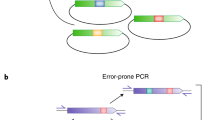
Abstract
Evolution of eukaryotes is mediated by sexual recombination of parental genomes. Crossovers occur in random, but homologous, positions at a frequency that depends on DNA length. As exons occupy only 1% of the human genome and introns about 24%, by far most of the crossovers occur between exons, rather than inside. The natural process of creating new combinations of exons by intronic recombination is called exon shuffling. Our group is developing in vitro formats for exon shuffling and applying these to the directed evolution of proteins. Based on the splice frame junctions, nine classes of exons and three classes of introns can be distinguished. Splice frame diagrams of natural genes show how the splice frame rules govern exon shuffling. Here, we review various approaches to constructing libraries of exon-shuffled genes. For example, exon shuffling of human pharmaceutical proteins can generate libraries in which all of the sequences are fully human, without the point mutations that raise concerns about immunogenicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gilbert, W. Why genes in pieces? Nature 271, 501 (1978).
Kuiper, J. et al. Interaction of mutants of tissue-type plasminogen activator with liver cells: effect of domain deletions. Biochem. J. 313, 775–780 (1996).
Browne, M.J. et al. A tissue-type plasminogen activator mutant with prolonged clearance in vivo. Effect of removal of the growth factor domain. J. Biol. Chem. 263, 1599–1602 (1988).
Doolittle, R.F. The multiplicity of domains in proteins. Annu. Rev. Biochem. 64, 287–314 (1995).
Bork, P. Mobile modules and motifs. Curr. Opin. Struct. Biol. 2, 413–421 (1992).
Patthy, L. Modular exchange principles in proteins. Curr. Opin. Struct. Biol. 1, 351–361 (1991).
Patthy, L. Introns and exons. Curr. Opin. Struct. Biol. 4, 383–392 (1994).
Belfort, M. & Perlman, P.S. Mechanisms of intron mobility. J. Biol. Chem. 270, 30237–30240 (1995).
Patthy, L. Intron-dependent evolution: preferred types of exons and introns. FEBS Lett. 214, 1–7 (1987).
Sharp, P.A. Speculations on RNA splicing. Cell 23, 643–646 (1981).
Patthy, L. Evolution of the proteases of blood coagulation and fibrinolysis by assembly from modules. Cell 41, 657–663 (1985).
Tordai, H., Banyai, L. & Patthy, L. The PAN module: the N-terminal domains of plasminogen and hepatocyte growth factor are homologous with the apple domains of the prekallikrein family and with a novel domain found in numerous nematode proteins. FEBS Lett. 461, 63–67 (1999).
Gitschier, J. et al. Characterization of the human factor VIII gene. Nature 312, 326–330 (1984).
Cripe, L.D., Moore, K.D., & Kane, W.H. Structure of the gene for human coagulation factor V. Biochemistry 31, 3777–3385 (1992).
Bottenus, R.E., Ichinose, A. &, Davie, E.W. Nucleotide sequence of the gene for the b subunit of human factor XIII. Biochemistry 29, 11195–11209 (1990).
Mancuso, D.J. et al. Structure of the gene for human von Willebrand factor. J. Biol. Chem. 264, 19514–19527 (1989).
Crameri, A., Cwirla, S. & Stemmer, W.P. Construction and evolution of antibody-phage libraries by DNA shuffling. Nat. Med. 2, 100–102 (1996).
Lollar, P., Parker, E.T. & Fay P.J. Coagulant properties of hybrid human/porcine factor VIII molecules. J. Biol. Chem. 267, 23652–23657 (1992).
Chang, J.Y., Monroe, D.M., Stafford, D.W., Brinkhous, K.M. & Roberts, H.R. Replacing the first epidermal growth factor-like domain of factor IX with that of factor VII enhances activity in vitro and in canine hemophilia B. J. Clin. Invest. 100, 886–892 (1997).
Langer-Safer, P.R. et al. Replacement of finger and growth factor domains of tissue plasminogen activator with plasminogen kringle 1. Biochemical and pharmacological characterization of a novel chimera containing a high affinity fibrin-binding domain linked to a heterologous protein. J. Biol. Chem. 266, 3715–3723 (1991).
Bell, G.I. et al. Human epidermal growth factor precursor: cDNA sequence, expression in vitro and gene organization. Nucleic Acids Res. 14, 8427–8446 (1986).
