Abstract
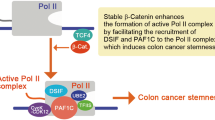
The E2F1 transcription factor can promote proliferation or apoptosis when activated, and is a key downstream target of the retinoblastoma tumour suppressor protein (pRB). Here we show that E2F1 is a potent and specific inhibitor of β-catenin/T-cell factor (TCF)-dependent transcription, and that this function contributes to E2F1-induced apoptosis. E2F1 deregulation suppresses β-catenin activity in an adenomatous polyposis coli (APC)/glycogen synthase kinase-3 (GSK3)-independent manner, reducing the expression of key β-catenin targets including c-MYC. This interaction explains why colorectal tumours, which depend on β-catenin transcription for their abnormal proliferation, keep RB1 intact. Remarkably, E2F1 activity is also repressed by cyclin-dependent kinase-8 (CDK8), a colorectal oncoprotein1. Elevated levels of CDK8 protect β-catenin/TCF-dependent transcription from inhibition by E2F1. Thus, by retaining RB1 and amplifying CDK8, colorectal tumour cells select conditions that collectively suppress E2F1 and enhance the activity of β-catenin.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Firestein, R. et al. CDK8 is a colorectal cancer oncogene that regulates β-catenin activity. Nature 10.1038/nature07179 (this issue)
Morris, E. J. et al. Functional identification of Api5 as a suppressor of E2F-dependent apoptosis in vivo . PLoS Genet 2, e196 (2006)
Freeman, M. & Bienz, M. EGF receptor/Rolled MAP kinase signalling protects cells against activated Armadillo in the Drosophila eye. EMBO Rep. 2, 157–162 (2001)
Clevers, H. Wnt/β-catenin signaling in development and disease. Cell 127, 469–480 (2006)
Kinzler, K. W. & Vogelstein, B. Lessons from hereditary colorectal cancer. Cell 87, 159–170 (1996)
Morin, P. J. et al. Activation of β-catenin-Tcf signaling in colon cancer by mutations in β-catenin or APC. Science 275, 1787–1790 (1997)
Hallstrom, T. C. & Nevins, J. R. Specificity in the activation and control of transcription factor E2F-dependent apoptosis. Proc. Natl Acad. Sci. USA 100, 10848–10853 (2003)
Black, E. P., Hallstrom, T., Dressman, H. K., West, M. & Nevins, J. R. Distinctions in the specificity of E2F function revealed by gene expression signatures. Proc. Natl Acad. Sci. USA 102, 15948–15953 (2005)
Sadot, E., Geiger, B., Oren, M. & Ben-Ze’ev, A. Down-regulation of β-catenin by activated p53. Mol. Cell. Biol. 21, 6768–6781 (2001)
Rother, K. et al. Identification of Tcf-4 as a transcriptional target of p53 signalling. Oncogene 23, 3376–3384 (2004)
Ueda, Y. et al. p73β, a variant of p73, enhances Wnt/β-catenin signaling in Saos-2 cells. Biochem. Biophys. Res. Commun. 283, 327–333 (2001)
Sansom, O. J. et al. Myc deletion rescues Apc deficiency in the small intestine. Nature 446, 676–679 (2007)
Liu, J. et al. Siah-1 mediates a novel β-catenin degradation pathway linking p53 to the adenomatous polyposis coli protein. Mol. Cell 7, 927–936 (2001)
Matsuzawa, S. I. & Reed, J. C. Siah-1, SIP, and Ebi collaborate in a novel pathway for β-catenin degradation linked to p53 responses. Mol. Cell 7, 915–926 (2001)
Hughes, T. A. & Brady, H. J. E2F1 up-regulates the expression of the tumour suppressor axin2 both by activation of transcription and by mRNA stabilisation. Biochem. Biophys. Res. Commun. 329, 1267–1274 (2005)
Hallstrom, T. C., Mori, S. & Nevins, J. R. An E2F1-dependent gene expression program that determines the balance between proliferation and cell death. Cancer Cell 13, 11–22 (2008)
Gope, R. et al. Increased expression of the retinoblastoma gene in human colorectal carcinomas relative to normal colonic mucosa. J. Natl Cancer Inst. 82, 310–314 (1990)
Kucherlapati, M. H., Nguyen, A. A., Bronson, R. T. & Kucherlapati, R. S. Inactivation of conditional Rb by Villin-Cre leads to aggressive tumors outside the gastrointestinal tract. Cancer Res. 66, 3576–3583 (2006)
