Abstract
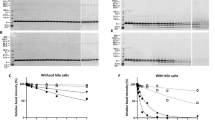
An integral part of the safety assessment of genetically modified plants is consideration of possible human health effects, especially food allergy. Prospective testing for allergenicity of proteins obtained from sources with no prior history of causing allergy has been difficult because of the absence of valid methods and models. Food allergens may share physicochemical properties that distinguish them from nonallergens, properties that may be used as a tool to predict the inherent allergenicity of proteins newly introduced into the food supply by genetic engineering. One candidate property is stability to digestion. We have systematically evaluated the stability of food allergens that are active via the gastrointestinal tract in a simple model of gastric digestion, emphasizing the major allergens of plant-derived foods such as legumes (peanuts and soybean). Important food allergens were stable to digestion in the gastric model (simulated gastric fluid). For example, soybean ß-conglycinin was stable for 60 min. In contrast, nonallergenic food proteins, such as spinach ribulose bis-phosphate carboxylase/oxygenase, were digested in simulated gastric fluid within 15 sec. The data are consistent with the hypothesis that food allergens must exhibit sufficient gastric stability to reach the intestinal mucosa where absorption and sensitization (development of atopy) can occur. Thus, the stability to digestion is a significant and valid parameter that distinguishes food allergens from nonallergens.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lavrik, P.B., Bartnicki, D.E., Feldman, J., Hammond, B.G., Keck, P.J., et al. 1995. Safety assessment of potatoes resistant to Colorado potato beetle, pp. 148–158 in ACS Symposium Series 605. Genetically modified foods. Safety issues. Engel, K-H., Takeoka, G.R., and Teranishi, R. (eds.). American Chemical Society, Washington, D.C.
Padgette, S.R., Re, D.B., Barry, G.F., Eichholtz, D.E., Delanny, X., et al. 1996. New weed control opportunities: Development of soybeans with a Roundup Ready™ gene, pp. 53–84 in Herbicide-resistant crops. Agricultural, environmental, economic, regulatory, and technical aspects. Duke, S.O. (ed.). CRC Lewis Publishers, Boca Baton, FL
US Food and Drug Administration, Department of Health and Human Services, 1992. Statement of policy: Foods derived from new plant varieties. Fed. Regist. 57: 22984–23005.
Nordlee, J.A., Taylor, S.L., Townsend, J.A., Thomas, L.A., and Bush, R.K. 1996. Identification of a Brazil-nut allergen in transgenic soybeans. N. Engl. J. Med. 334: 688–692.
Vieths, S., Aulepp, H., Becker, W-M., and Buschmann, L. 1996. Characterization of labile and stable allergens in foods of plant origin, pp. 130–149 in Food allergies and intolerances. Eisenbrand, G., Aulepp, H., Dayan, A.D., Bias, RS., Grunow, W., et al. (eds.). VCH Verlagsgesellschaft mbH, Weinheim.
Hefle, S. 1996. The chemistry and biology of food allergens. Food Tech. 50: 86–92.
Fuchs, R.L., Ream, J.E., Hammond, B.G., Naylor, M.W., Leimgruber, R.M., et al. 1993. Safety assessment of the neomycin phosphotransferase II (NPTII) protein. Bio/Technology 11: 1543–1547.
Reed, A.J., Kretzmer, K.A., Naylor, M.W., Finn, R.F., Magin, K.M., et al. 1996. Safety assessment of 1-aminocyclopropane-1-carboxylic acid deaminase protein expressed in delayed ripening tomatoes. J. Agric. Food Chem. 44: 388–394.
Sampson, H.A., Mendelson, L., and Rosen, J.P. 1992. Fatal and near-fatal analphylactic reactions to food in children and adolescents. N. Engl. J. Med. 327: 380–384.
Panconesi, E., Sertoli, A., Fabbri, P., Giorgini, S., and Spallanzi, P. 1980. Anaphylactic shock from mustard after ingestion of pizza. Contact Dermatitis 6: 294–295.
Shibasaki, M., Suzuki, S., Tajima, S., Nemoto, H., and Kuroume, T. 1980. Allergenicity of major component proteins of soybean. Int. Arch. Allergy Appl. Immunol. 61: 441–448.
Ogawa, T., Bando, N., Tsuji, H., Okajima, H., Nishikawa, K., et al. 1991. Investigation of the IgE-binding proteins in soybeans by immunoblotting with the sera of the soybean-sensitive patients with atopic dermatitis. J. Nutr. Sci. Vitaminol. 37: 555–565.
Ogawa, T., Tsuji, H., Bando, N., Kitamura, K., Zhu, Y-L., et al. 1993. Identification of the soybean allergenic protein, Gly m Bd 30K, with the soybean seed 34-kDa oil-body-associated protein. Biosci. Biotech. Biochem. 57: 1030–1033.
Burks, A.W., Cockrell, G., Connaughton, C., Guin, J., Alien, W., et al. 1994. Identification of peanut agglutinin and soybean trypsin inhibitor as minor legume allergens. Int. Arch. Allergy Immunol. 105: 143–149.
Burks, A.W., Williams, L.W., Helm, R.N., Connaughton, C., Cockrell, G., et al. 1991. Identification of a major peanut allergen, Ara h I, in patients with atopic dermatitis and positive peanut challenges. J. Allergy Clin. Immunol. 88: 172–179.
Burks, A.W., Williams, L.W., Connaughton, C., Cockrell, G., O'Brien, T.J., et al. 1992. Identification and characterization of a second major peanut allergen, Ara h II, with use of sera of patients with atopic dermatatis and positive peanut challenge. J. Allergy Clin. Immunol. 90: 962–969.
