Abstract
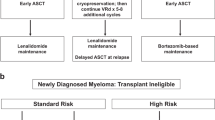
New systems have emerged for diagnosis, staging and response assessment in multiple myeloma (MM). The diagnostic and response criteria recommended are primarily derived from the International Myeloma Working Group, with certain updates and clarifications. The International Staging System is the current standard for staging of myeloma. A new risk stratification model is provided to specifically define high-risk patients who may benefit from novel therapeutic strategies. This paper provides the current criteria for diagnosis, staging, risk stratification and response assessment of MM.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kyle RA, Gertz MA, Witzig TE, Lust JA, Lacy MQ, Dispenzieri A et al. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clinic Proc 2003; 78: 21–33.
Snozek CLH, Katzmann JA, Kyle RA, Dispenzieri A, Larson D, Therneau TM et al. Prognostic value of the serum free light chain ratio in newly diagnosed myeloma: proposed incorporation into the International Staging System. Leukemia 2008; 22: 1933–1937.
Rajkumar SV, Kyle RA, Therneau TM, Melton III LJ, Bradwell AR, Clark RJ et al. Serum free light chain ratio is an independent risk factor for progression in monoclonal gammopathy of undetermined significance (MGUS). Blood 2005; 106: 812–817.
Dispenzieri A, Kyle RA, Katzmann JA, Therneau TM, Larson D, Benson J et al. Immunoglobulin free light chain ratio is an independent risk factor for progression of smoldering (asymptomatic) multiple myeloma. Blood 2008; 111: 785–789.
Dingli D, Kyle RA, Rajkumar SV, Nowakowski GS, Larson DR, Bida JP et al. Immunoglobulin free light chains and solitary plasmacytoma of bone. Blood 2006; 108: 1979–1983.
The International Myeloma Working Group. Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol 2003; 121: 749–757.
Rajkumar SV, Kyle RA . Multiple myeloma: diagnosis and treatment.[see comment]. Mayo Clinic Proc 2005; 80: 1371–1382.
Kyle RA, Therneau TM, Rajkumar SV, Remstein ED, Offord JR, Larson DR et al. Long-term follow-up of IgM monoclonal gammopathy of undetermined significance. Blood 2003; 102: 3759–3764.
Gobbi PG, Baldini L, Broglia C, Goldaniga M, Comelli M, Morel P et al. Prognostic validation of the international classification of immunoglobulin M gammopathies: a survival advantage for patients with immunoglobulin M monoclonal gammopathy of undetermined significance? Clin Cancer Res 2005; 11: 1786–1790.
Kyle RA, Therneau TM, Rajkumar SV, Offord JR, Larson DR, Plevak MF et al. Long-term follow-up of IgM monoclonal gammopathy of undetermined significance. Semin Oncol 2003; 30: 169–171.
Baldini L, Goldaniga M, Guffanti A, Broglia C, Cortelazzo S, Rossi A et al. Immunoglobulin M monoclonal gammopathies of undetermined significance and indolent Waldenstrom's macroglobulinemia recognize the same determinants of evolution into symptomatic lymphoid disorders: proposal for a common prognostic scoring system. J Clin Oncol 2005; 23: 4662–4668.
Owen RG, Treon SP, Al-Katib A, Fonseca R, Greipp PR, McMaster ML et al. Clinicopathological definition of Waldenstrom's macroglobulinemia: consensus panel recommendations from the Second International Workshop on Waldenstrom's Macroglobulinemia. Semin Oncol 2003; 30: 110–115.
Dimopoulos MA, Moulopoulos LA, Maniatis A, Alexanian R . Solitary plasmacytoma of bone and asymptomatic multiple myeloma. Blood 2000; 96: 2037–2044.
Dimopoulos MA, Kiamouris C, Moulopoulos LA . Solitary plasmacytoma of bone and extramedullary plasmacytoma. Hematol Oncol Clin North Am 1999; 13: 1249–1257.
Rajkumar SV, Dispenzieri A, Kyle RA . Monoclonal gammopathy of undetermined significance, Waldenstrom macroglobulinemia, AL amyloidosis, and related plasma cell disorders: diagnosis and treatment. Mayo Clin Proc 2006; 81: 693–703.
Dispenzieri A, Kyle RA, Lacy MQ, Rajkumar SV, Therneau TM, Larson DR et al. POEMS syndrome: definitions and long-term outcome. Blood 2003; 101: 2496–2506.