Haigis, K., Sage, J., Glickman, J., Shafer, S. & Jacks, T. The related retinoblastoma (pRb) and p130 proteins cooperate to regulate homeostasis in the intestinal epithelium. J. Biol. Chem. 281, 638–647 (2006)
Williams, J. P. et al. The retinoblastoma protein is required for Ras-induced oncogenic transformation. Mol. Cell. Biol. 26, 1170–1182 (2006)
Dickins, R. A. et al. Probing tumor phenotypes using stable and regulated synthetic microRNA precursors. Nature Genet. 37, 1289–1295 (2005)
Faux, M. C. et al. Restoration of full-length adenomatous polyposis coli (APC) protein in a colon cancer cell line enhances cell adhesion. J. Cell Sci. 117, 427–439 (2004)
Kim, S., Xu, X., Hecht, A. & Boyer, T. G. Mediator is a transducer of Wnt/β-catenin signaling. J. Biol. Chem. 281, 14066–14075 (2006)
Malik, S. & Roeder, R. G. Dynamic regulation of pol II transcription by the mammalian Mediator complex. Trends Biochem. Sci. 30, 256–263 (2005)
He, L. et al. A microRNA polycistron as a potential human oncogene. Nature 435, 828–833 (2005)
O'Donnell, K. A., Wentzel, E. A., Zeller, K. I., Dang, C. V. & Mendell, J. T. c-Myc-regulated microRNAs modulate E2F1 expression. Nature 435, 839–843 (2005)
Lee, Y. S. & Carthew, R. W. Making a better RNAi vector for Drosophila: use of intron spacers. Methods 30, 322–329 (2003)
Dick, F. A., Sailhamer, E. & Dyson, N. J. Mutagenesis of the pRB pocket reveals that cell cycle arrest functions are separable from binding to viral oncoproteins. Mol. Cell. Biol. 20, 3715–3727 (2000)
Di Stefano, L., Jensen, M. R. & Helin, K. E2F7, a novel E2F featuring DP-independent repression of a subset of E2F-regulated genes. EMBO J. 22, 6289–6298 (2003)
Hurford, R. K., Cobrinik, D., Lee, M. H. & Dyson, N. pRB and p107/p130 are required for the regulated expression of different sets of E2F responsive genes. Genes Dev. 11, 1447–1463 (1997)
Acknowledgements
We thank many investigators for their gifts of cell lines, plasmids and fly stocks, especially S. Artavanis-Tsakonas. We thank D. Rennie and the Massachusetts General Hospital Cutaneous Biology Research Center Transgenic Fly Core for embryo injections, and B. Fowle for his help with SEM imaging. We thank A. McClatchey, J. Settleman, C. Seum and T. Orr-Weaver for their gifts of antibodies. We thank our colleagues at the Massachusetts General Hospital (MGH) Cancer Center for discussions. E.J.M. and J.-Y.J. are supported in part by a Ruth L. Kirschstein Award and a Tosteson Postdoctoral Fellowship, respectively. L.D.S. is supported by the MGH ECOR Fund for Medical Discovery. N.-S.M. is a Leukemia and Lymphoma Society Special Fellow. K.M.H. was supported by a Career Development award from the Harvard Gastrointestinal Specialized Program of Research Excellence (GI-SPORE) (P50-CA127003). N.J.D. was supported by a scholarship from the Saltonstall Foundation. This study was supported by grants from the National Institutes of Health to N.J.D. (GM81607, GM053203) and A.M.N. (GM071449).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-10 with Legends (PDF 5338 kb)
Rights and permissions
About this article
Cite this article
Morris, E., Ji, JY., Yang, F. et al. E2F1 represses β-catenin transcription and is antagonized by both pRB and CDK8. Nature 455, 552–556 (2008). https://doi.org/10.1038/nature07310
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07310
This article is cited by
-
Knockout of ICAT in Adipose Tissue Alleviates Fibro-inflammation in Obese Mice
Inflammation (2023)
-
MicroRNA-9 facilitates hypoxia-induced injury and apoptosis in H9c2 cells via targeting CDK8
Journal of Biosciences (2021)
-
Rebelled epigenome: histone H3S10 phosphorylation and H3S10 kinases in cancer biology and therapy
Clinical Epigenetics (2020)
-
Multiomics global landscape of stemness-related gene clusters in adipose-derived mesenchymal stem cells
Stem Cell Research & Therapy (2020)
-
Dissecting the Pol II transcription cycle and derailing cancer with CDK inhibitors
Nature Chemical Biology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.