Abstract
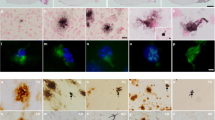
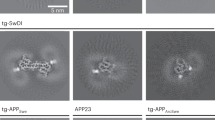
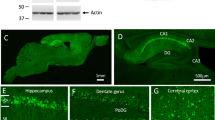
DEPOSITS of β-amyloid are one of the main pathological charac-teristics of Alzheimer's disease. The β-amyloid peptide constituent (relative molecular mass 4,200) of the deposits is derived from the β-amyloid precursor protein (β-APP) which is expressed in several different isoforms1–6. The two most prevalent β-APP isoforms are distinguished by either the presence (β-APP751) or absence (β-APP695) of a Kunitz serine protease inhibitor domain. Changes in the abundance of different β-APP messenger RNAs in brains of Alzheimer's disease victims have been widely reported7–12. Although these results have been controversial, most evidence favours an increase in the mRNAs encoding protease inhibitor-containing isoforms of β-APP and it is proposed that this change contributes to β -amyloid formation9–12. We have now produced an imbalance in the normal neuronal ratio of β-APP isoforms by preparing transgenic mice expressing additional β-APP751 under the control of a neural-specific promoter. The cortical and hip-pocampal brain regions of the transgenic mice display extracellular β -amyloid immunoreactive deposits varying in size (<5–50μm) and abundance. These results suggest that one mechanism of β-amyloid formation may involve a disruption of the normal ratio of neuronal β-APP isoform expression and support a direct relationship between increased expression of Kunitz inhibitor-bearing β-APP isoforms and β-amyloid deposition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Glenner, G. G. & Wong, C. W. Biochem. biophys. Res. Commun. 120, 885–890 (1984).
Masters, C. L. et al. Proc. natn. Acad. Sci. U.S.A. 82, 4245–4249 (1985).
Kang, J. et al. Nature 325, 733–736 (1987).
Ponte, P. et al. Nature 331, 525–528 (1988).
Tanzi, R. E. et al. Nature 331, 528–530 (1988).
Kitaguchi, N., Takahashi, Y., Tokushima, Y., Shiojiri, S. & Ito, H. Nature 331, 530–532 (1988).
Palmert, M. R. et al. Science 241, 1080–1084 (1988).
Neve, R. L., Finch, E. A. & Dawes, L. R. Neuron 1, 669–677 (1988).
Johnson, S. A. et al. Expl Neurol. 102, 264–268 (1988).
Tanaka, S. et al. Biochem. biophys. Res. Commun. 157, 472–479 (1988).
Johnson, S. A., McNeill, T., Cordell, B. & Finch, C. E. Science 248, 854–857 (1990).
Neve, R. L., Rogers, J. & Higgens, G. A. Neuron 5, 329–338 (1990).
Forss-Petter, S. et al. Neuron 5, 187–197 (1990).
Shivers, B. D. et al. EMBO J. 7, 1365–1370 (1988).
Takio, K., Hasegawa, M., Titani, K. & Ihara, Y. Biochem. biophys. Res. Commun. 160, 1296–1301 (1989).
Murphy, G. M. et al. Prog. Neuro-psychopharmac. 14, 309–317 (1990).
Goate, A. et al. Nature 349, 704–706 (1991).
Yamaguchi, H., Hirai, S., Morimatsu, M., Shoji, M. & Ihara, Y. Acta neuropath. 76, 541–549 (1988).
Tagliavini, F., Giaccone, G., Frangione, B. & Bugiani, O. Neurosci. Lett. 103, 191–196 (1988).
Ikeda, S., Allsop, D. & Glenner, G. G. Lab. Invest. 60, 113–122 (1989).
Bugiani, O., Giaconne, G., Frangione, B., Ghetti, B. & Tagliavini, F. Neurosci. Lett. 103, 263–268 (1989).
Sakimura, K., Kushiya, E., Takahashi, Y. & Suzuki, Y. Gene 60, 103–113 (1987).
Okayama, H. & Berg, P. Mol. cell. Biol. 3, 280–289 (1983).
Eklund, J. & Bradford, G. E. Anim. Prod. 22, 127–130 (1976).
Southern, E. M. J. molec. Biol. 98, 503–517 (1975).
Saiki, R. K. et al. Science 239, 487–491 (1988).
Laemmli, U. K. Nature 227, 680–685 (1970).
Tobin, H., Staehelin, T. & Gordon, J. Proc. natn. Acad. Sci. U.S.A. 76, 4350–4354 (1979).
Taggart, R. T. & Samloff, I. M. Science 219, 1228–1230 (1983).
Allsop, D. et al. Neuropath. appl. Neurobiol. 15, 531–542 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Quon, D., Wang, Y., Catalano, R. et al. Formation of β-amyloid protein deposits in brains of transgenic mice. Nature 352, 239–241 (1991). https://doi.org/10.1038/352239a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/352239a0
This article is cited by
-
Diplazium esculentum (Retz.) Sw. reduces BACE-1 activities and amyloid peptides accumulation in Drosophila models of Alzheimer’s disease
Scientific Reports (2021)
-
The Interplay of Tau Protein and β-Amyloid: While Tauopathy Spreads More Profoundly Than Amyloidopathy, Both Processes Are Almost Equally Pathogenic
Cellular and Molecular Neurobiology (2021)
-
Prominent amyloid plaque pathology and cerebral amyloid angiopathy in APP V717I (London) carrier – phenotypic variability in autosomal dominant Alzheimer’s disease
Acta Neuropathologica Communications (2020)
-
History and progress of hypotheses and clinical trials for Alzheimer’s disease
Signal Transduction and Targeted Therapy (2019)
-
Alzheimer’s disease-like APP processing in wild-type mice identifies synaptic defects as initial steps of disease progression
Molecular Neurodegeneration (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.