Durie BG, Salmon SE . A clinical staging system for multiple myeloma. Correlation of measured myeloma cell mass with presenting clinical features, response to treatment, and survival. Cancer 1975; 36: 842–854.
Bataille R, Durie BG, Grenier J . Serum beta2 microglobulin and survival duration in multiple myeloma: a simple reliable marker for staging. Br J Haematol 1983; 55: 439–447.
Greipp PR, San Miguel JF, Durie BG, Crowley JJ, Barlogie B, Blade J et al. International Staging System for multiple myeloma. J Clin Oncol 2005; 23: 3412–3420.
Tricot G, Barlogie B, Jagannath S, Bracy D, Mattox S, Vesole DH et al. Poor prognosis in multiple myeloma is associated only with partial or complete deletions of chromosome 13 or abnormalities involving 11q and not with other karyotype abnormalities. Blood 1995; 86: 4250–4256.
Smadja NV, Bastard C, Brigaudeau C, Leroux D, Fruchart C, Groupe Francais de Cytogenetique H . Hypodiploidy is a major prognostic factor in multiple myeloma. Blood 2001; 98: 2229–2238.
Fonseca R, Barlogie B, Bataille R, Bastard C, Bergsagel PL, Chesi M et al. Genetics and cytogenetics of multiple myeloma: a workshop report. Cancer Res 2004; 64: 1546–1558.
Fassas AB, Spencer T, Sawyer J, Zangari M, Lee CK, Anaissie E et al. Both hypodiploidy and deletion of chromosome 13 independently confer poor prognosis in multiple myeloma. Br J Haematol 2002; 118: 1041–1047.
Rajkumar SV, Kyle RA . Multiple myeloma: diagnosis and treatment. Mayo Clin Proc 2005; 80: 1371–1382.
Fonseca R, Blood E, Rue M, Harrington D, Oken MM, Kyle RA et al. Clinical and biologic implications of recurrent genomic aberrations in myeloma. Blood 2003; 101: 4569–4575.
Facon T, Avet-Loiseau H, Guillerm G, Moreau P, Genevieve F, Zandecki M et al. Chromosome 13 abnormalities identified by FISH analysis and serum beta2-microglobulin produce a powerful myeloma staging system for patients receiving high-dose therapy. Blood 2001; 97: 1566–1571.
Greipp PR, Lust JA, WM OF, Katzmann JA, Witzig TE, Kyle RA . Plasma cell labeling index and beta 2-microglobulin predict survival independent of thymidine kinase and C-reactive protein in multiple myeloma [see comments]. Blood 1993; 81: 3382–3387.
Blade J, Samson D, Reece D, Apperley J, Bjorkstrand B, Gahrton G et al. Criteria for evaluating disease response and progression in patients with multiple myeloma treated by high-dose therapy and haemopoietic stem cell transplantation. Myeloma Subcommittee of the EBMT. European Group for Blood and Marrow Transplant. Br J Haematol 1998; 102: 1115–1123.
Durie BGM, Harousseau J-L, Miguel JS, Blade J, Barlogie B, Anderson K et al. International uniform response criteria for multiple myeloma. Leukemia 2006; 20: 1467–1473.
Anderson KC, Kyle RA, Rajkumar SV, Stewart AK, Weber D, Richardson P . Clinically relevant end points and new drug approvals for myeloma. Leukemia 2008; 22: 231–239.
Acknowledgements
This study was supported by Grants CA62242 and CA107476 from the National Cancer Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kyle, R., Rajkumar, S. Criteria for diagnosis, staging, risk stratification and response assessment of multiple myeloma. Leukemia 23, 3–9 (2009). https://doi.org/10.1038/leu.2008.291
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2008.291
Keywords
This article is cited by
-
Circulating biosignatures in multiple myeloma and their role in multidrug resistance
Molecular Cancer (2023)
-
Image quality in whole-body MRI using the MY-RADS protocol in a prospective multi-centre multiple myeloma study
Insights into Imaging (2023)
-
Radiomics analysis of bone marrow biopsy locations in [18F]FDG PET/CT images for measurable residual disease assessment in multiple myeloma
Physical and Engineering Sciences in Medicine (2023)
-
Microfluidic isolation of aptamers with affinity towards multiple myeloma monoclonal immunoglobulins
Biomedical Microdevices (2023)
-
Effectiveness of biosimilar pegfilgrastim in patients with multiple myeloma after high-dose melphalan and autologous stem cell transplantation
Annals of Hematology (2023)