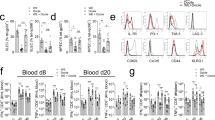
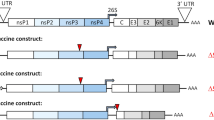
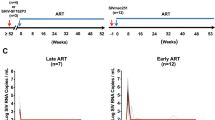
Abstract
Infection of adult mice with lymphocytic choriomeningitis virus (LCMV), a non-cytopathic segmented RNA virus, leads initially to generalized infection, followed by clearance and subsequent lifelong immunity. Indirect evidence has suggested that viral antigens may persist in lymphoid tissues during the phase of immunological memory, but viral genomic RNA has not been detected in previous studies1,2. During a search for persistent virus in the spleen, we identified LCMV-specific sequences present as DNA by polymerase chain reaction (PCR) in mice over 200 days after infection. In vivo and in vitro studies revealed that reverse transcription of viral RNA into complementary DNA occurred after acute infection of cells of its natural hosts, mouse and hamster, but not of other species and could be inhibited in vitro by azidothymidine (AZT), indicating that this was mediated by endogenous reverse transcriptase activity. These findings reveal a surprising and new pathway of interaction between exogenous RNA viruses and endogenous retroviral, and perhaps other host components, that results in the persistence of virally determined DNA. We speculate that the latter may function in vivo as a form of DNA vaccine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lau, L. L., Jamieson, B. D., Somasundaram, T. & Ahmed, R. Cytotoxic T-cell memory without antigen. Nature 369, 648–652 (1994).
Slifka, M., Matloubian, M. & Ahmed, R. Bone Marrow is a major site of long-term antibody production after acute viral infection. J. Virol. 69, 1895–1902 (1995).
Zinkernagel, R. M. et al. On immunological memory. A. Rev. Immunol. 14, 333–368 (1996).
Kundig, T. et al. On the role of antigen in maintaining CTL memory. Proc. Natl Acad. Sci. USA 93, 9716–8723 (1996).
Ahmed, R. & Gray, D. Immunological memory and protective immunity: understanding their relation. Science 272, 54–60 (1996).
Battegay, M. et al. Quantification of LCMV with immunological focus assay in 24 or 96 well plates. J. Virol. Methods 33, 191–198 (1991).
Pircher, H. et al. Viral escape by selection of cytotoxic T cell-resistant variants in vivo. Nature 346, 629–233 (1990).
Kagi, D. et al. Cytotoxicity mediated by T cells and natural killer cells is greatly impaired in perforin-deficient mice. Nature 369, 31–37 (1994).
Smith, M., Brian, E. L. & Pagano, J. Resumption of virus production after HIV infection of T lymphocytes in the presence of AZT. J. Virol. 61, 3769–3773 (1987).
Rawls, W., Banerjee, S., McMillan, C. & Buchmeier, M. Inhibition of Pichinde virus replication by Actinomycin D. J. Gen. Virol. 33, 421–434 (1976).
Planz, O., Seiler, P., Hengartner, H. & Zinkernagel, R. Specific CTL eliminate cells producing neutralizing antibodies. Nature 382, 726–730 (1996).
Lehmann-Grube, F. Lymphocytic choriomeningitis virus. Virol. Monogr. 10, 28–106 (1971).
Pyra, H., Boeni, J. & Schupbach, J. Ultrasensitive retrovirus detection by a RT assay based on product enhancement. Proc. Natl Acad. Sci. USA 51, 1544–1548 (1994).
Oxenius, A. et al. Presentation of endogenous viral proteins in association with MHC Class II. Eur. J. Immunol. 25, 3402–3411 (1995).
Oldstone, M. & Buchmeier, M. Restricted expression of viral glycoprotein in cells of persistently infected mice. Nature 300, 360–362 (1982).
Lehmann-Grube, F. Acarrier state of LCMV in L cell cultures. Nature 213, 770–773 (1967).
