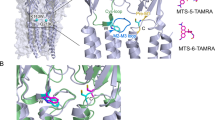
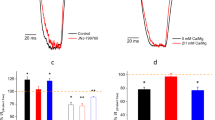
Abstract
Among ion channels, only the nicotinic-receptor superfamily has evolved to generate both cation- and anion-selective members. Although other, structurally unrelated, neurotransmitter-gated cation channels exist, no other type of neurotransmitter-gated anion channel, and thus no other source of fast synaptic inhibitory signals, has been described so far. In addition to the seemingly straightforward electrostatic effect of the presence (in the cation-selective members) or absence (in the anion-selective ones) of a ring of pore-facing carboxylates, mutational studies have identified other features of the amino-acid sequence near the intracellular end of the pore-lining transmembrane segments (M2) that are also required to achieve the high charge selectivity shown by native channels1,2,3,4,5,6,7,8,9,10. However, the mechanism underlying this more subtle effect has remained elusive11 and a subject of speculation. Here we show, using single-channel electrophysiological recordings to estimate the protonation state of native ionizable side chains, that anion-selective-type sequences favour whereas cation-selective-type sequences prevent the protonation of the conserved, buried basic residues at the intracellular entrance of the pore (the M2 0′ position). We conclude that the previously unrecognized tunable charge state of the 0′ ring of buried basic side chains is an essential feature of these channels’ versatile charge-selectivity filter.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
23 June 2011
Fig. 1a was corrected.
References
Galzi, J. L. et al. Mutations in the ion channel domain of a neuronal nicotinic receptor convert ion selectivity from cationic to anionic. Nature 359, 500–505 (1992)
Corringer, P.-J. et al. Mutational analysis of the charge selectivity filter of the α7 nicotinic acetylcholine receptor. Neuron 22, 831–843 (1999)
Keramidas, A., Moorhouse, A. J., French, C. R., Schofield, P. R. & Barry, P. H. M2 pore mutations convert the glycine receptor channel from being anion- to cation-selective. Biophys. J. 79, 247–259 (2000)
Gunthorpe, M. J. & Lummis, S. C. R. Conversion of the ion selectivity of the 5-HT3A receptor from cationic to anionic reveals a conserved feature of the ligand-gated ion channel superfamily. J. Biol. Chem. 276, 10977–10983 (2001)
Jensen, M. L. et al. The β subunit determines the ion selectivity of the GABAA receptor. J. Biol. Chem. 277, 41438–41447 (2002)
Keramidas, A., Moorhouse, A. J., Pierce, K. D., Schofield, P. R. & Barry, P. H. Cation-selective mutations in the M2 domain of the inhibitory glycine receptor channel reveal determinants of ion-charge selectivity. J. Gen. Physiol. 119, 393–410 (2002)
Thompson, A. J. & Lummis, S. C. R. A single ring of charged amino acids at one end of the pore can control ion selectivity in the 5-HT3 receptor. Br. J. Pharmacol. 140, 359–365 (2003)
Wotring, V. E., Miller, T. S. & Weiss, D. S. Mutations at the GABA receptor selectivity filter: a possible role for effective charges. J. Physiol. (Lond.) 548, 527–540 (2003)
Menard, C., Horvitz, H. R. & Cannon, S. Chimeric mutations in the M2 segment of the 5-hydroxytryptamine-gated chloride channel MOD-1 define a minimal determinant of anion/cation permeability. J. Biol. Chem. 280, 27502–27507 (2005)
Sunesen, M. et al. Mechanism of Cl− selection by a glutamate-gated chloride (GluCl) receptor revealed through mutations in the selectivity filter. J. Biol. Chem. 281, 14875–14881 (2006)
Keramidas, A., Moorhouse, A. J., Schofield, P. R. & Barry, P. H. Ligand-gated ion channels: mechanisms underlying ion selectivity. Prog. Biophys. Mol. Biol. 86, 161–204 (2004)
Cymes, G. D., Ni, Y. & Grosman, C. Probing ion-channel pores one proton at a time. Nature 438, 975–980 (2005)
Cymes, G. D. & Grosman, C. Pore-opening mechanism of the nicotinic acetylcholine receptor evinced by proton transfer. Nature Struct. Mol. Biol. 15, 389–396 (2008)
Krishek, B. J., Moss, S. J. & Smart, T. G. Homomeric β1 γ-aminobutyric acidA receptor-ion channels: evaluation of pharmacological and physiological properties. Mol. Pharmacol. 49, 494–504 (1996)
Cully, D. F. et al. Cloning of an avermectin-sensitive glutamate-gated chloride channel from Caenorhabditis elegans . Nature 371, 707–711 (1994)
van Nierop, P. et al. Identification of molluscan nicotinic acetylcholine receptor (nAChR) subunits involved in formation of cation- and anion-selective nAChRs. J. Neurosci. 25, 10617–10626 (2005)
