Abstract
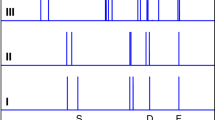
WATER has been studied more extensively than any other liquid, yet its microscopic properties remain poorly understood. The difficulty in obtaining a rigorous molecular-scale description of water structure is largely a consequence of the extended, dynamic hydrogen-bonded network that exists throughout the liquid1. Studies of the structure and dynamics of isolated small clusters of water molecules2–6 provide a means of quantifying the intermolecular forces and hydrogen-bond rearrangements that occur in condensed phases. Experiments2–7 and theory8 strongly suggest that the water trimer, tetramer and pentamer have cyclic minimum energy structures. Larger water clusters are expected8 to have three-dimensional geometries, with the hexamer representing the transition from cyclic to such three-dimensional structures. Here we report investigations by terahertz laser vibration–rotation tunnelling spectroscopy3 of the structure of the water hexamer. A comparison of our results with quantum Monte Carlo simulations of this species suggests that the most stable form of (H2O)6 is indeed a cage-like structure, held together by eight hydrogen bonds (Fig. 1).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Luzar, A. & Chandler, D. Nature 379, 55–57 (1996).
Liu, K., Cruzan, J. D. & Saykally, R. J. Science 271, 929–933 (1996).
Saykally, R. J. & Blake, G. A. Science 259, 1570–1575 (1993).
Pugliano, N. & Saykally, R. J. Science 257, 1937–1940 (1992).
Cruzan, J. D. et al. Science 271, 59–62 (1996).
Liu, K., Brown, M. G., Cruzan, J. D. & Saykally, R. J. Science 271, 62–64 (1996).
Pribble, R. N. & Zwier, Z. S. Science 265, 75–79 (1994).
Kim, K., Jordan, K. D. & Zwier, T. S. J. Am. chem. Soc. 116, 11568–11569 (1994).
Blake, G. A. et al. Rev. Sci. Instrum. 62, 1701–1716 (1991).
Liu, K. et al. Rev. Sci. Instrum. 67, 410–416 (1996).
Tsai, C. J. & Jordan, K. D. Chem. Phys. Lett. 213, 181–188 (1993).
Millot, C. & Stone, A. J. Molec. Phys. 77, 439–462 (1992).
Gregory, J. K. & Clary, D. C. J. chem. Phys. 103, 8924–8930 (1995).
Anderson, J. B. J. chem. Phys. 63, 1499–1503 (1986).
Coker, D. F. & Watts, R. O. Molec. Phys. 58, 1113–1123 (1986).
Buch, V. J. J. chem. Phys. 97, 726–729 (1992).
Speedy, R. J., Madura, J. D. & Jorgensen, W. L. J. phys. Chem. 91, 909–913 (1987).
Belch, A. C. & Rice, S. A. J. Chem. Phys. 86, 5676–5682 (1987).
Narten, A. H., Thiessen, W. E. & Blum, L. Science 217, 1033–1034 (1982).
Peterson, S. W. & Levy, H. A. Acta Crystallogr. 10, 70–76 (1957).
Kuhs, W. F. & Lehmann, M. S. J. phys. Chem. 87, 4312–4313 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, K., Brown, M., Carter, C. et al. Characterization of a cage form of the water hexamer. Nature 381, 501–503 (1996). https://doi.org/10.1038/381501a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/381501a0
This article is cited by
-
Effect of Orientational Isomerism in Neutral Water Hexamers on Their Thermodynamic Properties and Concentrations in the Gas Phase
Journal of Cluster Science (2023)
-
Ortho-para interconversion of nuclear states of H2O through replica transition state: prospect of quantum entanglement at homodromic Bjerrum defect site
Journal of Molecular Modeling (2023)
-
Structure and nature of ice XIX
Nature Communications (2021)
-
Computational molecular spectroscopy
Nature Reviews Methods Primers (2021)
-
Synthesis, structural, and DFT studies of mixed ligand copper(II) malonates
Journal of Chemical Sciences (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.