Ny, T., Elgh, F. & Lund, B. The structure of the human tissue-type plasminogen activator gene: correlation of intron and exon structures to functional and structural domains. Proc. Natl. Acad. Sci. USA 81, 5355–5359 (1984).
Leytus, S.P., Foster, D.C., Kurachi, K. & Davie, E.W. Gene for human factor X: a blood coagulation factor whose gene organization is essentially identical with that of factor IX and protein C. Biochemistry 25, 5098–5102 (1986).
Miyazawa, K., Kitamura, A. & Kitamura, N. Structural organization and the transcription initiation site of the human hepatocyte growth factor gene. Biochemistry 30, 9170–9176 (1991).
Degen, S.J. & Davie, E.W. Nucleotide sequence of the gene for human prothrombin. Biochemistry 26, 6165–6177 (1987).
Patel, R.S., Odermatt, E., Schwarzbauer, J.E. & Hynes, R.O. Organization of the fibronectin gene provides evidence for exon shuffling during evolution. EMBO J. 6, 2565–2572 (1987).
Cool, D.E. & MacGillivray, R.T. Characterization of the human blood coagulation factor XII gene. Intron/exon gene organization and analysis of the 5′-flanking region. J. Biol. Chem. 262, 13662–13673 (1987).
Kim, S.J., Ruiz, N., Bezouska, K. & Drickamer K. Organization of the gene encoding the human macrophage mannose receptor (MRC1). Genomics 14, 721–727 (1992).
Schwarzbauer, J.E., Patel, R.S., Fonda, D. & Hynes, R.O. Multiple sites of alternative splicing of the rat fibronectin gene transcript. EMBO J. 6, 2573–2580 (1987).
Giger, R.J. et al. The gene of chicken axonin–1. Complete structure and analysis of the promoter. Eur. J. Biochem. 227, 617–628 (1995).
del Castillo, I., Cohen-Salmon, M., Blanchard, S., Lutfalla, G. & Petit, C. Structure of the X-linked Kallmann syndrome gene and its homologous pseudogene on the Y chromosome. Nat. Genet. 2, 305–310 (1992).
Petersen, T.E., Martzen, M.R., Ichinose, A. & Davie, E.W. Characterization of the gene for human plasminogen, a key proenzyme in the fibrinolytic system. J. Biol. Chem. 265, 6104–6111 (1990).
Beaubien, G. et al. Gene structure and chromosomal localization of plasma kallikrein. Biochemistry 30, 1628–1635 (1991).
Asakai, R., Davie, E.W. & Chung, D.W. Organization of the gene for human factor XI. Biochemistry 26 7221–7228 (1987).
Schmidel, D.K., Tatro, A.V., Phelps, L.G., Tomczak, J.A. & Long, G.L. Organization of the human protein S genes. Biochemistry 29, 7845–7852 (1990).
Edenbrandt, C.M., Lundwall, A., Wydro, R. & Stenflo, J. Molecular analysis of the gene for vitamin K dependent protein S and its pseudogene. Cloning and partial gene organization. Biochemistry 29, 7861–7868 (1990).
Ploos van Amstel, H.K., Reitsma, P.H., van der Logt C.P. & Bertina, R.M. Intron-exon organization of the active human protein S gene PS alpha and its pseudogene PS beta: duplication and silencing during primate evolution. Biochemistry 29 7853–7861 (1990).
Johnston, G.I., Bliss, G.A., Newman, P.J. & McEver, R.P. Structure of the human gene encoding granule membrane protein–140, a member of the selectin family of adhesion receptors for leukocytes. J. Biol. Chem. 265, 21381–21385 (1990).
Bensi, G., Raugei, G., Klefenz, H. & Cortese, R. Structure and expression of the human haptoglobin locus. EMBO J. 4, 119–126 (1985).
Girard, T.J. et al. Structure of the human lipoprotein-associated coagulation inhibitor gene. Intro/exon gene organization and localization of the gene to chromosome 2. J. Biol. Chem. 266, 5036–5041 (1991).
Christiano, A.M. et al. Structural organization of the human type VII collagen gene (COL7A1), composed of more exons than any previously characterized gene. Genomics 21, 169–179 (1994).
Creighton, T.E. & Charles, I.G. Sequences of the genes and polypeptide precursors for two bovine protease inhibitors. J. Mol. Biol. 194, 11–22 (1987).
Shimasaki, S. et al. Primary structure of the human follistatin precursor and its genomic organization. Proc. Natl. Acad. Sci. USA 85, 4218–4222 (1988).