Beg, A. A. & Jorgensen, E. M. EXP-1 is an excitatory GABA-gated cation channel. Nature Neurosci. 6, 1145–1152 (2003)
Bocquet, N. et al. A prokaryotic proton-gated ion channel from the nicotinic acetylcholine receptor family. Nature 445, 116–119 (2007)
Lee, D. J.-S., Keramidas, A., Moorhouse, A. J., Schofield, P. R. & Barry, P. H. The contribution of proline 250 (P-2′) to pore diameter and ion selectivity in the human glycine receptor channel. Neurosci. Lett. 351, 196–200 (2003)
Schutz, C. N. & Warshel, A. What are the dielectric ‘constants’ of proteins and how to validate electrostatic models? Proteins 44, 400–417 (2001)
Harms, M. J. et al. The pK a values of acidic and basic residues buried at the same internal location in a protein are governed by different factors. J. Mol. Biol. 389, 34–47 (2009)
Kamerlin, S. C. L., Haranczyk, M. & Warshel, A. Progress in ab initio QM/MM free-energy simulations of electrostatic energies in proteins: accelerated QM/MM studies of pK, redox reactions and solvation free energies. J. Phys. Chem. B 113, 1253–1272 (2009)
Karp, D. A., Stahley, M. R. & García-Moreno, E. B. Conformational consequences of ionization of Lys, Asp, and Glu buried at position 66 in staphylococcal nuclease. Biochemistry 49, 4138–4146 (2010)
Chimenti, M. S., Castañeda, C. A., Majumdar, A. & García-Moreno, E. B. Structural origins of high apparent dielectric constants experienced by ionizable groups in the hydrophobic core of a protein. J. Mol. Biol. 405, 361–377 (2011)
Papke, D., Gonzalez-Gutierrez, G. & Grosman, C. Desensitization of neurotransmitter-gated ion channels during high-frequency stimulation: a comparative study of Cys-loop, AMPA and purinergic receptors. J. Physiol. (Lond.) 589, 1571–1585 (2011)
Engel, A. G. et al. New mutations in acetylcholine receptor subunit genes reveal heterogeneity in the slow-channel congenital myasthenic syndrome. Hum. Mol. Genet. 5, 1217–1227 (1996)
Grosman, C. & Auerbach, A. Asymmetric and independent contribution of the second transmembrane segment 12′ residues to diliganded gating of acetylcholine receptor channels. A single-channel study with choline as the agonist. J. Gen. Physiol. 115, 637–651 (2000)
Ohno, K. et al. Congenital myasthenic syndrome caused by prolonged acetylcholine receptor channel openings due to a mutation in the M2 domain of the epsilon subunit. Proc. Natl Acad. Sci. USA 92, 758–762 (1995)
Qin, F. Restoration of single-channel currents using the segmental k-means method based on hidden Markov modeling. Biophys. J. 86, 1488–1501 (2004)
Qin, F., Auerbach, A. & Sachs, F. Estimating single-channel kinetic parameters from idealized patch-clamp data containing missed events. Biophys. J. 70, 264–280 (1996)
Elenes, S., Ni, Y., Cymes, G. D. & Grosman, C. Desensitization contributes to the synaptic response of gain-of-function mutants of the muscle nicotinic receptor. J. Gen. Physiol. 128, 615–627 (2006)
Barry, P. H. & Lynch, J. W. Liquid junction potentials and small cell effects in patch-clamp analysis. J. Membr. Biol. 121, 101–117 (1991)
Acknowledgements
We thank S. Sine for wild-type muscle AChR cDNA; M. Slaughter and D. Papke for wild-type α1 GlyR cDNA; S. Elenes for critical advice on fast-perfusion experiments; E. Jakobsson and H. Robertson for discussions; and G. Papke, M. Maybaum, J. Pizarek and C. Staehlin for technical assistance. This work was supported by a grant from the US National Institutes of Health (R01-NS042169 to C.G.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Text, Supplementary Figures 1-10 with legends, Supplementary Tables 1-4 and additional references. (PDF 2047 kb)
Rights and permissions
About this article
Cite this article
Cymes, G., Grosman, C. Tunable pKa values and the basis of opposite charge selectivities in nicotinic-type receptors. Nature 474, 526–530 (2011). https://doi.org/10.1038/nature10015
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10015
This article is cited by
-
Subunit gating resulting from individual protonation events in Kir2 channels
Nature Communications (2023)
-
Approaching the 5-HT3 receptor heterogeneity by computational studies of the transmembrane and intracellular domains
Journal of Computer-Aided Molecular Design (2013)
-
Emerging approaches to probing ion channel structure and function
Neuroscience Bulletin (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.