. J. Biol. Chem. 276, 39206–39212 (2001).
Krauskopf, A., Eriksson, O., Craigen, W. J., Forte, M. A. & Bernardi, P. Properties of the permeability transition in VDAC1−/− mitochondria. Biochim. Biophys. Acta. 1757, 590–595 (2006).
Sheiko, T. V., Fisher, J. K., Craigen, W. J. & Korsmeyer, S. J. VDAC2 inhibits BAK activation and mitochondrial apoptosis. Science 301, 513–517 (2003).
Banerjee, J. & Ghosh, S. Bax increases the pore size of rat brain mitochondrial voltage-dependent anion channel in the presence of tBid. Biochem. Biophys. Res. Commun. 323, 310–314 (2004).
Shimizu, S., Ide, T., Yanagida, T. & Tsujimoto, Y. Electrophysiological study of a novel large pore formed by Bax and the voltage-dependent anion channel that is permeable to cytochrome c. J. Biol. Chem. 275, 12321–12325 (2000).
Sugiyama, T., Shimizu, S., Matsuoka, Y., Yoneda, Y. & Tsujimoto, Y. Activation of mitochondrial voltage-dependent anion channel by a pro-apoptotic BH3-only protein Bim. Oncogene 21, 4944–4956 (2002).
Shimizu, S., Narita, M. & Tsujimoto, Y. Bcl-2 family proteins regulate the release of apoptogenic cytochrome c by the mitochondrial channel VDAC. Nature 399, 483–487 (1999).
Petronilli, V. et al. Transient and long-lasting openings of the mitochondrial permeability transition pore can be monitored directly in intact cells by changes in mitochondrial calcein fluorescence. Biophys. J. 76, 725–734 (1999).
Baines, C. P. et al. Loss of cyclophilin D reveals a critical role for mitochondrial permeability transition in cell death. Nature 434, 658–662 (2005).
Bernardi, P. et al. The mitochondrial permeability transition from in vitro artifact to disease target. FEBS. J. 273, 2077–2099 (2006).
Halestrap, A. P., McStay, G. P. & Clarke, S. J. The permeability transition pore complex: another view. Biochimie 84, 153–166 (2002).
Nakagawa, T. et al. Cyclophilin D-dependent mitochondrial permeability transition regulates some necrotic but not apoptotic cell death. Nature 434, 652–658 (2005).
Basso, E. et al. Properties of the permeability transition pore in mitochondria devoid of cyclophilin D. J. Biol. Chem. 280, 18558–18561 (2005).
Schinzel, A. C. et al. Cyclophilin D is a component of mitochondrial permeability transition and mediates neuronal cell death after focal cerebral ischemia. Proc. Natl Acad. Sci. USA 102, 12005–12010 (2005).
Kokoszka, J. E. et al. The ADP/ATP translocator is not essential for the mitochondrial permeability transition pore. Nature 427, 461–465 (2004).
Crompton, M., Virji, S. & Ward, J. M. Cyclophilin-D binds strongly to complexes of the voltage-dependent anion channel and the adenine nucleotide translocase to form the permeability transition pore. Eur. J. Biochem. 258, 729–735 (1998).
Woodfield, K., Ruck, A., Brdiczka, D. & Halestrap, A. P. Direct demonstration of a specific interaction between cyclophilin-D and the adenine nucleotide translocase confirms their role in the mitochondrial permeability transition. Biochem. J. 336, 287–290 (1998).
Vander Heiden, M. G., et al. Outer mitochondrial membrane permeability can regulate coupled respiration and cell survival. Proc. Natl Acad. Sci. USA 97, 4666–4671 (2000).
Vander Heiden, M. G. et al. Bcl-xL promotes the open configuration of the voltage-dependent anion channel and metabolite passage through the outer mitochondrial membrane. J. Biol. Chem. 276, 19414–19419 (2001).
Lai, J. C. et al. A pharmacologic target of G3139 in melanoma cells may be the mitochondrial VDAC. Proc. Natl Acad. Sci. USA 103, 7494–7499 (2006).
Priault, M., Chaudhuri, B., Clow, A., Camougrand, N. & Manon, S. Investigation of bax-induced release of cytochrome c from yeast mitochondria permeability of mitochondrial membranes, role of VDAC and ATP requirement. Eur. J. Biochem. 260, 684–691 (1999).
Polcic, P. & Forte, M. Response of yeast to the regulated expression of proteins in the Bcl-2 family. Biochem. J. 374, 393–402 (2003).
Rostovtseva, T. K. et al. Bid, but not Bax, regulates VDAC channels. J. Biol. Chem. 279, 13575–13583 (2004).
Antonsson, B., Montessuit, S., Lauper, S., Eskes, R. & Martinou, J. C. Bax oligomerization is required for channel-forming activity in liposomes and to trigger cytochrome c release from mitochondria. Biochem. J. 345, 271–278 (2000).
Kagawa, S., et al. A binary adenoviral vector system for expressing high levels of the proapoptotic gene bax. Gene Ther. 7, 75–79 (2000).
Kaiser, R. A., et al. Targeted inhibition of p38 mitogen-activated protein kinase antagonizes cardiac injury and cell death following ischemia-reperfusion in vivo. J. Biol. Chem. 279, 15524–15530 (2004).
Acknowledgements
We are very grateful to B. Fang for the generous gift of the Bax adenovirus, R. Gottlieb for recombinant Bax and tBid, and the late S. Korsmeyer for Bax–Bak gene-targeted mice. This work was supported by grants from the National Institutes of Health (J.D.M. and W.J.C.), an American Heart Association Scientist Development Grant (C.P.B.) and Established Investigator Grant (J.D.M.), a National Institutes of Health NRSA award (R.A.K.), The Children's Hospital Research Foundation (C.P.B.), and the Fondation Leducq (Heart failure network grant to J.D.M).
Author information
Authors and Affiliations
Contributions
C.P.B. and J.D.M. conceived the research project, C.P.B. performed all experimentation except the ischaemia–reperfusion experiment, which was performed by R.A.K. T.S. and W.J.C. provided critical reagents and valuable discussion.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4 and S5 (PDF 663 kb)
Rights and permissions
About this article
Cite this article
Baines, C., Kaiser, R., Sheiko, T. et al. Voltage-dependent anion channels are dispensable for mitochondrial-dependent cell death. Nat Cell Biol 9, 550–555 (2007). https://doi.org/10.1038/ncb1575
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1575
This article is cited by
-
Identity, structure, and function of the mitochondrial permeability transition pore: controversies, consensus, recent advances, and future directions
Cell Death & Differentiation (2023)
-
Molecular mechanisms of mitochondrial DNA release and activation of the cGAS-STING pathway
Experimental & Molecular Medicine (2023)
-
Construction and Evaluation of an M2 Macrophage-Related Prognostic Model for Colon Cancer
Applied Biochemistry and Biotechnology (2023)
-
Kastor and Polluks polypeptides encoded by a single gene locus cooperatively regulate VDAC and spermatogenesis
Nature Communications (2022)
-
Molecular mechanisms and consequences of mitochondrial permeability transition
Nature Reviews Molecular Cell Biology (2022)