Abstract
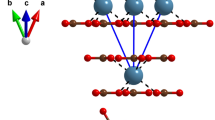
IN two very interesting papers (Proc. Roy. Soc., vol. 105, p. 370, and vol. 106, p. 346, 1924) Prof. W. L. Bragg put forward an explanation of the strong birefringence exhibited by the crystalline carbonates and nitrates. Expressed very briefly, his theory is that in the carbonate and nitrate ions the oxygen atoms are situated in one plane around the central carbon or nitrogen atom, as the case may be, and that, as the result of this arrangement and of the mutual influence of the electric doublets induced in the atoms by the field of the light-waves, the refractivity of the group depends to a marked extent on the direction of the light-vector. The refractive indices of the crystal were successfully computed on this basis. The crystalline sulphates are known, on the other hand, to have a very weak birefringence, and the suggestion was made that very probably the oxygen atoms are arranged tetrahedrally round the sulphur atom in the sulphate ion group, thus making it optically isotropic.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
RAMAN, C. The Birefringence of Crystalline Carbonates, Nitrates and Sulphates. Nature 118, 264–265 (1926). https://doi.org/10.1038/118264b0
Issue Date:
DOI: https://doi.org/10.1038/118264b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.