Abstract
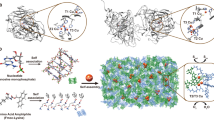
EACH individual excretes roughly 10 kg of urea per year, as a result of the hydrolysis of arginine in the final cytosolic step of the urea cycle1. This reaction allows the disposal of nitrogenous waste from protein catabolism, and is catalysed by the liver arginase enzyme2. In other tissues that lack a complete urea cycle, arginase regulates cellular arginine and ornithine concentrations for biosynthetic reactions3, including nitric oxide synthesis: in the macrophage, arginase activity is reciprocally coordinated with that of NO synthase to modulate NO-dependent cytotoxicity4–9. The bioinorganic chemistry of arginase is particularly rich because this enzyme is one of very few that specifically requires a spin-coupled Mn2+ –Mn2+ cluster for catalytic activity in vitro and in vivo10. The 2.1 Å-resolution crystal structure of trimeric11 rat liver arginase reveals that this unique metal cluster resides at the bottom of an active-site cleft that is 15 Å deep. Analysis of the structure indicates that arginine hydrolysis is achieved by a metal-activated solvent molecule which symmetrically bridges the two Mn2+ ions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Krebs, H. A. & Henseleit, K. Hoppe-Seyler's Z. Physiol. Chem. 210, 33–66 (1932).
Herzfeld, A. & Raper, S. M. Biochem. J. 153, 469–478 (1976).
Yip, M. C. M. & Knox, W. E. Biochem. J. 127, 893–899 (1972).
Corraliza, I. M., Soler, G., Eichmann, K. & Modolell, M. Biochem. Biophys. Res. Commun. 206, 667–673 (1995).
Daghigh, F., Fukuto, J. M. & Ash, D. E. Biochem. Biophys. Res. Commun. 202, 174–180 (1994).
Chénais, B., Yapo, A., Lepoivre, M. & Tenu, J.-P. Biochem. Biophys. Res. Commun. 196, 1558–1565 (1993).
Klatt, P., Schmidt, K., Uray, G. & Mayer, B. J. Biol. Chem. 268, 14781–14787 (1993).
Keller, R., Gehri, R., Keist, R., Huf, E. & Kayser, F. H. Cell. Immunol. 134, 249–256 (1991).
Albina, J. E., Henry, W. L., Mastrofrancesco, B., Martin, B.-A. & Reichner, J. S. J. Immunol. 155, 4391–4396 (1995).
Reczkowski, R. S. & Ash, D. E. J. Am. Chem. Soc. 114, 10992–10994 (1992).
Kanyo, Z. F., Chen, C.-Y., Daghigh, F., Ash, D. E. & Christianson, D. W. J. Mol. Biol. 224, 1175–1177 (1992).
Christianson, D. W. & Alexander, R. S. J. Am. Chem. Soc. 111, 6412–6419 (1989).
Rardin, R. L., Tolman, W. B. & Lippard, S. J. New J. Chem. 15, 417–430 (1991).
Cavalli, R. C., Burke, C. J., Kawamoto, S., Soprano, D. R. & Ash, D. E. Biochemistry 33, 10652–10657 (1994).
Christianson, D. W. & Fierke, C. A. Acc. Chem. Res. 29, 331–339 (1996).
Reczkowski, R. S. & Ash, D. E. Arch. Biochem. Biophys. 312, 31–37 (1994).
Kuhn, N. J., Talbot, J. & Ward, S. Arch. Biochem. Biophys. 286, 217–221 (1991).
Jabri, E., Carr, M. B., Hausinger, R. P. & Karplus, P. A. Science 268, 998–1004 (1995).
Park, I.-S. & Hausinger, R. P. Biochemistry 35, 5345–5352 (1996).
Nyborg, J. & Wonacott, A. J. in The Rotation Method in Crystallography (eds Arndt, U. W. & Wonacott, A. J.) 139–152 (North-Holland, Amsterdam, 1977).
Collaborative Computational Project No. 4 Acta Crystallogr. D 50, 760–763 (1994).
Furey, W. & Swaminathan, S. Am. Crystallogr. Assoc. Mtg Prog. Abstr. 18, 73 (1990).
Brünger, A. T., Kuriyan, J. & Karplus, M. Science 235, 458–460 (1987).
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Acta. Crystallogr. A 47, 110–119 (1991).
Kraulis, P. J. Appl. Crystallogr. 24, 946–950 (1991).
Bacon, D. J. & Anderson, W. F. J. Mol. Graph. 6, 219–220 (1988).
Merritt, E. A. & Murphy, M. E. P. Acta Crystallogr. D 50, 869–873 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kanyo, Z., Scolnick, L., Ash, D. et al. Structure of a unique binuclear manganese cluster in arginase. Nature 383, 554–557 (1996). https://doi.org/10.1038/383554a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/383554a0
This article is cited by
-
Guanidino acid hydrolysis by the human enzyme annotated as agmatinase
Scientific Reports (2022)
-
X-ray Crystallography and Electron Paramagnetic Resonance Spectroscopy Reveal Active Site Rearrangement of Cold-Adapted Inorganic Pyrophosphatase
Scientific Reports (2020)
-
Thermostable arginase from Sulfobacillus acidophilus with neutral pH optimum applied for high-efficiency l-ornithine production
Applied Microbiology and Biotechnology (2020)
-
Histone deacetylase 10 structure and molecular function as a polyamine deacetylase
Nature Communications (2017)
-
Arginase-1 deficiency
Journal of Molecular Medicine (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.