Abstract
bcl-2 overexpression in synovial sarcomas has been recently reported. Although it is widely known that bcl-2 suppresses apoptosis in various cells, there are no studies that have examined the significance of apoptosis in synovial sarcoma. In the present study, we visualized apoptotic tumor cells by the terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate in situ nick end-labeling (TUNEL) method in 49 cases of primary synovial sarcoma. The degree of apoptosis was analyzed in relation to several clinicopathologic parameters, cell proliferative activity, and immunohistochemical expression of apoptosis-related proteins, including bcl-2, bax, bcl-x, bak, p53, p21 (WAF1/CIP1), Fas, and Fas ligand. TUNEL index (TUNEL-I) significantly correlated with the mitotic index (MI) (ñ = 0.60, P < .0001) and Ki-67 labeling index (MIB1-I) (ñ = 0.52, P = 0.0005). There was a highly significant association between high TUNEL-I value (>.8%) and poor prognosis (log-rank test; P < .0001). Many synovial sarcomas were diffusely positive for bcl-2 family proteins (bcl-2, bax, bcl-x, and bak) and were negative or only sporadically positive for Fas, Fas ligand, p53, and p21 (WAF1/CIP1) proteins. The results indicated that increased rate of apoptosis in primary synovial sarcoma was considered to be an indicator of poor prognosis. In addition, apoptosis in synovial sarcoma may be controlled by multiple apoptosis-regulating mechanisms, including the bcl-2 family.
Similar content being viewed by others
INTRODUCTION
Synovial sarcoma is a soft tissue sarcoma of unknown histogenesis, occurring most frequently in adolescents and young adults. It is classified into three subtypes: the biphasic type composed of both epithelial and spindle-cell components, the monophasic type composed of either epithelial or spindle-cell component, and the poorly differentiated type (1). Most synovial sarcomas are characterized by nonrandom chromosomal translocation, t(18;X)(p11.2;q11.2) (2). Reverse transcription-polymerase chain reaction (RT-PCR) for chimeric SYT-SSX fusion transcript originating from the specific translocation is now regarded as a diagnostic tool of this tumor (2). A number of investigators regard synovial sarcoma as “soft tissue carcinosarcoma” because it shares morphologic and immunologic properties of the epithelium as well as mesenchyme (3). The clinical behavior of synovial sarcoma has been described to correlate with some morphologic and biologic parameters (e.g., histologic subtype, percentage glandularity, tumor necrosis, and cell proliferative activity determined by mitotic rate or immunohistochemical staining of proliferating cell nuclear antigen) (4, 5).
Recent studies have shown overexpression of bcl-2 protein in synovial sarcomas (6, 7, 8). bcl-2 proto-oncogene was initially described in B-cell lymphomas (9), and the bcl-2 protein is known to prolong the survival of a variety of cells by blocking apoptosis (10). Apoptosis is a highly conserved, genetically mediated process of selective cell deletion that occurs during embryogenesis and in normal tissue homeostasis as well as in various diseases (11). Apoptosis is also a fundamental process in tumor cell kinetics, and susceptibility to apoptosis in untreated malignant tumors varies with tumor type (12). Although the precise mechanisms that regulate apoptosis of tumor cells are not completely understood, there are numerous endogenous and exogenous initiating factors (e.g., growth factor deprivation, Fas and its ligand, viral infection, irradiation, cytotoxic chemotherapy) (11, 12, 13). In addition, apoptosis is controlled by a number of proto-oncogenes and tumor suppressor genes, including bcl-2 (and its related genes), p53, Rb, and c-myc (11, 12, 13, 14).
Because apoptosis in malignant tumors may account for spontaneous cell loss, its potential prognostic value and therapeutic application have been of great interest in recent years. In the present study, we evaluated the extent of apoptosis in primary synovial sarcomas with the terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate in situ nick end-labeling (TUNEL) method (15). The extent of apoptosis was analyzed in correlation with several clinicopathologic parameters, extent of cell proliferation, and immunohistochemical expression of various apoptosis-regulating factors (e.g., bcl-2 family (16), p53 (13, 17), p21(WAF1/CIP1) (18), Fas (19), and Fas ligand (20) proteins). The aims of this study were (1) to assess the extent of apoptosis in primary synovial sarcomas; (2) to clarify the relationship among apoptosis, cell proliferative activity, and expression of apoptosis-related proteins; and (3) to elucidate the significance of apoptosis in the prognosis of synovial sarcoma.