Menéndez-Arias, L., Moneo, I., Domínguez, J., and Rodríguez, R. 1988. Primary structure of the major allergen of yellow mustard (Sinapis alba L) seed, Sin a I. Eur. J. Biochem. 177: 159–166.
Gonz´lez de la Pena, M.A., Menéndez-Arias, L., Monsalve, R.I. and Rodríguez, R. 1991. Isolation and characterization of a major allergen from oriental mustard seeds, Bra j I. Int. Arch. Allergy Appl. Immunol. 96: 263–270.
Taylor, S.L. 1986. Immunologic and allergic properties of cow's milk proteins in humans. J. Food Protect. 49: 239–250.
Haddad, Z.H., Kalra, V., and Verma, S. 1979. IgE antibodies to peptic and peptic-tryptic digests of betalactogbbulin: significance in food hypersensitivity. Ann. Allergy 42: 368–371.
Schwartz, H.R., Nerurkar, L.S., Spies, J.R., Scanlon, R.T., and Bellanti, J.A. 1980. Milk hypersensitivity: RAST studies using new antigens generated by pepsin hydrolysis of beta-lactoglobulin. Ann. Allergy 45: 242–245.
Board of Trustees (ed.). 1995. Simulated Gastric Fluid, TS., pp. 2053 in The United States Pharmacopeia 23, The National Formulary 18. United States Pharmacopeial Convention, Inc., Rockville, MD.
Ryle, A.P. 1984. Pepsins, gastricsins and their zymogens, pp. 228–233 in Methods of Enzymatic Analysis. Bergmeyer, H.U. (ed.). Verlag Chemie, Deerfield Beach, FL.
Shägger, H. and Von Jagow, H. 1987. Tricine-sodium dodecyl sulfate-palyacryl-amide gel electrophoresis for separation of proteins in the range from 1 to 100 kDa. Anal. Biochem. 166: 368–379.
Charpentier, B.A. and Lemmel, D.E. 1984. A rapid automated procedure for the determination of trypsin inhibitor activity in soy products and common foodstuffs. J. Agric. Food Chem. 32: 908–911.
Liener, I.E. 1976. Legume toxins in relation to protein digestibility: A review. J. Food Sci. 41: 1076.
Deshpande, S.S. and Nielsen, S.S. 1987. In vitro enzymatic hydrolysis of phase-olin, the major storage protein of Phaseolus vilgaris L. J. Food Sci. 52: 1326–1329.
Asselin, J., Hébert, J., and Amiot, J. 1989. Effects of in vitro proteolysis on the allergenicity of major whey proteins. J. Food Sci. 54: 1037–1039.
Metcalfe, D.D., Fuchs, R.L., Astwood, J.D., Sampson, H.A., Taylor, S.L., et al. 1996. Assessment of the allergenic potential of foods derived from genetically engineered crop plants, in Allergenicity of foods produced by genetic modification. Fuchs, R.D., Fordham, J., Metcalfe, D., Sampson, H., Taylor, S., et al. (eds.). CRC Press, Boca Raton, FL. In press.
Fuchs, R.L., Rogan, G.J., Keck, P.J., Love, S.L., and Lavrik, P.B. 1995. Safety evaluation of Colorado potato beetle-protected potatoes, pp. 63–78 in Application of the principles of substantial equivalence to the safety evaluation of foods or food components from plants derived by modern biotechnology. World Health Organization, Food Safety Unit.
Astwood, J.D. and Fuchs, R.L. 1996. Preventing food allergy: emerging technologies. Trends Food Sci.Technol. 7: 219–226.
Tsuji, H., Bando, N., Kimoto, M., Okada, N., and Ogawa, T. 1993. Preparation and application of monoclonal antibodies for a sandwich enzyme-linked immunosor-bant assay of the major soybean allergen, Gly m Bd 30K. J. Nutr. Sci. Vitaminol. 39: 389–397.
Monsalve, R.I. Gonz´lez del la Pena, M.A., Menéndez-Arias, L., Lopez-Otin, C., Villalba, M., et al. 1993. Characterization of a new oriental-mustard (Brassica juncea) allergen, Bra j IE: detection of an allergenic epitope. Biochem. J. 293: 625–632.
Harlow, E. and Lane, D. 1988. Immunoblotting, pp. 1–726 in Antibodies: A laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY.
Christy, K.G., Jr., LaTart, D.B., and Osterhoudt, H.W. 1989. Modifications for SDS-PAGE of proteins. BioTechniques 7: 692–693.
Neuhoff, V., Arold, N., Taube, D., and Ehrhardt, W. 1988. Improved staining of proteins in polyacrylamide gels including isoelectric focusing gels with clear background at nanogram sensitivity using Coumassie brilliant blue G-250 and R-250. Electrophoresis 9: 255–262.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Astwood, J., Leach, J. & Fuchs, R. Stability of food allergens to digestion in vitro. Nat Biotechnol 14, 1269–1273 (1996). https://doi.org/10.1038/nbt1096-1269
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt1096-1269
This article is cited by
-
Re-evaluation of dietary interventions in rheumatoid arthritis: can we improve patient conversations around food choices?
Rheumatology International (2024)
-
Physicochemical Characterization and In Vitro Digestibility Study of an In Silico Designed Recombinant Protein Enriched with Large Neutral Amino Acids and Lacking Phenylalanine for Phenylketonuria
The Protein Journal (2022)
-
Are Physicochemical Properties Shaping the Allergenic Potency of Animal Allergens?
Clinical Reviews in Allergy & Immunology (2022)
-
Mass spectrometric analysis of digesta does not improve the allergenicity assessment of GM crops
Transgenic Research (2021)
-
Molecular characterization and safety assessment of biofortified provitamin A rice
Scientific Reports (2020)