Abstract
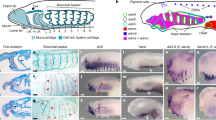
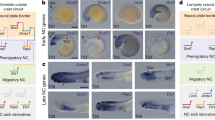
The neural crest, a defining character of vertebrates1, is of prime importance to their evolutionary origin2. To understand neural crest evolution, we explored molecular mechanisms underlying craniofacial development in the basal jawless vertebrate, sea lamprey (Petromyzon marinus), focusing on the SoxE (Sox8, Sox9 and Sox10) gene family. In jawed vertebrates, these are important transcriptional regulators of the neural crest3, and the loss of Sox9 causes abnormal craniofacial development4,5. Here we report that two lamprey SoxE genes are expressed in migrating neural crest and crest-derived prechondrocytes in posterior branchial arches, whereas a third paralogue is expressed later in the perichondrium and mandibular arch. Morpholino knock-down of SoxE1 reveals that it is essential for posterior branchial arch development, although the mandibular arch is unaffected. The results show that chondrogenic function of SoxE regulators can be traced to the lamprey–gnathostome common ancestor and indicate that lamprey SoxE genes might have undergone independent duplication to have distinct functions in mandibular versus caudal branchial arches. This work sheds light on the homology of vertebrate branchial arches and supports their common origin at the base of vertebrates.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Gans, C. & Northcutt, R. G. Neural crest and the origin of vertebrates: a new head. Science 220, 268–274 (1983)
Holland, N. D. & Chen, J.-Y. Origin and early evolution of the vertebrates: new insights from advances in molecular biology, anatomy, and palaeontology. BioEssays 23, 142–151 (2001)
Cheung, M. & Briscoe, J. Neural crest development is regulated by the transcription factor Sox9. Development 130, 5681–5693 (2003)
Spokony, R. F., Aoki, Y., Saint-Germain, N., Magner-Fink, E. & Saint-Jeannet, J. P. The transcription factor Sox9 is required for cranial neural crest development in Xenopus. Development 129, 421–432 (2002)
Yan, Y. L. et al. A zebrafish sox9 gene required for cartilage morphogenesis. Development 129, 5065–5079 (2002)
Horigome, N. et al. Development of cephalic neural crest cells in embryos of Lampetra japonica, with special reference to the evolution of the jaw. Dev. Biol. 207, 287–308 (1999)
Langille, R. M. & Hall, B. K. Role of the neural crest in development of the trabeculae and branchial arches in embryonic sea lamprey, Petromyzon marinus (L). Development 102, 301–310 (1988)
Neidert, A. H., Virupannavar, V., Hooker, G. W. & Langeland, J. A. Lamprey Dlx genes and early vertebrate evolution. Proc. Natl Acad. Sci. USA 98, 1665–1670 (2001)
McCauley, D. W. & Bronner-Fraser, M. Neural crest contributions to the lamprey head. Development 130, 2317–2327 (2003)
Janvier, P. Early Vertebrates (Oxford Univ. Press, Oxford, 1996)
Kimmel, C. B., Miller, C. T. & Keynes, R. J. Neural crest patterning and the evolution of the jaw. J. Anat. 199, 105–120 (2001)
Mallatt, J. Early vertebrate evolution—pharyngeal structure and the origin of gnathostomes. J. Zool. 204, 169–183 (1984)
Schultze, M. Die Entwickelungsgeschichte von Petromyzon planeri, (Haarlem, 1856)
Thompson, J. D., Higgins, D. G. & Gibson, T. J. Clustal-W—improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680 (1994)
Tomsa, J. M. & Langeland, J. A. Otx expression during lamprey embryogenesis provides insights into the evolution of the vertebrate head and jaw. Dev. Biol. 207, 26–37 (1999)
Kusakabe, R., Takechi, M., Tochinai, S. & Kuratani, S. Lamprey contractile protein genes mark different populations of skeletal muscles during development. J. Exp. Zool. B 302, 121–133 (2004)
McCauley, D. W. & Bronner-Fraser, M. Conservation of Pax gene expression in ectodermal placodes of the lamprey. Gene 287, 129–139 (2002)
Damas, H. Observations sur le développement des ganglions craniens chez Lampetra fluviatilis (L.). Arch. Biol. (Liege) 62, 65–95 (1951)
Takio, Y. et al. Evolutionary biology: lamprey Hox genes and the evolution of jaws. Nature Brief Communication arising, doi:10.1038/nature02616 (20 May 2004).
