Abstract
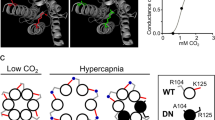
Extracellular signalling by the purine nucleotide ATP has long been associated with sensory function1,2,3,4,5,6,7,8. In the periphery, ATP mediates nociception3,4,5, mechanosensitivity3,6, thermal sensitivity7 and O2 chemosensitivity8. These processes share a common mechanism that involves the release of ATP to excite afferent fibres via activation of ionotropic P2X and/or metabotropic P2Y receptors. Chemosensors located in the brainstem are crucial for the maintenance of physiological levels of blood gases through the regulation of breathing9,10,11. Here we show that an increase in pCO2 in the arterial blood triggers the immediate release of ATP from three chemosensitive regions located on the ventral surface of the medulla oblongata. Blockade of ATP receptors at these sites diminishes the chemosensory control of breathing, suggesting that ATP release constitutes a key step in central chemosensory transduction. These new data suggest that ATP, a phylogenetically ancient, unique and simple molecule, has been widely used in the evolution of afferent systems to mediate distinct forms of sensory transduction not only in the periphery but also within the central nervous system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chen, C. C. et al. A P2X purinoceptor expressed by a subset of sensory neurons. Nature 377, 428–431 (1995)
Lewis, C. et al. Coexpression of P2X2 and P2X3 receptor subunits can account for ATP-gated currents in sensory neurons. Nature 377, 432–435 (1995)
Cook, S. P., Vulchanova, L., Hargreaves, K. M., Elde, R. & McCleskey, E. W. Distinct ATP receptors on pain-sensing and stretch-sensing neurons. Nature 387, 505–508 (1997)
Cockayne, D. A. et al. Urinary bladder hyporeflexia and reduced pain-related behaviour in P2X3-deficient mice. Nature 407, 1011–1015 (2000)
Cook, S. P. & McCleskey, E. W. Cell damage excites nociceptors through release of cytosolic ATP. Pain 95, 41–47 (2002)
Vlaskovska, M. et al. P2X3 knock-out mice reveal a major sensory role for urothelially released ATP. J. Neurosci. 21, 5670–5677 (2001)
Souslova, V. et al. Warm-coding deficits and aberrant inflammatory pain in mice lacking P2X3 receptors. Nature 407, 1015–1017 (2000)
Rong, W. et al. Pivotal role of nucleotide P2X2 receptor subunit of the ATP-gated ion channel mediating ventilatory responses to hypoxia. J. Neurosci. 23, 11315–11321 (2003)
Loeschcke, H. H. Central chemosensitivity and the reaction theory. J. Physiol. (Lond.) 32, 1–24 (1982)
Feldman, J. L., Mitchell, G. S. & Nattie, E. E. Breathing: rhythmicity, plasticity, chemosensitivity. Annu. Rev. Neurosci. 26, 239–266 (2003)
Mulkey, D. K. et al. Respiratory control by ventral surface chemoreceptor neurons in rats. Nature Neurosci. 7, 1360–1369 (2004)
Llaudet, E., Botting, N. P., Crayston, J. A. & Dale, N. A three-enzyme microelectrode sensor for detecting purine release from central nervous system. Biosens. Bioelectron. 18, 43–52 (2003)
Llaudet, E., Hatz, S., Droniou, M. & Dale, N. Microelectrode biosensor for real-time measurement of ATP in biological tissue. Anal. Chem. 77, 3267–3273 (2005)
Richter, D. W. & Spyer, K. M. Studying rhythmogenesis of breathing: comparison of in vivo and in vitro models. Trends Neurosci. 24, 464–472 (2001)
Kawai, A., Ballantyne, D., Muckenhoff, K. & Scheid, P. Chemosensitive medullary neurones in the brainstem-spinal cord preparation of the neonatal rat. J. Physiol. (Lond.) 492, 277–292 (1996)
Gourine, A. V., Atkinson, L., Deuchars, J. & Spyer, K. M. Purinergic signalling in the medullary mechanisms of respiratory control in the rat: respiratory neurones express the P2X2 receptor subunit. J. Physiol. (Lond.) 552, 197–211 (2003)
Acknowledgements
We thank M. E. Droniou for help with the initial development of the ATP sensor. We also thank the Biotechnology and Biological Sciences Research Council (K.M.S., A.V.G.) and the Wellcome Trust (N.D.) for support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
N.D. and E.L. are founders of, and hold equity in, Sarissa Biomedical Ltd. Sarissa Biomedical Ltd. offers for sale ATP biosensors similar to some of those used in this article.
Supplementary information
Rights and permissions
About this article
Cite this article
Gourine, A., Llaudet, E., Dale, N. et al. ATP is a mediator of chemosensory transduction in the central nervous system. Nature 436, 108–111 (2005). https://doi.org/10.1038/nature03690
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03690
This article is cited by
-
Kir4.1 channels contribute to astrocyte CO2/H+-sensitivity and the drive to breathe
Communications Biology (2024)
-
Carbon dioxide and MAPK signalling: towards therapy for inflammation
Cell Communication and Signaling (2023)
-
Modulatory dynamics of periodic and aperiodic activity in respiration-brain coupling
Nature Communications (2023)
-
Purinergic signaling mediates neuroglial interactions to modulate sighs
Nature Communications (2023)
-
A Controlled Transcription-Driven Light-Up Aptamer Amplification for Nucleoside Triphosphate Detection
BioChip Journal (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.