Villarreal, X.C., Mann, K.G. & Long, G.L. Structure of human osteonectin based upon analysis of cDNA and genomic sequences. Biochemistry 28, 6483–6491 (1989).
Scott, M.J. et al. Ovoinhibitor introns specify functional domains as in the related and linked ovomucoid gene. J. Biol. Chem. 262, 5899–5907 (1987).
Gott, P. et al. Human trefoil peptides: genomic structure in 21q22.3 and coordinated expression. Eur. J. Hum. Genet. 4, 308–315 (1996).
Metsaranta, M., Toman, D., de Crombrugghe, B. & Vuorio, E. Mouse type II collagen gene. Complete nucleotide sequence, exon structure, and alternative splicing. J. Biol. Chem. 266, 16862–16869 (1991).
Martinerie, C. et al. Structural analysis of the human nov proto-oncogene and expression in Wilms tumor. Oncogene 9, 2729–2732 (1994).
Lawler, J. et al. Characterization of the murine thrombospondin gene. Genomics 11, 587–600 (1991).
Ogata, R.T., Rosa, P.A. & Zepf, N.E. Sequence of the gene for murine complement component C4. J. Biol. Chem. 264, 16565–16572 (1989).
Grassel, S., Sicot, F.X., Gotta, S. & Chu, M.L. Mouse fibulin-2 gene. Complete exon-intron organization and promoter characterization. Eur. J. Biochem. 263, 471–477 (1999).
Takahara, K., Lee, S., Wood, S. & Greenspan, D.S. Structural organization and genetic localization of the human bone morphogenetic protein 1/mammalian tolloid gene. Genomics 29, 9–15 (1995).
Rossignol, M., Gagnon, M.L. & Klagsbrun, M. Genomic organization of human neuropilin-1 and neuropilin-2 genes: identification and distribution of splice variants and soluble isoforms. Genomics 70, 211–222 (2000).
Zimmermann, K., Hoischen, S., Hafner, M. & Nischt, R. Genomic sequences and structural organization of the human nidogen gene (NID). Genomics 27, 245–250 (1995).
Parma, J., Christophe, D., Pohl, V. & Vassart, G. Structural organization of the 5′ region of the thyroglobulin gene. Evidence for intron loss and “exonization” during evolution. J. Mol. Biol. 196, 769–779 (1987).
Linnenbach, A.J. et al. Retroposition in a family of carcinoma-associated antigen genes. Mol. Cell. Biol. 13, 1507–1515 (1993).
Sudhof, T.C., Goldstein, J.L., Brown, M.S. & Russell, D.W. The LDL receptor gene: a mosaic of exons shared with different proteins. Science 228, 815–822 (1985).
Marazziti, D., Eggertsen, G., Fey, G.H. & Stanley, K.K. Relationships between the gene and protein structure in human complement component C9. Biochemistry 27, 6529–6534 (1988).
Kirschbaum, N.E., Gumina, R.J. & Newman, P.J. Organization of the gene for human platelet/endothelial cell adhesion molecule-1 shows alternatively spliced isoforms and a functionally complex cytoplasmic domain. Blood 84, 4028–4037 (1994).
Coutelle, O. et al. The neural cell adhesion molecule L1: genomic organisation and differential splicing is conserved between man and the pufferfish Fugu. Gene 208, 7–15 (1998).
Nolan, K.F., Kaluz, S., Higgins, J.M., Goundis, D. & Reid, K.B. Characterization of the human properdin gene. Biochem. J. 287, 291–297 (1992).
Kiss, I. et al. Structure of the chicken link protein gene: exons correlate with the protein domains. Proc. Natl. Acad. Sci. USA 84, 6399–6403 (1987).
Screaton, G.R. et al. Genomic structure of DNA encoding the lymphocyte homing receptor CD44 reveals at least 12 alternatively spliced exons. Proc. Natl. Acad. Sci. USA 89, 12160–12164 (1992).
Naso, M.F., Zimmermann, D.R. & Iozzo, R.V. Characterization of the complete genomic structure of the human versican gene and functional analysis of its promoter. J. Biol. Chem. 269, 32999–33008 (1994).
Ord, D.C. et al. Structure of the gene encoding the human leukocyte adhesion molecule–1 (TQ1, Leu-8) of lymphocytes and neutrophils. J. Biol.Chem. 265, 7760–7767 (1990).
Collins, T. et al. Structure and chromosomal location of the gene for endothelial-leukocyte adhesion molecule 1. J. Biol. Chem. 266, 2466–2473 (1991).