Coates, M. I. & Cohn, M. J. Fins, limbs and tails: outgrowths and axial patterning in vertebrate evolution. Bioessays 20, 371–381 (1998)
Cerny, R. et al. Developmental origins and evolution of jaws: new interpretation of ‘maxillary’ and ‘mandibular’. Dev. Biol. 276, 225–236 (2004)
Kuratani, S., Murakami, Y., Nobusada, Y., Kusakabe, R. & Hirano, S. Developmental fate of the mandibular mesoderm in the lamprey, Lethenteron japonicum: Comparative morphology and development of the gnathostome jaw with special reference to the nature of the trabecula cranii. J. Exp. Zoolog. B Mol. Dev. Evol. 302, 458–468 (2004)
Graham, A. The development and evolution of the pharyngeal arches. J. Anat. 199, 133–141 (2001)
Veitch, E., Begbie, J., Schilling, T. F., Smith, M. M. & Graham, A. Pharyngeal arch patterning in the absence of neural crest. Curr. Biol. 9, 1481–1484 (1999)
Acknowledgements
We thank J. Langeland for providing embryonic cDNA libraries; D. Meulemans for SoxE oligonucleotide primer sequences, T. Sauka-Spengler for SoxE2 and SoxE3 library screens, R. Bergstedt and the staff at Hammond Bay Biological Station for facilities and technical assistance; R. Kusakabe for providing the LjMA2 DNA construct, and L. Trinh for assistance with confocal microscopy imaging techniques. This work was supported by a grant to M.B.F. from the National Aeronautics and Space Administration.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
PmSoxE1, PmSoxE2, PmSoxE3 and PmSoxF sequences are deposited in GenBank under accession numbers AY830453, DQ328983, DQ328984 and AY830454, respectively. Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods, Supplementary Figures 1–4 and Supplementary Discussion. (PDF 361 kb)
Supplementary Movie 1
Quicktime Movie through a dorsal-to-ventral Z-series of confocal images illustrating SoxE1 RNA expression in a stage 25 embryo at different dorsoventral levels within developing branchial arches. Presumptive cartilage cells, shown in red, express SoxE1 in branchial arches. Rostrally (righthand side) pharyngeal endodermal outpockets contact ectoderm to form branchial arches and expression of SoxE1 in the rostralmost arch is in differentiating chondrocytes.. In the caudal region shown (lefthand side), endoderm does not contact ectoderm and SoxE1 expression is present in undifferentiated ectomesenchymal neural crest. Dorsally (start of movie), the caudal pharyngeal outpocketing nearly contacts the ectoderm to form a branchial arch. Ventrally (end of movie), the endodermal outpocket is shown at a greater distance from ectoderm than its dorsal position at the start of the movie. Lamprey autofluorescence shown in green. (MOV 1417 kb)
Supplementary Movie 2
Quicktime movie through a dorsal-to-ventral Z-series of confocal images illustrating SoxE2 RNA expression in a stage 25 embryo at different dorsoventral levels within branchial arches. SoxE2 RNA expression is present only in stacks of prechondrocytes within each arch. (MOV 420 kb)
Supplementary Movie 3
Quicktime movie through a dorsal-to-ventral Z-series of confocal images illustrating SoxE3 expression in a stage 25 embryo at different dorsoventral levels within a developing branchial arch. SoxE3 RNA is expressed in the perichondrial cells surrounding the chondrocytes, but is not present in the stacked chondrocytes within the branchial arch shown. (MOV 1945 kb)
Rights and permissions
About this article
Cite this article
McCauley, D., Bronner-Fraser, M. Importance of SoxE in neural crest development and the evolution of the pharynx. Nature 441, 750–752 (2006). https://doi.org/10.1038/nature04691
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04691
This article is cited by
-
Cartilage diversification and modularity drove the evolution of the ancestral vertebrate head skeleton
EvoDevo (2023)
-
Evolution of the endothelin pathway drove neural crest cell diversification
Nature (2020)
-
Evolution of the new head by gradual acquisition of neural crest regulatory circuits
Nature (2019)
-
Embryonic expression patterns and phylogenetic analysis of panarthropod sox genes: insight into nervous system development, segmentation and gonadogenesis
BMC Evolutionary Biology (2018)
-
Highly conserved molecular pathways, including Wnt signaling, promote functional recovery from spinal cord injury in lampreys
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.