Abstract
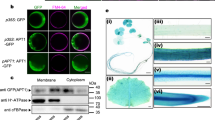
GLUTATHIONE S-transferases (GSTs) are enzymes that detoxify heterocyclic compounds (xenobiotics) by covalently linking glutathione to the substrate, forming a glutathione S-conjugate1,2. A glutathione pump in the vacuolar membrane of barley actively sequesters herbicide–glutathione S-conjugates; glutathionation allows recognition and entry of the conjugates into vacuoles3. The protein encoded by the Bronze-2 gene in maize performs the last genetically defined step in anthocyanin biosynthesis, resulting in the deposition of red and purple pigments in the vacuoles of maize tissues4. We show here that Bz2 encodes a GST with activity in maize, transformed Arabidopsis thaliana plants and Escherichia coli. We demonstrate that anthocyanins extracted from maize protoplasts expressing BZ2 are conjugated with glutathione, and that vanadate, a known inhibitor of the glutathione pump3 in plant vacuolar membranes, inhibits the accumulation of anthocyanins in the vacuole. These results provide a biochemical function for BZ2, and suggest a common mechanism for the ability of plants to sequester structurally similar but functionally diverse molecules in the vacuole.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Pickett, C. B. & Lu, A. Y. H. A. Rev. Biochem. 58, 743–764 (1989).
Ishikawa, T. Trends biochem. Sci. 17, 463–468 (1992).
Martinoia, E., Grill, E., Tommasini, R., Kreuz, K. & Amrhein, N. Nature 364, 247–249 (1993).
Coe, E. H. Jr, Neuffer, M. G. & Hoisington, D. A. Corn and Corn Improvement, 3rd edn (eds Sprague, G. F. & Dudley, J. W.) 81–258 (American Society of Agronomy, Madison, 1988).
Harborne, J. B., Mabry, T. J. & Mabry, H. (eds) The Flavonoids (Chapman & Hall, London, 1975).
Nash, J. & Walbot, V. Pl. Physiol. 100, 464–471 (1993).
Walbot, V., Benito, M.-I., Bodeau, J. P. & Nash, J. Maydica 39, 19–28 (1994).
Schmitz, G. & Theres, K. Molec. Gen. Genet. 233, 269–277 (1992).
Czarnecka, E., Nagao, R. J., Key, J. L. & Gurley, W. B. Molec. cell. Biol. 8, 113–122 (1988).
van der Zaal, E. J. et al. Pl. molec. Biol. 16, 983–998 (1991).
Droog, F. N. J., Hooykaas, P. J. J., Libbenga, K. R. & van der Zaal, E. J. Pl. molec. Biol. 21, 965–972 (1993).
Takahashi, T., Kusaba, M., Hiraoka, Y. & Nagata, Y. Pl. J. 1, 327–332 (1991).
Taylor, J. L. et al. Molec. Pl. Microbe Interact. 3, 72–77 (1990).
Grove, G. et al. Nucleic Acids Res. 16, 425–438 (1988).
Shah, D. M. et al. Pl. molec. Biol. 6, 203–211 (1986).
Irzyk, G. P. & Fuerst, E. P. Pl. Physiol. 102, 803–810 (1993).
Dudler, R., Hertig, C., Rebmann, G., Bull, J. & Mauch, F. Molec. Pl. Microbe Interact. 4, 14–18 (1991).
Timmerman, K. P. Physiologia Pl. 77, 465–471 (1989).
Mannervik, B. & Guthenberg, C. Meth. Enzym. 77, 231–235 (1981).
Goff, S. A. et al. EMBO J. 9, 2517–2522 (1990).
Bodeau, J. P. & Walbot, V. Molec. gen. Genet. 233, 379–387 (1992).
Lloyd, A. M., Walbot, V. & Davis, R. W. Science 258, 1773–1775 (1992).
Piluka, S., Hayden, J. B., Awasthi, S., Awasthi, Y. C. & Zimniak, P. J. biol. Chem. 269, 27566–27573 (1994).
Bowman, E. J., Siebers, A. & Altendorf, K. H. Proc. natn. Acad. Sci. U.S.A. 85, 7972–7976 (1988).
Lamoureux, G. L. & Rusness, D. G. Sulfur in Pesticide Action and Metabolism (eds Rosen, J. D., Magee, P. S. & Casada, J. E.) 133–164 (Am. Chem. Soc., Washington DC, 1981).
Lamoureux, G. L. & Rusness, D. G. Pesticide Chemistry: Human Welfare and the Environment (eds Miyamoto, J. & Kearney, P. C.) 295–300 (Pergamon Press, New York, 1983).
Harborne, J. B. & Self, R. Phytochemistry 26, 2417–2418 (1987).
Harborne, J. B. Phytochemistry 25, 1887–1894 (1986).
Sandermann, H. Jr, Trends biol. Sci. 17, 82–84 (1992).
Valvekens, D., Van Montagu, M. & Van Lijsebettens, M. Proc. natn. Acad. Sci. U.S.A. 85, 5536–5540 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Marrs, K., Alfenito, M., Lloyd, A. et al. A glutathione S-transferase involved in vacuolar transfer encoded by the maize gene Bronze-2. Nature 375, 397–400 (1995). https://doi.org/10.1038/375397a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/375397a0
This article is cited by
-
Genome-wide association study of kernel colour traits and mining of elite alleles from the major loci in maize
BMC Plant Biology (2024)
-
Genome-wide identification and characterization of Glutathione S-Transferases (GSTs) and their expression profile under abiotic stresses in tobacco (Nicotiana tabacum L.)
BMC Genomics (2023)
-
The catalytic role of glutathione transferases in heterologous anthocyanin biosynthesis
Nature Catalysis (2023)
-
Functional Analysis of the Anthocyanin-Associated Glutathione S-Transferase Gene StGST1 in Potato
Potato Research (2023)
-
Flavonoids: a review on biosynthesis and transportation mechanism in plants
Functional & Integrative Genomics (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.