MATERIALS AND METHODS
Cases of primary synovial sarcoma, which had been procured from 1963 to 1996, were selected from three sources: medical records in the Second Department of Pathology Laboratory, Kyushu University; Department of Pathology, National Kyushu Medical Center; and Department of Pathology, National Kyushu Cancer Center. Formalin-fixed, paraffin-embedded tissue blocks were available for 49 patients. All patients had not had preoperative chemotherapy or radiotherapy at the time of initial surgery. All tumors were staged according to the American Joint Committee on Cancer (AJCC) staging of soft tissue sarcomas (21).
Synovial sarcomas were diagnosed using hematoxylin and eosin (H&E), periodic acid-Schiff with and without diastase digestion, colloidal iron for mucin with and without hyaluronidase digestion, silver impregnation for reticulin, and/or immunohistochemical staining of cytokeratin and vimentin. RT-PCR for SYT-SSX fusion transcript was used for diagnostic confirmation in four tumors, including one from which a previously reported synovial sarcoma cell line, SN-SY-1, was established (22). All microscopic slides were reexamined to confirm the diagnosis, and the most representative one or two paraffin-embedded tissue blocks were used for subsequent studies.
Histologic Study and Assessment of Mitosis
Necrosis was categorized as absent (“not”) and present (“any”) by examining H&E sections of each synovial sarcoma. Mitotic nuclei were counted on each representative H&E section. The mitotic index (MI) was determined by counting the number of mitoses in four separate sets of 10 high-power fields (× 400) and selection of the highest counts.
TUNEL and Assessment of Apoptosis
Visualization of DNA fragmentation, a marker of apoptosis, was performed by the TUNEL method using the In Situ Apoptosis Detection Kit (Takara Biomedicals, Kusatsu, Japan) according to the instructions provided by the manufacturer. Briefly, formalin-fixed, paraffin-embedded synovial sarcoma tissue sections were dewaxed and rehydrated, and nucleic acids were stripped from proteins by treatment with 15 μg/mL proteinase K (Sigma, St. Louis, MO) in 0.01 mol/L phosphate-buffered saline (PBS), pH 7.4, for 15 min at room temperature. Sections were reacted with terminal deoxynucleotidyl transferase (TdT; Takara Biomedicals) and fluorescein isothiocyanate (FITC)- deoxyuridine triphosphate (dUTP; Takara Biomedicals) for 60 min at 37° C in a humid chamber. Sections were then incubated with horseradish peroxidase (HRP)-conjugated murine anti-FITC antibody (Takara Biomedicals) for 30 min at 37° C. End labeling of fragmented DNA was visualized with hydrogen peroxide containing 3,3′-diaminobenzidine tetrahydrochloride (DAB)/PBS. Paraffin-embedded sections of normal human lymph nodes and gastric mucosa were used as positive controls. The presence of a clear nuclear staining was indicative of apoptotic cells.
In each section, at least 5000 synovial sarcoma cells were examined in the most evenly and distinctly labeled areas at high-power field (× 400), and the number of TUNEL-positive tumor cells was counted. In counting TUNEL-positive synovial sarcoma cells, viable tumor areas just around necrosis were avoided. The TUNEL index (TUNEL-I) was calculated as percentage of TUNEL-positive tumor cells per examined tumor cells. Assessment of TUNEL-I was performed independently by three authors (SK, TF, YO), and the validity was statistically confirmed by the Friedman test using the StatView 4.02 software (Abacus Concepts, Inc., Berkeley, CA). TUNEL-I data assessed by the first author (SK) was used in the following study.
Ki-67 Immunostaining and Assessment
Four-μm-thick synovial sarcoma tissue sections were cut from formalin-fixed, paraffin-embedded tissue blocks. They were dewaxed in xylene, rehydrated through graded ethanol solutions, rinsed in PBS for 5 min, and then immersed in 0.3% hydrogen peroxide in methanol for 30 min to block endogenous peroxidase. For antigen retrieval, sections were then microwaved in 0.01 mol/L sodium citrate-buffered saline (CBS), pH 6.0, for 30 min at 95° C using a Microwave Processor, model H2800 (Energy Beam Science, Inc., Washington, DC). After slides were rinsed in PBS for 5 min, they were blocked with a solution of 10% normal rabbit serum in PBS at room temperature for 10 min and incubated at 4° C overnight with the murine monoclonal anti–Ki-67 antibody (MIB1, Immunotech, Marseille, France) at a dilution of 1:100 in 1.0% bovine serum albumin (BSA)/PBS. Subsequent reaction was made by the streptavidin-biotin complex/horseradish peroxidase (SAB/HRP) method using a HISTOFINE SAB-PO (M) Immunohistochemical Staining Kit (Nichirei, Tokyo, Japan) according to the protocol provided by the manufacturer. Positive reactions were visualized with hydrogen peroxide containing DAB/PBS.
