Abstract
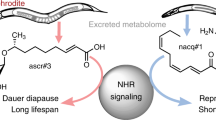
Pheromones are cell type-specific signals used for communication between individuals of the same species. When faced with overcrowding or starvation, Caenorhabditis elegans secrete the pheromone daumone, which facilitates communication between individuals for adaptation to adverse environmental stimuli1,2,3,4. Daumone signals C. elegans to enter the dauer stage, an enduring and non-ageing stage of the nematode life cycle with distinctive adaptive features and extended life. Because daumone is a key regulator of chemosensory processes in development and ageing5,6, the chemical identification of daumone is important for elucidating features of the daumone-mediated signalling pathway. Here we report the isolation of natural daumone from C. elegans by large-scale purification, as well as the total chemical synthesis of daumone. We present the stereospecific chemical structure of purified daumone, a fatty acid derivative. We demonstrate that both natural and chemically synthesized daumones equally induce dauer larva formation in C. elegans (N2 strain) and certain dauer mutants, and also result in competition between food and daumone. These results should help to elucidate the daumone-mediated signalling pathway, which might in turn influence ageing and obesity research and the development of antinematodal drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Cassada, R. C. & Russell, R. L. The dauer larva, a post-embryonic developmental variant of the nematode Caenorhabditis elegans . Dev. Biol. 46, 326–342 (1975)
Golden, J. W. & Riddle, D. L. A pheromone influences larval development in the nematode Caenorhabditis elegans . Science 218, 578–580 (1982)
Golden, J. W. & Riddle, D. L. The Caenorhabditis elegans dauer larva: developmental effects of pheromone, food, and temperature. Dev. Biol. 102, 368–378 (1984)
Golden, J. W. & Riddle, D. L. A Caenorhabditis elegans dauer-inducing pheromone and an antagonistic component of the food supply. J. Chem. Ecol. 10, 1265–1280 (1984)
Bargmann, C. I. & Horvitz, H. R. Control of larval development by chemosensory neurons in Caenorhabditis elegans . Science 251, 1243–1246 (1991)
Schackwitz, W. S., Inoue, T. & Thomas, J. H. Chemosensory neurons function in parallel to mediate a pheromone response in C. elegans . Neuron 17, 719–728 (1996)
Thomas, J. H., Birnby, D. A. & Vowels, J. J. Evidence for parallel processing of sensory information controlling dauer formation in Caenorhabditis elegans . Genetics 134, 1105–1117 (1993)
Patterson, G. I., Koweek, A., Wong, A., Liu, Y. & Ruvkun, G. The DAF-3 Smad protein antagonizes TGF-beta-related receptor signaling in the Caenorhabditis elegans dauer pathway. Genes Dev. 11, 2679–2690 (1997)
Apfeld, J. & Kenyon, C. Regulation of lifespan by sensory perception in Caenorhabditis elegans . Nature 402, 804–809 (1999)
Perkins, L. A., Hedgecock, E. M., Thomson, J. N. & Culotii, J. G. Mutant sensory cilia in the nematode Caenorhabditis elegans . Dev. Biol. 117, 456–487 (1986)
Antebi, A., Culotti, J. G. & Hedgecock, E. M. daf-12 regulates developmental age and the dauer alternative in Caenorhabditis elegans . Development 125, 1191–1205 (1998)
Bird, D. M. & Opperman, C. H. Caenorhabditis elegans: A genetic guide to parasitic nematode biology. J. Nematol. 30, 299–308 (1998)
Viney, M. F. & Franks, N. R. Is dauer pheromone of Caenorhabditis elegans really a pheromone? Naturwissenschaften 91, 123–124 (2004)
Regnier, F. E. & Law, J. H. Insect pheromone. J. Lipid Res. 9, 541–551 (1968)
Koga, M., Take-uchi, M., Tameishi, T. & Ohshima, Y. Control of DAF-7 TGF-(alpha) expression and neuronal process development by a receptor tyrosine kinase KIN-8 in Caenorhabditis elegans . Development 126, 5387–5398 (1999)
Brenner, S. The genetics of Caenorhabditis elegans . Genetics 77, 71–94 (1974)
Choi, B. K., Chitwood, D. J. & Paik, Y.-K. Proteomic changes during disturbance of cholesterol metabolism by azacoprostane treatment in Caenorhabditis elegans . Mol. Cell. Proteomics 2, 1086–1095 (2003)
Vowels, J. J. & Thomas, J. H. Multiple chemosensory defects in daf-11 and daf-21 mutants of Caenorhabditis elegans . Genetics 138, 303–316 (1994)
Kimura, K. D., Tissenbaum, H. A., Liu, Y. & Ruvkun, G. daf-2, an insulin receptor-like gene that regulates longevity and diapause in Caenorhabditis elegans . Science 277, 942–946 (1997)
