Abstract
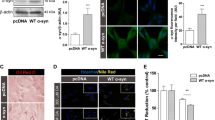
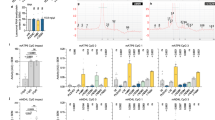
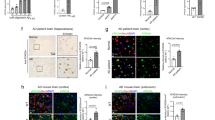
Amyloid beta peptide (Aβ) has a key role in the pathological process of Alzheimer's disease (AD), but the physiological function of Aβ and of the amyloid precursor protein (APP) is unknown1,2. Recently, it was shown that APP processing is sensitive to cholesterol and other lipids3,4,5,6,7,8,9,10. Hydroxymethylglutaryl-CoA reductase (HMGR) and sphingomyelinases (SMases) are the main enzymes that regulate cholesterol biosynthesis and sphingomyelin (SM) levels, respectively. We show that control of cholesterol and SM metabolism involves APP processing. Aβ42 directly activates neutral SMase and downregulates SM levels, whereas Aβ40 reduces cholesterol de novo synthesis by inhibition of HMGR activity. This process strictly depends on γ-secretase activity. In line with altered Aβ40/42 generation, pathological presenilin mutations result in increased cholesterol and decreased SM levels. Our results demonstrate a biological function for APP processing and also a functional basis for the link that has been observed between lipids and Alzheimer's disease (AD).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hardy, J. & Selkoe, D. J. The amyloid hypothesis of Alzheimer's disease: progress and problems on the road to therapeutics. Science 297, 353–356 (2002).
Sisodia, S. S. & St George-Hyslop, P. H. γ-Secretase, Notch, Aβ and Alzheimer's disease: where do the presenilins fit in? Nature Rev. Neurosci. 3, 281–290 (2002).
Wahrle, S. et al. Cholesterol-dependent γ-secretase activity in buoyant cholesterol-rich membrane microdomains. Neurobiol. Dis. 9, 11–23 (2002).
Burns, M. et al. Presenilin redistribution associated with aberrant cholesterol transport enhances β-amyloid production in vivo. J. Neurosci. 23, 5645–5649 (2003).
Puglielli, L., Tanzi, R. E. & Kovacs, D. M. Alzheimer's disease: the cholesterol connection. Nature Neurosci. 6, 345–351 (2003).
Fassbender, K. et al. Simvastatin strongly reduces levels of Alzheimer's disease β-amyloid peptides Aβ42 and Aβ40 in vitro and in vivo. Proc. Natl Acad. Sci. USA 98, 5856–5861 (2001).
Runz, H. et al. Inhibition of intracellular cholesterol transport alters presenilin localization and amyloid precursor protein processing in neuronal cells. J. Neurosci. 22, 1679–1689 (2002).
Yamazaki, T., Chang, T. Y., Haass, C. & Ihara, Y. Accumulation and aggregation of amyloid β-protein in late endosomes of Niemann-pick type C cells. J. Biol. Chem. 276, 4454–4460 (2001).
Sawamura, N. et al. Modulation of amyloid precursor protein cleavage by cellular sphingolipids. J. Biol. Chem. 279, 11984–11991 (2004).
Refolo, L. M. et al. A cholesterol-lowering drug reduces β-amyloid pathology in a transgenic mouse model of Alzheimer's disease. Neurobiol. Dis. 8, 890–899 (2001).
Herreman, A. et al. Total inactivation of γ-secretase activity in presenilin-deficient embryonic stem cells. Nature Cell Biol. 2, 461–462 (2000).
Grziwa, B. et al. The transmembrane domain of the amyloid precursor protein in microsomal membranes is on both sides shorter than predicted. J. Biol. Chem. 278, 6803–6808 (2003).
Brown, M. S., Ye, J., Rawson, R. B. & Goldstein, J. L. Regulated intramembrane proteolysis: a control mechanism conserved from bacteria to humans. Cell 100, 391–398 (2000).
Simons, M. et al. Cholesterol depletion inhibits the generation of β-amyloid in hippocampal neurons. Proc. Natl Acad. Sci. USA 95, 6460–6464 (1998).
Abad-Rodriguez, J. et al. Neuronal membrane cholesterol loss enhances amyloid peptide generation. J. Cell Biol. 167, 953–960 (2004).
Simons, M. et al. Treatment with simvastatin in normocholesterolemic patients with Alzheimer's disease: a 26-week randomized, placebo-controlled, double-blind trial. Ann. Neurol. 52, 346–350 (2002).
