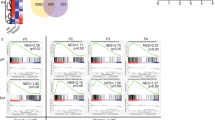
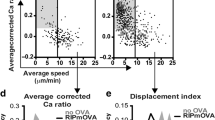
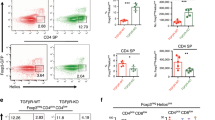
Abstract
Positive selection allows thymocytes that recognize an individual's own major histocompatibility complex (self-MHC) molecules to survive and differentiate, whereas negative selection removes overtly self-reactive thymocytes1. Although both forms of thymic selection are mediated by the αβ T-cell receptor (TCR) and require self-MHC recognition, an important question is whether they are controlled by distinct signalling cascades2. We have shown that mutation of an essential motif within the TCR α-chain-connecting peptide domain (α-CPM) profoundly affects positive but not negative selection3. Using transgenic mice expressing a mutant α-CPM TCR we examined the contribution of several mitogen-activated protein kinase (MAPK) cascades to thymic selection. Here we show that in thymocytes expressing a mutant α-CPM receptor, a positively selecting peptide failed to activate the extracellular signal-regulated kinase (ERK), although other MAPK cascades were induced normally. The defect in ERK activation was associated with impaired recruitment of the activated tyrosine kinases Lck and ZAP-70, phosphorylated forms of the TCR component CD3ζ and the adaptor protein LAT to detergent-insoluble glycolipid-enriched microdomains (DIGs). Therefore, an intact DIG-associated signalosome is essential for sustained ERK activation, which leads to positive selection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
von Boehmer, H., Teh, H. S. & Kisielow, P. The thymus selects the useful, neglects the useless and destroys the harmful. Immunol. Today 10, 57–61 (1989).
Mariathasan, S., Jones, R. G. & Ohashi, P. S. Signals involved in thymocyte positive and negative selection. Semin. Immunol. 11, 263– 272 (1999).
Bäckström, B. T., Müller, U., Hausmann, B. & Palmer, E. Positive selection through a motif in the αβ T cell receptor. Science 281, 835–838 ( 1998).
Hogquist, K. A. et al. T cell receptor antagonist peptides induce positive selection. Cell 76, 17–27 (1994).
Zijlstra, M. et al. β 2-microglobulin deficient mice lack CD4-8+cytolytic T cells. Nature 344, 742–746 (1990).
Hogquist, K. A., Jameson, S. C. & Bevan, M. J. Strong agonist ligands for the T cell receptor do not mediate positive selection of functional CD8+ T cells. Immunity 3, 79–86 (1995).
Swat, W., Ignatowicz, L., von Boehmer, H. & Kisielow, P. Clonal deletion of immature CD4+8+thymocytes in suspension culture by extrathymic antigen-presenting cells. Nature 351, 150–153 ( 1991).
Alberola-Ila, J., Forbush, K. A., Seger, R., Krebs, E. G. & Perlmutter, R. M. Selective requirement for MAP kinase activation in thymocyte differentiation. Nature 373, 620–623 (1995).
Alberola-Ila, J., Hogquist, K. A., Swan, K. A., Bevan, M. J. & Perlmutter, R. M. Positive and negative selection invoke distinct signaling pathways. J. Exp. Med. 184 , 9–18 (1996).
Swan, K. A. et al. Involvement of p21ras distinguishes positive and negative selection in thymocytes. EMBO J. 14, 276 –285 (1995).
O'Shea, C. C., Crompton, T., Rosewell, I. R., Hayday, A. C. & Owen, M. J. Raf regulates positive selection. Eur. J. Immunol. 26, 2350– 2355 (1996).
Pagès, G. et al. Defective thymocyte maturation in p44 MAP kinase (Erk 1) knockout mice. Science 286, 1374– 1377 (1999).
Sugawara, T., Moriguchi, T., Nishida, E. & Takahama, Y. Differential roles of ERK and p38 MAP kinase pathways in positive and negative selection of T lymphocytes. Immunity 9, 565–574 (1998).
Yang, D. D. et al. Differentiation of CD4+ T cells to Th1 cells requires MAP kinase JNK2. Immunity 9, 575 –585 (1998).
Sabapathy, K. et al. JNK2 is required for efficient T-cell activation and apoptosis but not for normal lymphocyte development. Curr. Biol. 9, 116–125 (1999).
