Abstract
Understanding how nerves regenerate is an important step towards developing treatments for human neurological disease1, but investigation has so far been limited to complex organisms (mouse and zebrafish2) in the absence of precision techniques for severing axons (axotomy). Here we use femtosecond laser surgery for axotomy in the roundworm Caenorhabditis elegans and show that these axons functionally regenerate after the operation. Application of this precise surgical technique should enable nerve regeneration to be studied in vivo in its most evolutionarily simple form.
Similar content being viewed by others
Main
Femtosecond laser pulses provide high peak intensities that reduce the energy threshold for tissue removal (ablation)3 and enable laser surgery to be carried out with a low-energy source4,5. (For methods, see supplementary information.) We successfully cut single axons inside C. elegans by using pulse energies of 10–40 nanojoules at the specimen and tightly focused, 200-femtosecond, near-infrared laser pulses. This results in the vaporization of axon volumes of about 0.1–0.3 femtolitres, assuming an average axon diameter of 0.3 micrometres (see supplementary information). Dye-filling of axotomized neurons confirmed that the observed axon gaps are not due to photobleaching, but to physical disconnection of the axons (see supplementary information).
The minimum energy used is consistent with measured optical breakdown thresholds in transparent materials3,6. At these low energies, we would expect mechanical effects due to plasma expansion and shock waves to be significantly reduced5,6 with respect to other laser-surgery techniques that require much higher energies (for example, 0.4 microjoules with 0.5-nanosecond pulses7). The use of pulses at a low repetition rate (1 kilo-hertz, 10 microwatts average power) should reduce heat accumulation and extended thermal damage to the environment. We were able to cut individual processes within a few micrometres of each other without damaging the nearby processes (see supplementary information).
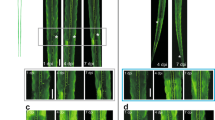
The D-type motor neurons in L4 larval-stage worms were selected as targets for laser surgery. These neurons have ventral cell bodies and extend circumferential axons towards the dorsal side; they form synapses to body muscles8. We cut the circumferential axons (labelled with green fluorescent protein9) at their mid-body positions, leaving the rest of the axon intact (Fig. 1a). Both ends of the severed axons initially retracted. Among 52 operated axons (in 11 worms), 54% regrew towards their distal ends within 12–24 hours (Fig. 1b). Axons that showed partial, aberrant, or no regrowth within 24 hours did not show further improvement over longer observation times (up to 36 hours).
a, Fluorescence images of axons labelled with green fluorescent protein before, immediately after, and in the hours following axotomy. Arrow indicates point of severance. Scale bar, 5 µm. b, Statistics of axon growth 24 h after axotomy, based on fluorescence images (n=52 axons). c, Time-course analysis of backward motion of worms following axotomy. Seventeen worms were scored blindly at different time points (for criteria, see supplementary information). Improvement in backward motion was graded as four levels from ‘shrinker’ behaviour (dark red) up to ‘wild type’ behaviour (yellow) in the hours following axotomy.
To evaluate functional recovery associated with nerve regeneration, we tested the behaviour of operated worms as it related to motor-neuron function. Loss of D-neuron function results in simultaneous contraction of dorsal and ventral body muscles (‘shrinker’ phenotype)10, which prevents backward locomotion. Operated worms (17 in total) showed this expected ‘shrinker’ phenotype immediately after axotomy (15 axons per worm), whereas sham-operated animals (6 in total) moved like wild-type worms.
Remarkably, the locomotion of operated worms improved, approaching that of the wild type within 24 hours of surgery (Fig. 1c), indicating that the regenerated axons were functional (see movie in supplementary information). By contrast, the shrinker phenotype caused by laser ablation of D-neuron cell bodies did not recover after 48 hours (results not shown). The correlation of axonal regrowth with behavioural recovery in C. elegans indicates that these nerves must have regenerated.
Femtosecond laser axotomy is a new technique that can be performed with 100% efficiency, sub-micrometre precision and high speed. As simple organisms such as C. elegans have amenable genetics, application of the femtosecond laser axotomy technique we describe here should help in the rapid identification of genes and molecules that affect nerve regeneration and development.
References
Horner, P. J. & Gage, F. H. Nature 407, 963–970 (2000).
Bhatt, D. H., Otto, S. J., Depoister, B. & Fetcho, J. R. Science 305, 254–258 (2004).
Perry, M. D. et al. J. Appl. Phys. 85, 6803–6810 (1999).
Tirlapur, U. K. & König, K. Nature 418, 290–291 (2002).
Shen, N. Thesis, Harvard Univ. (2003).
Vogel, A. et al. Appl. Phys. B 68, 271–280 (1999).
Colombelli, J., Grill, S. W. & Stelzer, E. H. K. Rev. Sci. Instrum. 75, 472–478 (2004).
White, J. G., Southgate, E., Thomson, J. N. & Brenner, S. Phil. Trans. R. Soc. Lond. B 314, 1–340 (1986).
Huang, X., Cheng, H. J., Tessier-Lavigne, M. & Jin, Y. Neuron 34, 563–576 (2002).
McIntire, S. L., Jorgensen, E., Kaplan, J. & Horvitz, H. R. Nature 364, 337–341 (1993).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Yanik, M., Cinar, H., Cinar, H. et al. Functional regeneration after laser axotomy. Nature 432, 822 (2004). https://doi.org/10.1038/432822a
Published:
Issue Date:
DOI: https://doi.org/10.1038/432822a
This article is cited by
-
Delivering broadband light deep inside diffusive media
Nature Photonics (2024)
-
Molecular mechanisms of neurite regeneration and repair: insights from C. elegans and Drosophila
Cell Regeneration (2023)
-
Nanotechnology development in surgical applications: recent trends and developments
European Journal of Medical Research (2023)
-
Emerging roles of mitochondria in animal regeneration
Cell Regeneration (2023)
-
Femtosecond laser microdissection for isolation of regenerating C. elegans neurons for single-cell RNA sequencing
Nature Methods (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.