Positively stained tumor cell nuclei were counted in the most evenly and distinctly labeled areas. The Ki-67 labeling index (MIB1-I) was expressed as the percentage of positively stained nuclei based on the count of at least 2000 tumor cells.
Immunostaining for Apoptosis-Related Proteins
Immunohistochemical stainings were performed with primary antibodies listed in Table 1. For antigen retrieval of bcl-2, bax, Fas, and Fas ligand proteins, dewaxed tumor tissue sections were incubated in 0.1% trypsin (Sigma)/PBS for 15 min (for bcl-2, Fas, and Fas ligand proteins) and 30 min (for bax protein) at 37° C. For antigen retrieval of bcl-x, bak, p53, and p21(WAF1/CIP1) proteins, dewaxed tumor tissue sections were microwaved in CBS for 30 min at 100° C using a H2800 Microwave Processor. The procedure of immunohistochemical staining was similar to that described in Ki-67 (MIB1) immunohistochemical staining, using a HISTOFINE SAB-PO (M) (for mouse primary antibodies), SAB-PO (R) (for rabbit primary antibodies) or SAB-PO (G) (for goat primary antibody) Immunohistochemical Staining Kit (Nichirei). Positive reactions were visualized with hydrogen peroxide containing DAB/PBS.
The expression of apoptosis-related proteins was semiquantitatively evaluated as negative (−; no positively stained tumor cells were present), low expression (+; only a few [<10%] positively stained tumor cells were present), moderate expression (++), or extensive expression (+++; numerous [>50%] positively stained tumor cells were present).
Statistical Analysis
Results were analyzed using the StatView 4.02 software. Correlation between TUNEL-I and categorized data of age of patients; gender; necrosis; tumor size; and expression of p53, p21 (WAF1/CIP1), bcl-x, or bak protein was analyzed by the Mann-Whitney U test. Correlation between TUNEL-I and histologic type, categorized expression of bcl-2 or bax protein, was analyzed by the Kruskal-Wallis test. Correlations among TUNEL-I, MI, and MIB1-I were examined by the Spearman rank correlation test and Mann-Whitney U test. Survival curves were constructed by the Kaplan-Meier method and compared by the log-rank test. Cox proportional hazards regression analysis with stepwise selection of variables was used to assess the relative importance of different factors on survival. A two-tailed P value of less than 0.05 was considered significant.
Double Fluorescence Labeling with TUNEL and bcl-2 Immunohistochemistry
Double fluorescence labeling with TUNEL and bcl-2 immunohistochemistry was performed using formalin-fixed, paraffin-embedded tissues of five synovial sarcomas. After dewaxing and rehydration, sections were treated with 15 μg/mL proteinase K for 5 to 7 min at room temperature. After sections were washed with PBS, they were incubated with a murine monoclonal antibody to bcl-2 protein (124; DAKO, Glostrup, Denmark) at a dilution of 1:40 for 60 min at room temperature and detected with a tetramethylrhodamine isothiocyanate (rhodamine)-conjugated rabbit antimouse IgG antibody (DAKO) diluted at 1:40. After sections were washed with PBS, they were then incubated in a humid chamber with a solution containing TdT and FITC-dUTP for 60 min at 37° C. After sections were washed with PBS and mounted with Antifade (Oncor, Gaithersburg, MD), they were examined under an Axiophot Fluorescence Microscope (Carl Zeiss Japan, Tokyo, Japan).
RESULTS
Clinicopathologic Features
Patients included 23 male and 26 females, with a mean age of 39.6 years (range, 9 to 70 years). Primary tumors were located in the lower extremities in 35 patients, upper extremities in 12 patients, and the trunk in 2 patients. Staging of the primary tumors was as follows: Stage I, 18 patients; Stage II, 25 patients; and Stage III, 6 patients. The length of follow-up period was calculated from the date of initial surgical treatment and ranged from 2 to 229 months (mean, 60.9 months). Histologically, 14 tumors were of the biphasic type, 34 were of the monophasic fibrous type, and 1 was a poorly differentiated tumor. At the conclusion of this study, 20 patients were still alive and 29 had died of the disease.
Apoptosis
TUNEL-positive synovial sarcoma cells, many showing homogeneously stained condensed nuclei, were found scattered among viable tumor cells (Fig. 1). In general, the nuclear labeling of TUNEL reaction was distinct, making it easy to identify reactive nuclei. On occasion, TUNEL-positive tumor cells were more frequently observed in viable tumor areas around necrosis. In a small number of well-differentiated biphasic synovial sarcomas, TUNEL-positive tumor cells were more frequently observed in epithelial glandular areas than in fibrous areas.