Rance, M. et al. Improved spectral resolution in cosy 1H NMR spectra of proteins via double quantum filtering. Biochem. Biophys. Res. Commun. 117, 479–485 (1983)
Bax, A. & Subramanian, S. Sensitivity-enhanced two-dimensional heteronuclear shift correlation NMR spectroscopy. J. Magn. Reson. 67, 565–570 (1986)
Bax, A. & Davis, D. G. Practical aspects of two-dimensional transverse NOE spectroscopy. J. Magn. Reson. 63, 207–213 (1985)
Bax, A., Griffey, R. H. & Hawkins, B. L. Correlation of proton and nitrogen-15 chemical shifts by multiple quantum NMR. J. Magn. Reson. 55, 301–315 (1983)
Bax, A. & Summers, M. F. 1H and 13C assignments from sensitivity enhanced detection of heteronuclear multiple-bond connectivity by two-dimensional multiple quantum NMR. J. Am. Chem. Soc. 108, 2093–2094 (1986)
Schleucher, J. et al. A general enhancement scheme in heteronuclear multidimensional NMR employing pulsed field gradients. J. Biomol. NMR 4, 301–306 (1994)
Acknowledgements
This study was supported by a grant to Y.K.P. from the Korea Health 21 R&D Project, Ministry of Health and Welfare, Republic of Korea. We thank J.-M. Kim at KDR Biotech Co. for his support on this project, D.J. Chitwood at the USDA-ARS Nematology Lab for his critical reading and suggestions, R. Moyer at King College (USA) for editorial assistance, J. Lee at Seoul National University for discussions and the Caenorhabditis Genetics Center for kind provision of the C. elegans strains used in this study. Technical support from the LG Chem Research Center (Taejon, Korea) was appreciated.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Methods
Description of methods used for the chemical synthesis of daumone. (DOC 226 kb)
Supplementary Table 1
Chemical shift of daumone by NMR analysis. (JPG 156 kb)
Supplementary Table 2
Differential dauer formation by food and daumone, which exhibits a conditional competition. (JPG 240 kb)
Supplementary Table 3
HRMS, R f and elemental analysis data. (JPG 210 kb)
Supplementary Table 4
Comparison of spectral data. (JPG 135 kb)
Supplementary Figure 1
A representative chromatogram of columns, molecular structure and fragmentation patterns of daumone. (JPG 116 kb)
Supplementary Figure 2
DEPT spectrum of the daumone. (JPG 65 kb)
Supplementary Figure 3
2D 1H, 13C HMBC spectrum of the daumone. (JPG 68 kb)
Supplementary Figure 4
2D TOCSY spectrum of the daumone. (JPG 63 kb)
Supplementary Figure 5
The observed ROEs of the daumone. (JPG 72 kb)
Supplementary Figure 6
500MHz 1H NMR spectrum of compound 1 (synthetic) at 298 K. (JPG 67 kb)
Supplementary Figure 7
500MHz 1H NMR spectrum of compound 1 (natural) at 298 K. (JPG 46 kb)
Supplementary Figure 8
125.7MHz 13C NMR spectrum of compound 1 at 298 K. (JPG 63 kb)
Supplementary Figure 9
500MHz DEPT spectrum of compound 1 at 298 K. (JPG 74 kb)
Supplementary Figure 10
500MHz HMBC spectrum of compound 1 at 298 K. (JPG 55 kb)
Supplementary Figure 11
500MHz ROESY spectrum of compound 1 at 298 K (JPG 47 kb)
Supplementary Figure 12
500MHz NOESY spectrum of compound 1 at 298 K. (JPG 81 kb)
Supplementary Figure 13
FT-IR spectrum of compound 1 at 298 K. (JPG 124 kb)
Supplementary Figure 14
HRMS(FAB) spectrum of compound 1 at 298 K. (JPG 85 kb)
Supplementary Figure 15
High resolution Q-TOF mass spectrum of compound 1. (JPG 34 kb)
Rights and permissions
About this article
Cite this article
Jeong, PY., Jung, M., Yim, YH. et al. Chemical structure and biological activity of the Caenorhabditis elegans dauer-inducing pheromone. Nature 433, 541–545 (2005). https://doi.org/10.1038/nature03201
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03201
This article is cited by
-
Pathogenic bacteria modulate pheromone response to promote mating
Nature (2023)
-
On the role of dauer in the adaptation of nematodes to a parasitic lifestyle
Parasites & Vectors (2021)
-
Endocrine pheromones couple fat rationing to dauer diapause through HNF4α nuclear receptors
Science China Life Sciences (2021)
-
miR-31-5p regulates cold acclimation of the wood-boring beetle Monochamus alternatus via ascaroside signaling
BMC Biology (2020)
-
Plant metabolism of nematode pheromones mediates plant-nematode interactions
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.