Abstract
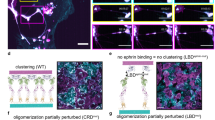
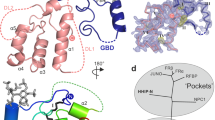
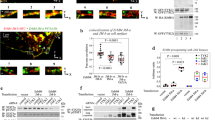
The Eph family of receptor tyrosine kinases and their membrane-anchored ephrin ligands are important in regulating cell–cell interactions as they initiate a unique bidirectional signal transduction cascade whereby information is communicated into both the Eph-expressing and the ephrin-expressing cells. Initially identified as regulators of axon pathfinding and neuronal cell migration, Ephs and ephrins are now known to have roles in many other cell–cell interactions, including those of vascular endothelial cells and specialized epithelia1,2. Here we report the crystal structure of the complex formed between EphB2 and ephrin-B2, determined at 2.7 Å resolution. Each Eph receptor binds an ephrin ligand through an expansive dimerization interface dominated by the insertion of an extended ephrin loop into a channel at the surface of the receptor. Two Eph–Ephrin dimers then join to form a tetramer, in which each ligand interacts with two receptors and each receptor interacts with two ligands. The Eph and ephrin molecules are precisely positioned and orientated in these complexes, promoting higher-order clustering and the initiation of bidirectional signalling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Flanagan, J. G. & Vanderhaeghen, P. The ephrins and Eph receptors in neural development. Annu. Rev. Neurosci. 21, 309–345 (1998).
Frisen, J., Holmberg, J. & Barbacid, M. Ephrins and their Eph receptors: multitalented directors of embryonic development. EMBO J. 18, 5159–5165 (1999).
Henkemeyer, M. et al. Nuk controls pathfinding of commissural axons in the mammalian central nervous system. Cell 86, 35–46 (1996).
Holland, S. J. et al. Bidirectional signalling through the EPH-family receptor Nuk and its transmembrane ligands. Nature 383, 722–725 (1996).
Bruckner, K., Pasquale, E. B. & Klein, R. Tyrosine phosphorylation of transmembrane ligands for Eph receptors. Science 275, 1640–1643 (1997).
Cowan, C. A. & Henkemeyer, M. The SH2/SH3 domain adaptor Grb4 transduces B-ephrin reverse signals. Nature 413, 174–179 (2001).
Eph Nomenclature Committee. Unified nomenclature for Eph family receptors and their ligands, the Ephrins. Cell 90, 403–404 (1997).
Gale, N. W. et al. Eph receptors and ligands comprise two major specificity subclasses and are reciprocally compartmentalized during embryogenesis. Neuron 17, 9–19 (1996).
Henkemeyer, M. et al. Immunolocalization of the Nuk receptor tyrosine kinase suggests roles in segmental patterning of the brain and axonogenesis. Oncogene 9, 1001–1014 (1994).
Davis, S. et al. Ligands for EPH-related receptor tyrosine kinases that require membrane attachment or clustering for activity. Science 266, 816–819 (1994).
Himanen, J. P., Henkemeyer, M. & Nikolov, D. B. Crystal structure of the ligand-binding domain of the receptor tyrosine kinase EphB2. Nature 396, 486–491 (1998).
Toth, J. et al. Crystal structure of an ephrin ectodomain. Dev. Cell 1, 83–92 (2001).
Labrador, J. P., Brambilla, R. & Klein, R. The N-terminal globular domain of Eph receptors is sufficient for ligand binding and receptor signaling. EMBO J. 16, 3889–3897 (1997).
Lackmann, M. et al. Distinct subdomains of the EphA3 receptor mediate ligand binding and receptor dimerization. J. Biol. Chem. 273, 20228–20237 (1998).
Hendrickson, W. A. Determination of macromolecular structures from anomalous diffraction of synchrotron radiation. Science 254, 51–58 (1991).
Lackmann, M. et al. Ligand for EPH-related kinase (LERK) 7 is the preferred high affinity ligand for the HEK receptor. J. Biol. Chem. 272, 16521–16530 (1997).
Stein, E. et al. Eph receptors discriminate specific ligand oligomers to determine alternative signaling complexes, attachment, and assembly responses. Genes Dev. 12, 667–678 (1998).
Bruckner, K. et al. EphrinB ligands recruit GRIP family PDZ adaptor proteins into raft membrane microdomains. Neuron 22, 511–524 (1999).
Stapleton, D., Balan, I., Pawson, T. & Sicheri, F. The crystal structure of an Eph receptor SAM domain reveals a mechanism for modular dimerization. Nature Struct. Biol. 6, 44–49 (1999).
Thanos, C. D., Goodwill, K. E. & Bowie, J. U. Oligomeric structure of the human EphB2 receptor SAM domain. Science 283, 833–836 (1999).
Hock, B. et al. PDZ-domain-mediated interaction of the Eph-related receptor tyrosine kinase EphB3 and the ras-binding protein AF6 depends on the kinase activity of the receptor. Proc. Natl Acad. Sci. USA 95, 9779–9784 (1998).
Torres, R. et al. PDZ proteins bind, cluster, and synaptically colocalize with Eph receptors and their ephrin ligands. Neuron 21, 1453–1463 (1998).
Buchert, M. et al. The junction-associated protein AF-6 interacts and clusters with specific Eph receptor tyrosine kinases at specialized sites of cell- cell contact in the brain. J. Cell Biol. 144, 361–371 (1999).
Lin, D., Gish, G. D., Songyang, Z. & Pawson, T. The carboxyl terminus of B class ephrins constitutes a PDZ domain binding motif. J. Biol. Chem. 274, 3726–3733 (1999).
Cowan, C. A., Yokoyama, N., Bianchi, L. M., Henkemeyer, M. & Fritzsch, B. EphB2 guides axons at the midline and is necessary for normal vestibular function. Neuron 26, 417–430 (2000).
Otwinowski, Z. & Minor, W. Data Collection and Processing 556-562 (SERC Daresbury Laboratory, Warrington, UK, 1993).
Project, C. C. The CCP4 suite: programs for X-ray crystallography. Acta Crystallogr. D 50, 760–763 (1994).
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard . Improved methods for binding protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Brünger, A. T. X-PLOR v. 3.1 Manual (Yale Univ. Press, New Haven, 1993).
Acknowledgements
We thank J. Coyle for help with figure preparation, and P. Jeffrey for help with X-ray measurements. D.B.N is a PEW fellow. This work was supported by the NIH and the New York Council Speaker's Fund for Biomedical Research (D.B.N.) and by the Welch Foundation (M.H.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Himanen, JP., Rajashankar, K., Lackmann, M. et al. Crystal structure of an Eph receptor–ephrin complex. Nature 414, 933–938 (2001). https://doi.org/10.1038/414933a
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/414933a
This article is cited by
-
Targeting EphA2: a promising strategy to overcome chemoresistance and drug resistance in cancer
Journal of Molecular Medicine (2024)
-
Oncogenic functions and therapeutic targeting of EphA2 in cancer
Oncogene (2021)
-
Human ribonuclease 1 serves as a secretory ligand of ephrin A4 receptor and induces breast tumor initiation
Nature Communications (2021)
-
EphrinB2 overexpression enhances osteogenic differentiation of dental pulp stem cells partially through ephrinB2-mediated reverse signaling
Stem Cell Research & Therapy (2020)
-
Targeting EphA2 in cancer
Journal of Hematology & Oncology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.