TUNEL-I ranged from 0.1 to 2.7% (mean, 0.84%). In categorized data analysis to examine the correlation between TUNEL-I and clinicopathologic parameters, TUNEL-I of the large tumor group (>8 cm) was significantly higher than that of the small tumor group (≤8 cm; P = .01; Table 2).
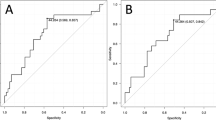
Univariate survival analysis by the Kaplan-Meier method and log-rank test using a TUNEL-I cutoff value of 0.8% showed that the survival rate of patients with low (≤0.8%) TUNEL-I values was significantly higher than that with high (>0.8%) TUNEL-I values (P < .0001; Fig. 2). In survival analysis, we tentatively defined 0.6, 0.7, 0.8, 0.9, or 1.0% as a cutoff for high versus low TUNEL-I value; 0.8% had the most significant prognostic effect on survival and finally was selected as the cutoff value.
Cell Proliferation
Cell proliferative activity of synovial sarcomas as determined by MIB1-I and MI demonstrated a considerable variability among cases. Immunohistochemical staining for Ki-67 antigen using MIB1 antibody was usually distinct in the nuclei of synovial sarcoma cells, making identification of Ki-67–positive tumor cells very easy (Fig. 3). In biphasic synovial sarcomas, the extent of Ki-67–positive tumor cells was similar in both epithelial glandular and spindle-cell fibrous areas.
MIB1-I ranged from 0.2 to 23.8% (mean, 7.12%), and MI ranged from 1 to 25 (mean, 8.63). There was a significant linear correlation between MI and MIB1-I (Spearman rank correlation test, ñ = .88; P < .0001; Fig. 4).
Univariate survival analysis using an MI cutoff value of 15 demonstrated that survival rate of patients with synovial sarcoma of low MI value (≤15) was significantly higher than that of high MI value (>15) (log-rank test; P = .023; Fig. 5). Univariate survival analysis using an MIB1-I cutoff value of 11% also showed that survival rate of patients with synovial sarcoma of low MIB1-I value (≤11%) was significantly higher than that of high MIB1-I value (>11%; log-rank test; P = .0095; Fig. 6).
Apoptosis and Cell Proliferation
With regard to apoptosis and cell proliferation, there were significant linear correlations between TUNEL-I and MI (Spearman rank correlation test, ñ = .60; P < .0001; Fig. 7) and between TUNEL-I and MIB1-I (Spearman rank correlation test, ñ = .52; P = .0005; Fig. 8).
Mann-Whitney U test showed a significant difference of TUNEL-I values between the synovial sarcoma group of low MI value (≤15) and that of high MI value (>15) (P = .0007; Table 3). Mann-Whitney U test also exhibited a significant difference of TUNEL-I values between synovial sarcoma group of low MIB1-I value (≤11%) and that of high MIB1-I value (>11%; P = .0057; Table 3).
Apoptosis-Related Proteins
Immunohistochemically, bcl-2, bax, bcl-x, and bak proteins were positive in the majority of examined synovial sarcomas and showed diffuse perinuclear and/or cytoplasmic staining (Fig. 9). A proportion of biphasic synovial sarcomas exhibited a topographic immunostaining pattern for bcl-2 (Fig. 10) and bax proteins, and their intense staining was observed predominantly in fibroblastic spindle-cell areas. p53 and p21 (WAF1/CIP1) proteins were negative or sporadically positive in the nuclei of spindle-shaped fibroblastic or epithelial glandular tumor cells of a small number of monophasic and biphasic synovial sarcomas (Fig. 11). Both Fas and Fas ligand proteins were immunohistochemically negative in most synovial sarcomas and were sporadically positive in infiltrating inflammatory cells.
Immunohistochemical staining for p53 and p21 (WAF1/CIP1) proteins in synovial sarcoma. A, sporadic immunoreactivity for p53 protein is observed in the spindle-shaped, fibroblastic tumor cells of monophasic fibrous synovial sarcoma; B, epithelial glandular tumor cells of biphasic synovial sarcoma are positive for p21 (WAF1/CIP1) protein.
Synovial sarcoma groups that were categorized by the extent of immunohistochemical staining of bcl-2, bax, bcl-x, bak, p53, and p21 (WAF1/CIP1) proteins failed to show any statistical differences of the TUNEL-I values (Table 4) and had no significant prognostic effect on survival (data not shown).
Multivariate Survival Analysis
In addition to MI, MIB1-I, and TUNEL-I, univariate survival analysis by the Kaplan-Meier method and log-rank test revealed that advanced AJCC stage (P < .0001), tumor necrosis (P = .014), and tumor size of more than 5 cm (P = .014) significantly correlated with poor survival. The six variables were entered into multivariate analysis by the Cox proportional hazards regression model with stepwise selection of variables, revealing that AJCC stage and TUNEL-I correlated significantly and independently with survival (Table 5).
