Abstract
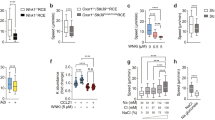
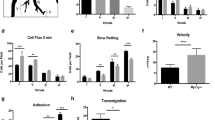
Cell migration is a fundamental biological process involving membrane polarization and cytoskeletal dynamics1, both of which are regulated by Rho family GTPases2,3,4,5. Among these molecules, Rac is crucial for generating the actin-rich lamellipodial protrusion, a principal part of the driving force for movement3,6. The CDM family proteins, Caenorhabditis elegans CED-5, human DOCK180 and Drosophila melanogaster Myoblast City (MBC), are implicated to mediate membrane extension by functioning upstream of Rac7,8,9,10,11,12. Although genetic analysis has shown that CED-5 and Myoblast City are crucial for migration of particular types of cells8,9,12, physiological relevance of the CDM family proteins in mammals remains unknown. Here we show that DOCK2, a haematopoietic cell-specific CDM family protein13, is indispensable for lymphocyte chemotaxis. DOCK2-deficient mice (DOCK2-/-) exhibited migration defects of T and B lymphocytes, but not of monocytes, in response to chemokines, resulting in several abnormalities including T lymphocytopenia, atrophy of lymphoid follicles and loss of marginal-zone B cells. In DOCK2-/- lymphocytes, chemokine-induced Rac activation and actin polymerization were almost totally abolished. Thus, in lymphocyte migration DOCK2 functions as a central regulator that mediates cytoskeletal reorganization through Rac activation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lauffenburger, D. A. & Horwitz, A. F. Cell migration: a physically integrated molecular process. Cell 84, 359–369 (1996).
Stowers, L., Yelon, D., Berg, L. J. & Chant, J. Regulation of the polarization of T cells toward antigen presenting cells by Ras-related GTPase Cdc42. Proc. Natl Acad. Sci. USA 92, 5027–5031 (1995).
Hall, A. Rho GTPases and the actin cytoskeleton. Science 279, 509–514 (1998).
Allen, W. E., Zicha, D., Ridley, A. J. & Jones, G. E. A role for Cdc42 in macrophage chemotaxis. J. Cell Biol. 141, 1147–1157 (1998).
Servant, G. et al. Polarization of chemoattractant receptor signaling durning neutrophil chemotaxis. Science 287, 1037–1040 (2000).
Bar-Sagi, D. & Hall, A. Ras and Rho GTPases: a new family reunion. Cell 103, 227–238 (2000).
Hasegawa, H. et al. DOCK180, a major CRK-binding protein, alters cell morphology upon translocation to the cell membrane. Mol. Cell. Biol. 16, 1770–1776 (1996).
Erickson, M. R. S., Galletta, B. J. & Abmayr, S. M. Drosophila myobast city encodes a conserved protein that is essential for myoblast fusion, dorsal closure, and cytoskeletal organization. J. Cell Biol. 138, 589–603 (1997).
Wu, Y.-C. & Horvitz, H. R. C. elegans phagocytosis and cell-migration protein CED-5 is similar to human DOCK180. Nature 392, 501–504 (1998).
Kiyokawa, E. et al. Activation of Rac1 by a Crk SH3-binding protein, DOCK180. Genes Dev. 12, 3331–3336 (1998).
Nolan, K. M. et al. Myobast city, the Drosophila homolog of DOCK180/CED-5, is required in a Rac signaling pathyway utilized for multiple developmental processes. Genes Dev. 12, 3337–3342 (1998).
Reddien, P. W. & Horvitz, H. R. CED-2/CrkII and CED-10/Rac control phagocytosis and cell migration in Caenorhabditis elegans. Nature Cell Biol. 2, 131–136 (2000).
Nishihara, H. et al. Non-adherent cell-specific expression of DOCK2, a member of the human CDM-family proteins. Biochem. Biophys. Acta 1452, 179–187 (1999).
Nagase, T. et al. Prediction of the coding sequences of unidentified human genes. VI. The coding sequences of 80 new genes (KIAA0201-KIAA0280) deduced by analysis of cDNA clones from cell line KG-1 and brain. DNA Res. 3, 321–329.