Dong, C. et al. Defective T cell differentiation in the absence of Jnk1. Science 282, 2092–2095 ( 1998).
Jacinto, E., Werlen, G. & Karin, M. Cooperation between Syk and Rac1 leads to synergistic JNK activation in T lymphocytes. Immunity 8, 31–41 (1998).
Werlen, G., Jacinto, E., Xia, Y. & Karin, M. Calcineurin preferentially synergizes with PKC-θ to activate JNK and IL-2 promoter in T lymphocytes. EMBO J. 17, 3101–3111 (1998).
Dong, C. et al. JNK is required for effector T-cell function but not for T-cell activation. Nature 405, 91– 94 (2000).
Dave, V. P. et al. CD3δ deficiency arrests development of the αβ but not the γδ T cell lineage. EMBO J. 16, 1360–1370 (1997).
Simons, K. & Ikonen, E. Functional rafts in cell membranes. Nature 387, 569–572 (1997).
Montixi, C. et al. Engagement of T cell receptor triggers its recruitment to low-density detergent-insoluble membrane domains. EMBO J. 17, 5334–5348 (1998).
Zhang, W., Trible, R. P. & Samelson, L. E. LAT palmitoylation: its essential role in membrane microdomain targeting and tyrosine phosphorylation during T cell activation. Immunity 9, 239–246 (1998).
Finco, T. S., Kadlecek, T., Zhang, W., Samelson, L. E. & Weiss, A. LAT is required for TCR-mediated activation of PLCγ and the Ras pathway. Immunity 9, 617– 626 (1998).
Marshall, C. J. Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell 80, 179–185 (1995).
York, R. D. et al. Rap1 mediates sustained MAP kinase activation induced by nerve growth factor. Nature 392, 622– 626 (1998).
Xia, Z., Dickens, M., Raingeaud, J., Davis, R. J. & Greenberg, M. E. Opposing effects of ERK and JNK-p38 MAP kinases on apoptosis. Science 270, 1326–1331 (1995).
Bäckström, B. T. et al. A motif within the T cell receptor α chain constant region connecting peptide domain controls antigen responsiveness. Immunity 5, 437–447 ( 1996).
Stotz, S. H., Bolliger, L., Carbone, F. R. & Palmer, E. T cell receptor (TCR) antagonism without a negative signal: evidence from T cell hybridomas expressing two independent TCRs. J. Exp. Med. 189, 253–264 ( 1999).
Alexander, J., Payne, J. A., Murray, R., Frelinger, J. A. & Cresswell, P. Differential transport requirements of HLA and H-2 class I glycoproteins. Immunogenetics 29, 380– 388 (1989).
Acknowledgements
We thank F. Carbone, M. J. Bevan and D. Kioussis for transgenic mice expressing wild-type OT-1 receptors; A. Peter for generating mutant α-CPM TCR constructs; U. Müller for generation of mutant transgenic mice; T. Potter for T2-Kb cells; M. Daniels, K. Hogquist and S. Jameson for tetramers; L. Samelson for LAT antisera; S. Stotz and T. Harder for discussions; and E. Jacinto, T. Baldari, P. Kisielow and J. Kirberg for reading the manuscript. The Basel Institute for Immunology was founded and is supported by F. Hoffmann-LaRoche Ltd.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Werlen, G., Hausmann, B. & Palmer, E. A motif in the αβ T-cell receptor controls positive selection by modulating ERK activity. Nature 406, 422–426 (2000). https://doi.org/10.1038/35019094
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35019094
This article is cited by
-
Dietary glucosamine overcomes the defects in αβ-T cell ontogeny caused by the loss of de novo hexosamine biosynthesis
Nature Communications (2022)
-
Structural understanding of T cell receptor triggering
Cellular & Molecular Immunology (2020)
-
Insertion of exogenous epitopes in the E3-19K of oncolytic adenoviruses to enhance TAP-independent presentation and immunogenicity
Gene Therapy (2015)
-
Antigen and cytokine receptor signals guide the development of the naïve mature B cell repertoire
Immunologic Research (2013)
-
How the TCR balances sensitivity and specificity for the recognition of self and pathogens
Nature Immunology (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.