Double Fluorescence Labeling with TUNEL and bcl-2 Immunohistochemistry
Double fluorescence labeling with TUNEL and bcl-2 immunostaining showed that a proportion of TUNEL-positive, apoptotic synovial sarcoma cells expressed bcl-2 protein (Fig. 12).
Double fluorescence labeling with triphosphate in situ nick end-labeling and bcl-2 immunohistochemistry in synovial sarcoma. Note a synovial sarcoma cell with triphosphate in situ nick end-labeling–positive (fluorescein isothiocyanate–labeled) nuclei and immunopositive (rhodamine-labeled) cytoplasm for bcl-2 protein.
DISCUSSION
We established a new synovial sarcoma cell line, SN-SY-1, which showed diffuse perinuclear and cytoplasmic immunoreactivity for bcl-2 protein, and Southern blot analysis demonstrated no rearrangement of the bcl-2 gene (22). In addition, ultrastructural localization of bcl-2 protein in cultured synovial sarcoma cells was similar to that previously reported in benign and malignant lymphoid and epithelial cells (23). We thus speculated that the bcl-2 protein might play a role in the tumorigenesis of synovial sarcoma by suppressing apoptosis in a manner similar to that described for lymphomas (22, 23). The present study represents an extension of these early findings and was designed to examine the significance of apoptosis in synovial sarcoma.
Since the establishment of morphologic criteria of apoptosis, several studies have examined apoptosis in various human tumors. Apoptotic cells can be recognized by nuclear shrinkage, compaction, segregation of chromatin against the nuclear envelope, and condensation of the cytoplasm (24). However, such recognition of apoptotic cells is not necessarily easy because of the variable degree of similarity between these cells and inflammatory cells or mitotic figures. In the present study, to facilitate the distinction of apoptotic synovial sarcoma cells, we used the TUNEL method (15), a standard technique for identifying fragmented nuclear DNA, which also enabled us to easily recognize apoptotic nuclear changes that were not always reliably identified on H&E sections.
Apoptosis in human cancers has been described in relation to high tumor grade, progression, and high mitotic rate (25, 26, 27, 28). However, the prognostic value of the frequency of apoptotic tumor cells is still controversial (25). In the present study, the extent of apoptosis (TUNEL-I) in primary synovial sarcomas showed a significant linear correlation with the mitotic rate (MI) and cell proliferative activity determined by Ki-67 immunostaining (MIB1-I). Similar relationships between apoptosis and cell proliferation have been reported in other tumor types (26, 29, 30), suggesting a mechanistic link between apoptosis and cell proliferation. The relationship between apoptosis and cell proliferation may be explained in part by the fact that many proteins operating in the cell cycle checkpoints (e.g., p53; Rb; cyclins D1, A, and B) are also regulators and inducers of apoptosis (25).
In the present study, elevated TUNEL-I significantly correlated with poor prognosis in synovial sarcoma. In multivariate analysis, it was proved that TUNEL-I had independent prognostic value. A similar association between high apoptotic rate and poor prognosis has been described in other tumors (e.g., non–small cell lung carcinoma (31), breast carcinoma (32), and ovarian carcinoma (27)). Because it is widely known that elevated cell proliferation generally is associated with poor prognosis in many cancers, including synovial sarcoma (4, 5), the association between high apoptotic rate and poor prognosis in synovial sarcoma could be explained by the correlation between apoptosis and cell proliferation.
Hirakawa et al. (6) first examined the expression of bcl-2 protein in synovial sarcomas using formalin-fixed, paraffin-embedded tissues and reported that 15 of 19 cases (79%) were immunohistochemically positive. More recent, Suster et al. (8) examined 70 cases of synovial sarcoma and detected a positive immunostaining for bcl-2 protein in all cases. In the present immunohistochemical study using paraffin-embedded tissues, bcl-2 protein was positive in all synovial sarcomas and showed a moderate (++) to extensive (+++) staining in 44 of the 49 tumors (90%). The mean TUNEL-I of the examined synovial sarcomas was relatively low (0.84%), compared with that reported in colon carcinomas (1.9%) (33), small cell lung carcinomas (2.3%) (34), and ovarian carcinomas (1.3%) (27). It might be possible that the low TUNEL-I value in synovial sarcoma is due to the suppressive effect of overexpressed bcl-2 protein.