Moskophidis, D. & Zinkernagel, R. M. Immunobiology of cytotoxic T-cell escape mutants of lymphocytic choriomeningitis virus. J. Virol. 69, 2187–2193 (1995).
Salvato, M. in The Arenaviridae(ed. Salvato, M.) 133–156 (Plenum, New York, (1993)).
Heidmann, O. & Heidmann, T. Retrotransposition of a mouse IAP sequence tagged with an indicator gene. Cell 64, 159–170 (1991).
Zhdanov, V. Integration of viral genomes. Nature 256, 471–473 (1975).
Wiener, A., Deininger, P. & Efstratiadis, A. Nonretroviral transposons: Genes, pseudogenes and transposable elements generated by reverse flow of genetic information. A. Rev. Biochem. 55, 631–661 (1986).
Coffin, J. in Reverse Transcriptase(eds Skalka, A. & Gough, S.) 445–479 (Cold Spring Harbour Laboratory Press, New York, (1993)).
Oldstone, M. & Dixon, F. Activation of murine leukaemia virus related antigen by LCMV. Science 174, 843–845 (1971).
Pease, L. & Murphy, W. Co-infection by LDV and C type retrovirus elicits neurological disease. Nature 286, 398–400 (1980).
Planz, O. et al. Acritical role for neutralizing-antibody-producing B cells, CD4+ T cells, and interferons in persistent and acute infections of mice with LCMV: implications for adoptive immunotherapy of virus carriers. Proc. Natl Acad. Sci. USA 94, 6874–6879 (1997).
Loewer, R., Loewer, J. & Kurth, R. The viruses in all of us: Characteristics and biological significance of human endogenous retrovirus sequences. Proc. Natl Acad. Sci. USA 93, 5177–5184 (1996).
Yokoyama, M., Zhang, J. & Whitton, J. DNA immunization confers protection against lethal LCMV. J. Virol. 69, 2684–2688 (1995).
Skyulev, Y., Joo, M., Vturina, I., Tsomides, T. & Eisen, H. Evidence that a single peptide-MHC complex on a target cell can elicit a CTL response. Immunity 4, 565–571 (1996).
Romanowski, V., Matsuura, Y. & Bishop, D. Complete sequence of the S RNA of LCMV (WE) compared to that of Pichinde. Virus Res. 3, 101–108 (1985).
Silver, J., Maudru, T., Fujita, K. & Repaske, R. An RT-PCR assay for the enzyme activity of reverse transcriptase capable of detecting single virions. Nucleic Acids Res. 21, 3593–3594 (1993).
Acknowledgements
This work was supported by the Wellcome Trust, the Swiss National Foundation for Science and the Kanton of Zurich, Switzerland. We thank E. Horvath, H. Krettli, A. Oxenius, S. Oehen, K.Riem, O. Planz, P. Seiler, N. Wey, D. Zimmerman and his group and J. Schupbach and J. Boeni of the Swiss National Centre for Retroviruses, University of Zurich for their technical help; we also thank L.Stitz (BFAV, Tübingen) and J. Klein (MPI, Tübingen) for providing mice and D.Bishop, J. Goudsmit, M.Billeter and R. Cattaneo for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klenerman, P., Hengartner, H. & Zinkernagel, R. A non-retroviral RNA virus persists in DNA form. Nature 390, 298–301 (1997). https://doi.org/10.1038/36876
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/36876
This article is cited by
-
ERASE: a novel nucleic-acid based antiviral mechanism
Cell Research (2021)
-
Endogenous reverse transcriptase and RNase H-mediated antiviral mechanism in embryonic stem cells
Cell Research (2021)
-
Enteric Virome and Carcinogenesis in the Gut
Digestive Diseases and Sciences (2020)
-
A diverse virome in kidney transplant patients contains multiple viral subtypes with distinct polymorphisms
Scientific Reports (2016)
-
Characterisation of cytoplasmic DNA complementary to non-retroviral RNA viruses in human cells
Scientific Reports (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.