Manser, E., Leung, T., Salihuddin, H., Zhao, Z.-s. & Lim, L. A brain serine/threonine protein kinase activated by Cdc42 and Rac1. Nature 367, 40–46 (1994).
Bleul, C. C. et al. The lymphocyte chemoattractant SDF-1 is a ligand for LESTR/fusin and blocks HIV-1 entry. Nature 382, 829–833 (1996).
Oberlin, E. et al. The CXC chemokine SDF-1 is the ligand for LETR/fusin and prevents infection by T-cell-line-adapted HIV-1. Nature 382, 833–835 (1996).
Yoshida, R. et al. Secondary lymphoid-tissue chemokine is a functional ligand for the CC chemokine receptor CCR7. J. Biol. Chem. 273, 7118–7122 (1998).
Sotsios, Y. & Ward, S. G. Phosphoinositide 3-kinase: a key biochemical signal for cell migration in response to chemokines. Immunol. Rev. 177, 217–235 (2000).
Thelen, M. Dancing to the tune of chemokines. Nature Immunol. 2, 129–134 (2001).
Förster, R. et al. A putative chemokine receptor, BLR1, directs B cell migration to defined lymphoid organs and specific anatomic compartments of the spleen. Cell 87, 1037–1047 (1996).
Nakano, H. et al. A novel mutant gene involved in T-lymphocyte-specific homing into peripheral lymphoid organs on mouse chromosome 4. Blood 91, 2886–2895 (1998).
Förster, R. et al. CCR7 coordinates the primary immune response by establishing functional microenvironments in secondary lymphoid organs. Cell 99, 23–33 (1999).
Gunn, M. D. et al. Mice lacking expression of secondary lymphoid organ chemokine have defects in lymphocyte homing and dendritic cell localization. J. Exp. Med. 189, 451–460 (1999).
Oliver, A. M., Martin, F., Gartland, G. L., Carter, R. H. & Kearney, J. F. Marginal zone B cells exhibit unique activation, proliferative and immunoglobulin secretory responses. Eur. J. Immunol. 27, 2366–2374 (1997).
Kim, C. H., Pelus, L. M., White, J. R. & Broxmeyer, H. E. Differential chemotactic behavior of developing T cells in response to thymic chemokines. Blood 91, 4434–4443 (1998).
Sasaki, T. et al. Function of PI3Kγ in thymocyte development, T cell activation, and neutrophil migration. Science 287, 1040–1046 (2000).
Li, Z. et al. Roles of PLC-β2 and -β3 and PI3Kγ in chemoattractant-mediated signal transduction. Science 287, 1046–1049 (2000).
Hirsch, E. et al. Central role for G protein-coupled phosphoinositide 3-kinase γ in inflammation. Science 287, 1049–1053 (2000).
Onai, N. et al. Impairment of lymphopoiesis and myelopoiesis in mice reconstituted with bone marrow-hematopoietic progenitor cells expressing SDF-1-intrakine. Blood 96, 2074–2080 (2000).
Acknowledgements
We thank K. Matsushima and N. Onai for anti-CXCR4 and anti-CCR7 antibodies; K.-i. Nakayama and K. Nakayama for E14 ES cells and helpful suggestions on ES cell culture; P. Marrack for BEα16-3; and Y. Yamada for blastocyst injection. This work was supported in part by a grant from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fukui, Y., Hashimoto, O., Sanui, T. et al. Haematopoietic cell-specific CDM family protein DOCK2 is essential for lymphocyte migration. Nature 412, 826–831 (2001). https://doi.org/10.1038/35090591
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35090591
This article is cited by
-
Multiple Immune Defects in Two Patients with Novel DOCK2 Mutations Result in Recurrent Multiple Infection Including Live Attenuated Virus Vaccine
Journal of Clinical Immunology (2023)
-
Extra-hematopoietic immunomodulatory role of the guanine-exchange factor DOCK2
Communications Biology (2022)
-
DOCK2 regulates antifungal immunity by regulating RAC GTPase activity
Cellular & Molecular Immunology (2022)
-
The DOCK protein family in vascular development and disease
Angiogenesis (2021)
-
The RUNX1-ETO target gene RASSF2 suppresses t(8;21) AML development and regulates Rac GTPase signaling
Blood Cancer Journal (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.