Recently, various forms of the bcl-2 family have been shown to be involved in the control of cell viability (16, 35, 36). Some members (e.g., bcl-2, bcl-xL [large molecular weight product of bcl-x gene], and mcl-1 proteins) inhibit apoptosis, whereas others (e.g., bax, bcl-xS [small molecular weight product of bcl-x gene], bak, and bad proteins) promote it, and the ratio of different family members (e.g., bcl-2/bax) seems to determine the survival or death of cells after apoptotic stimuli (16). In the present study, immunohistochemical staining for bax, bcl-x (including both bcl-xL and bcl-xS), and bak proteins also exhibited a diffuse perinuclear and cytoplasmic positivity in most monophasic and biphasic synovial sarcomas as well as bcl-2 protein. Those members of the bcl-2 family were supposed to be involved in the regulation of apoptosis in synovial sarcoma. However, our present study failed to elucidate any significant correlation between the extent of apoptosis and immunohistochemical expression of bcl-2, bax, bcl-x, or bak protein.
Although synovial sarcomas have been shown to diffusely express bcl-2 protein (6, 7, 8), it is unknown whether apoptotic synovial sarcoma cells simultaneously express bcl-2 protein in vivo. In this study, double fluorescence staining with bcl-2 immunohistochemistry and TUNEL identified a considerable number of apoptotic synovial sarcoma cells that expressed bcl-2 protein. The finding indicates that apoptosis occurs even in synovial sarcoma cells that express bcl-2. Accordingly, it was assumed that some apoptosis-promoting factors (e.g., bax, bak) may be involved in the promotion of apoptosis in such synovial sarcoma cells. Diffuse immunohistochemical expression of bax and bak proteins in synovial sarcomas seemed to support this hypothesis; however, double fluorescence labeling with TUNEL and immunohistochemistry for bax or bak protein failed to successfully detect apoptotic synovial sarcoma cells that express bax or bak protein (data not shown). Proteolytic pretreatment of the TUNEL might damage epitopes or cellular structures where the proteins were present and, as a result, impair the immunoreactivity. We used formalin-fixed, paraffin-embedded synovial sarcoma tissue in the double fluorescence experiment; however, fresh-frozen tissue might be preferable.
Recent studies have shown that many tumor cells express both Fas and Fas ligand on their surface. The Fas-Fas ligand system is known as one of the major pathways for induction of apoptosis in cells and tissues (37). On contact with Fas ligand, Fas-bearing cells rapidly undergo apoptosis (19, 20). In the present immunohistochemical study, synovial sarcomas showed rare immunopositivity for both Fas and Fas ligand proteins. Accordingly, Fas-Fas ligand system was considered not likely to play an important role in the induction of apoptosis in primary synovial sarcomas.
Wild-type p53 prevents cells with damaged DNA from proliferation by arresting the cell division cycle until such damage is repaired or by pushing the damaged cell down an irreversible apoptotic pathway; however, mutant p53 is known to inhibit apoptosis (13, 17, 38). Immunohistochemical staining of p53 protein using a monoclonal antibody, (e.g., PAb 1801), which recognizes domains present in both the wild and mutated p53 proteins, was believed predominantly to detect mutated p53 protein because the wild-type p53 protein is rapidly degraded in routinely handled tissue (39). Whereas recent studies have shown that immunohistochemical p53 overexpression is not necessarily linked with p53 gene mutation (40), p21 (WAF1/CIP1), a downstream p53 effector gene, is activated by wild-type p53 protein and is involved in the regulation of cell cycle G1-S transition and apoptosis (18, 41, 42). In the current immunohistochemical study, p53 and p21 (WAF1/CIP1) proteins both were sporadically positive mainly in the tumor cells of a small number of monophasic and biphasic synovial sarcomas, and the expression seemed to have no remarkable association with the extent of apoptosis, clinicopathologic parameters, and expression of bcl-2 family proteins.
In summary, the present study demonstrated that extent of apoptosis in primary synovial sarcomas correlated with that of cell proliferative activity and increased apoptosis was an indicator of poor prognosis and survival. A number of apoptotic synovial sarcoma cells that were detected by TUNEL expressed bcl-2 protein. In addition, our immunohistochemical study concerning apoptosis-related proteins revealed that multiple bcl-2 family proteins were expressed in synovial sarcomas and possibly involved in the regulation of apoptosis. Although the mechanism of apoptosis in primary synovial sarcoma is not completely clarified, increased rate of apoptosis is likely to be a useful marker for aggressive tumor growth and poor prognosis.
References
Enzinger FM, Weiss SW . Synovial sarcoma. In: Enzinger FM, Weiss SW, editors. Soft tissue tumors. 3rd ed. St. Louis: CV Mosby; 1995. p. 757–786.