Shoji, M. et al. The levels of cerebrospinal fluid Aβ40 and Aβ42(43) are regulated age-dependently. Neurobiol. Aging 22, 209–215 (2001).
Cutler, R. G. et al. Involvement of oxidative stress-induced abnormalities in ceramide and cholesterol metabolism in brain aging and Alzheimer's disease. Proc. Natl Acad. Sci. USA 101, 2070–2075 (2004).
Pappolla, M. A. et al. Mild hypercholesterolemia is an early risk factor for the development of Alzheimer amyloid pathology. Neurology 61, 199–205 (2003).
Wolozin, B., Kellman, W., Ruosseau, P., Celesia, G. G. & Siegel, G. Decreased prevalence of Alzheimer disease associated with 3-hydroxy-3- methyglutaryl coenzyme A reductase inhibitors. Arch. Neurol. 57, 1439–1443 (2000).
Sparks, D. L. et al. Atorvastatin for the treatment of mild to moderate Alzheimer disease: preliminary results. Arch. Neurol. 62, 753–757 (2005).
Saura, C. A. et al. Loss of presenilin function causes impairments of memory and synaptic plasticity followed by age-dependent neurodegeneration. Neuron 42, 23–36 (2004).
Beglopoulos, V. et al. Reduced β-amyloid production and increased inflammatory responses in presenilin conditional knock-out mice. J. Biol. Chem. 279, 46907–46914 (2004).
Ida, N. et al. Analysis of heterogeneous βA4 peptides in human cerebrospinal fluid and blood by a newly developed sensitive Western blot assay. J. Biol. Chem. 271, 22908–22914 (1996).
Lee, M. K. et al. Expression of presenilin 1 and 2 (PS1 and PS2) in human and murine tissues. J. Neurosci. 16, 7513–7525 (1996).
Slunt, H. H. et al. Expression of a ubiquitous, cross-reactive homologue of the mouse β-amyloid precursor protein (APP). J. Biol. Chem. 269, 2637–2644 (1994).
Jana, A. & Pahan, K. Fibrillar amyloid-β peptides kill human primary neurons via NADPH oxidase-mediated activation of neutral sphingomyelinase: implications for Alzheimer's disease. J. Biol. Chem. 279, 51451–51459 (2004).
Qi, X. L. et al. Oxidative stress induced by β-amyloid peptide (1–42) is involved in the altered composition of cellular membrane lipids and the decreased expression of nicotinic receptors in human SH-SY5Y neuroblastoma cells. Neurochem. Int. 46, 613–621 (2005).
Haze, K., Yoshida, H., Yanagi, H., Yura, T. & Mori, K. Mammalian transcription factor ATF6 is synthesized as a transmembrane protein and activated by proteolysis in response to endoplasmic reticulum stress. Mol. Biol. Cell 10, 3787–3799 (1999).
Huitema, K., van den Dikkenberg, J., Brouwers, J. F. & Holthuis, J. C. Identification of a family of animal sphingomyelin synthases. EMBO J. 23, 33–44 (2004).
Acknowledgements
We are grateful to I. Tomic and R. Stammann for excellent technical assistance; B. Penke for peptides; T. Ruppert for mass-spectrometry advice and support; and for funding received from the European Union via QLK-172-2002 Lipidiet, Deutsche Forschungsgemeinschaft and Bundesministerium für Bildung und Forschung.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures S1, S2, S3 ,S4 and Supplementary Materials and Methods plus Supplementary References (PDF 104 kb)
Rights and permissions
About this article
Cite this article
Grimm, M., Grimm, H., Pätzold, A. et al. Regulation of cholesterol and sphingomyelin metabolism by amyloid-β and presenilin. Nat Cell Biol 7, 1118–1123 (2005). https://doi.org/10.1038/ncb1313
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1313
This article is cited by
-
Brain areas lipidomics in female transgenic mouse model of Alzheimer's disease
Scientific Reports (2024)
-
Lipidomic analysis of brain and hippocampus from mice fed with high-fat diet and treated with fecal microbiota transplantation
Nutrition & Metabolism (2023)
-
Reexamining the Causes and Effects of Cholesterol Deposition in the Brains of Patients with Alzheimer’s Disease
Molecular Neurobiology (2023)
-
The role of DHCR24 in the pathogenesis of AD: re-cognition of the relationship between cholesterol and AD pathogenesis
Acta Neuropathologica Communications (2022)
-
Multi-Omic analyses characterize the ceramide/sphingomyelin pathway as a therapeutic target in Alzheimer’s disease
Communications Biology (2022)