Hoyle, G.W. & Hill, R.L. Structure of the gene for a carbohydrate-binding receptor unique to rat Kupffer cells. J. Biol. Chem. 266, 1850–1857 (1991).
Hahn, D., Illisson, R., Metspalu, A. & Sterchi, E.E. Human N-benzoyl-l-tyrosyl-p-aminobenzoic acid hydrolase (human meprin): genomic structure of the alpha and beta subunits. Biochem. J. 346, 83–91 (2000).
Kiss, I. et al. Structure of the gene for cartilage matrix protein, a modular protein of the extracellular matrix. Exon/intron organization, unusual splice sites, and relation to alpha chains of beta 2 integrins, von Willebrand factor, complement factors B and C2, and epidermal growth factor. J. Biol. Chem. 264, 8126–8134 (1989).
Hayman, A.R., Koppel, J. & Trueb, B. Complete structure of the chicken alpha 2(VI) collagen gene. Eur. J. Biochem. 197, 177–184 (1991).
Corbi A.L., Garcia–Aguilar, J. & Springer, T.A. Genomic structure of an integrin alpha subunit, the leukocyte p150,95 molecule. J. Biol. Chem. 265, 2782–2788 (1990).
Jenne, D. & Stanley, K.K. Nucleotide sequence and organization of the human S-protein gene: repeating peptide motifs in the "pexin" family and a model for their evolution. Biochemistry 26, 6735–6742 (1987).
Campbell, S.M., Rosen, J.M., Hennighausen, L.G., Strech-Jurk, U. & Sippel, A.E. Comparison of the whey acidic protein genes of the rat and mouse. Nucleic Acids Res. 12, 8685–8697 (1984).
Stetler, G., Brewer, M.T. & Thompson, R.C. Isolation and sequence of a human gene encoding a potent inhibitor of leukocyte proteases. Nucleic Acids Res. 14, 7883–7896 (1986).
Altruda, F., Poli, V., Restagno, G. & Silengo, L. Structure of the human hemopexin gene and evidence for intron-mediated evolution. J. Mol. Evol. 27, 102–108 (1988).
Huhtala, P., Chow, L.T. & Tryggvason, K. Structure of the human type IV collagenase gene. J. Biol. Chem. 265, 11077–11082 (1990).
Brauninger, A., Karn, T., Strebhardt, K. & Rubsamen-Waigmann, H. Characterization of the human CSK locus. Oncogene 8, 1365–1369 (1993).
Rohrer, J., Parolini, O., Belmont, J.W. & Conley, M.E. The genomic structure of human BTK, the defective gene in X-linked agammaglobulinemia. Immunogenetics 40, 319–324 (1994).
Tanaka, A. et al. DNA sequence encoding the amino-terminal region of the human c-src protein: implications of sequence divergence among src-type kinase oncogenes. Mol. Cell. Biol. 7, 1978–1983 (1987).
Anderson, S.K. et al. Human cellular src gene: nucleotide sequence and derived amino acid sequence of the region coding for the carboxy-terminal two-thirds of pp60c-src. Mol. Cell. Biol. 5, 1122–1129 (1985).
Airenne, T. et al. Structure of the human laminin gamma 2 chain gene (LAMC2): alternative splicing with different tissue distribution of two transcripts. Genomics 32, 54–64 (1996).
Kallunki, T., Ikonen, J., Chow, L.T., Kallunki, P. & Tryggvason, K. Structure of the human laminin B2 chain gene reveals extensive divergence from the laminin B1 chain gene. J. Biol. Chem. 266, 221–228 (1991).
Vuolteenaho, R., Chow, L.T. & Tryggvason, K. Structure of the human laminin B1 chain gene. J. Biol. Chem. 265, 15611–15616 (1990).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kolkman, J., Stemmer, W. Directed evolution of proteins by exon shuffling. Nat Biotechnol 19, 423–428 (2001). https://doi.org/10.1038/88084
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/88084
This article is cited by
-
Origin and Evolution of RAS Membrane Targeting
Oncogene (2023)
-
The abscisic acid receptor gene VvPYL4 positively regulates grapevine resistance to Plasmopara viticola
Plant Cell, Tissue and Organ Culture (PCTOC) (2020)
-
An insight into the evolution of introns in the gyrase A gene of plants
Plant Systematics and Evolution (2018)
-
Biomolecular engineering for nanobio/bionanotechnology
Nano Convergence (2017)
-
Public antibodies to malaria antigens generated by two LAIR1 insertion modalities
Nature (2017)