Fligman I, Lonardo F, Jhanwar SC, Gerald WL, Woodruff J, Ladanyi M . Molecular diagnosis of synovial sarcoma and characterization of a variant SYT-SSX2 fusion transcript. Am J Pathol 1995; 147: 1592–1599.
Miettinen M, Virtanen I . Synovial sarcoma: a misnomer. Am J Pathol 1984; 117: 18–25.
Oda Y, Hashimoto H, Tsuneyoshi M, Takeshita S . Survival in synovial sarcoma: a multivariate study of prognostic factors with special emphasis on the comparison between early death and long term survival. Am J Surg Pathol 1993; 17: 35–44.
Oda Y, Hashimoto H, Takeshita S, Tsuneyoshi M . The prognostic value of immunohistochemical staining for proliferating cell nuclear antigen in synovial sarcoma. Cancer 1993; 72: 478–485.
Hirakawa N, Naka T, Yamamoto I, Fukuda T, Tsuneyoshi M . Overexpression of bcl-2 protein in synovial sarcoma: a comparative study of other soft tissue spindle cell sarcomas and an additional analysis by fluorescence in-situ hybridization. Hum Pathol 1996; 27: 1060–1065.
Pilotti S, Mezzelani A, Azzarelli A, Rilke F, Pierotti MA . bcl-2 expression in synovial sarcoma. J Pathol 1998; 184: 337–338.
Suster S, Fisher C, Moran CA . Expression of bcl-2 oncoprotein in benign and malignant spindle cell tumors of soft tissue, skin, serosal surface, and gastrointestinal tract. Am J Surg Pathol 1998; 22: 863–872.
Tsujimoto Y, Croce CM . Analysis of the structure, transcripts, and protein products of bcl-2, the gene involved in human follicular lymphoma. Proc Natl Acad Sci U S A 1986; 83: 5214–5218.
Hockenbery DM, Nunez G, Milliman C, Schreiber, Korsmeyer SJ . Bcl-2 is an inner mitochondrial membrane protein that blocks programmed cell death. Nature 1990; 348: 334–336.
Wyllie AH . Apoptosis and the regulation of it in normal and neoplastic tissues: an overview. Cancer Metastasis Rev 1992; 11: 95–103.
Staunton MJ, Gaffiney EF . Tumor type is a determinant of susceptibility to apoptosis. Am J Clin Pathol 1995; 103: 300–307.
Kerr JFR, Winterford CM, Harmon AV . Apoptosis. Its significance in cancer and cancer therapy. Cancer 1994; 73: 2013–2016.
Hoffman B, Liebermann DA . Molecular controls of apoptosis: differentiation/growth arrest primary response genes, proto-oncogenes, and tumor suppressor genes as positive and negative modulators. Oncogene 1994; 9: 1807–1812.
Gavrieli Y, Shermen Y, Ben-Sasson SA . Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 1992; 119: 493–501.
Oltvai ZN, Milliman CL, Korsmeyer SJ . Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 1993; 74: 609–619.
Yonish-Rouach E, Resnitzky D, Lotem J, Sachs L, Kimchi A, Oren M . Wild-type p53 induces apoptosis of myeloid leukaemic cells that is inhibited by interleukin-6. Nature 1991; 352: 345–347.
El-Deiry WS, Harper JW, O’Connor PM, Velculescu VE, Canman CE, Jackman J, et al. WAF1/CIP1 is induced in p53-mediated G1 arrest and apoptosis. Cancer Res 1994; 54: 1169–1174.
Itoh N, Yonehara S, Ishii A, Yonehara M, Mizushima S, Sameshima M, et al. The polypeptide encoded by the cDNA for human cell surface antigen Fas can mediate apoptosis. Cell 1991; 26: 233–243.
Suda T, Takahashi T, Golstein P, Nagata S . Molecular cloning and expression of the Fas ligand, a novel member of the tumor necrosis factor family. Cell 1993; 75: 1169–1178.
Russell WO, Cohen J, Enzinger F, Hajdu SI, Heise H, Martin RG, et al. A clinical and pathological staging system for soft tissue sarcomas. Cancer 1977; 40: 1562–1570.
Noguchi S, Ueki T, Kawauchi S, Fukuda T, Matsuura H, Sonoda T, et al. Establishment and characterization of a new synovial sarcoma cell line, SN-SY-1: specific reference to bcl-2 protein and SYT-SSX1 hybrid transcripts. Int J Cancer 1997; 72: 995–1002.
Kawauchi S, Noguchi S, Fukuda T, Tsuneyoshi M . Subcellular localization of bcl-2 protein in synovial sarcoma. Pathol Int 1997; 47: 854–861.
Majno G, Joris I . Apoptosis, oncosis, and necrosis: an overview of cell death. Am J Pathol 1995; 146: 3–19.
Soini Y, Paakko P, Lehto V-P . Histopathological evaluation of apoptosis in cancer. Am J Pathol 1998; 153: 1041–1053.
Sasatomi E, Tokunaga O, Miyazaki K . Spontaneous apoptosis in gallbladder carcinoma: relationships with clinicopathologic factors, expression of E-cadherin, bcl-2 protooncogene, and p53 oncosuppressor gene. Cancer 1996; 78: 2101–2110.
McMenamin ME, O’Neil AJ, Gaffney EF . Extent of apoptosis in ovarian serous carcinoma: relation to mitotic and proliferative indices, p53 expression, and survival. J Clin Pathol Mol Pathol 1997; 50: 242–246.
Berardo MD, Elledge RM, Moor C, Clark GM, Osborne CK, Allred DC . bcl-2 and apoptosis in lymph node positive breast carcinoma. Cancer 1998; 82: 1296–1302.
Leoncini L, Del Vecchio MT, Megha T, Barbini P, Galieni P, Pileri S, et al. Correlations between apoptotic and proliferative indices in malignant non-Hodgkin’s lymphomas. Am J Pathol 1993; 142: 755–763.
Lipponen PK, Aaltomaa S . Apoptosis in bladder cancer as related to standard prognostic factors and prognosis. J Pathol 1994; 173: 333–339.
Tormanen U, Eerola A-K, Rainio P, Vahakangas K, Soini Y, Sormunen R . Enhanced apoptosis predicts shortened survival in non-small cell lung carcinoma. Cancer Res 1995; 55: 5595–5602.
Lipponen P, Aaltomaa S, Kosma V-M, Syrjanen K . Apoptosis in breast carcinoma as related to histopathological characteristics and prognosis. Eur J Cancer 1994; 30A: 2068–2073.
Baretton GB, Diebold J, Christoforis G, Vogt M, Muller C, Dopfer K, et al. Apoptosis and immunohistochemical bcl-2 expression in colorectal adenomas and carcinomas: aspects of carcinogenesis and prognostic significance. Cancer 1996; 77: 255–264.
Eerola A-K, Tormanen U, Rainio P, Sormunen R, Bloigu R, Vahakangas K, et al. Apoptosis in operated small cell lung carcinoma is inversely related to tumour necrosis and p53 immunoreactivity. J Pathol 1997; 181: 172–177.
Craig RW . The bcl-2 gene family. Semin Cancer Biol 1995; 6: 35–43.
Krajewska M, Moss SF, Krajewski S, Song K, Holt PR, Reed JC . Elevated expression of Bcl-X and reduced Bak in primary colorectal adenocarcinoma. Cancer Res 1996; 56: 2422–2427.
Nagata S . Apoptosis by death factor. Cell 1997; 88: 355–365.
Chang F, Syrjanen S, Tervahauta A, Syrjanen K . Tumourigenesis associated with the p53 tumour suppressor gene. Br J Cancer 1993; 68: 653–661.
Purdie CA, O’Grady J, Piris J, Wyllie AH, Bird CC . p53 expression in colorectal tumors. Am J Pathol 1991; 138: 807–813.
Tolbert DM, Noffsinger AE, Miller MA, DeVoe GW, Stemmermann GN, Macdonald JS, et al. p53 immunoreactivity and single-strand conformational polymorphism analysis often fail to predict p53 mutational status. Mod Pathol 1999; 12: 54–60.
El-Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM, et al. WAF1, a potential mediator of p53 tumor suppression. Cell 1993; 75: 817–825.
Harper JW, Adami GR, Wei N, Keyomarsi K, Elledge SJ . The p21 cdk-interacting protein Cip1 is a potent inhibitor of G1 cyclin-dependent kinases. Cell 1993; 75: 805–816.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kawauchi, S., Fukuda, T., Oda, Y. et al. Prognostic Significance of Apoptosis in Synovial Sarcoma: Correlation with Clinicopathologic Parameters, Cell Proliferative Activity, and Expression of Apoptosis-Related Proteins. Mod Pathol 13, 755–765 (2000). https://doi.org/10.1038/modpathol.3880131
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3880131
Keywords
This article is cited by
-
Synovial Sarcoma: A Complex Disease with Multifaceted Signaling and Epigenetic Landscapes
Current Oncology Reports (2020)
-
Tissue microarray profiling of primary and xenotransplanted synovial sarcomas demonstrates the immunophenotypic similarities existing between SYT-SSX fusion gene confirmed, biphasic, and monophasic fibrous variants
Virchows Archiv (2006)
-
Diagnosis of primary esophageal synovial sarcoma by demonstration of t(X;18) translocation: a case report
Virchows Archiv (2006)