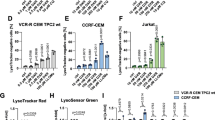
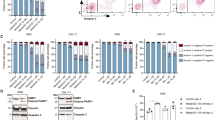
Abstract
Mutation of Bcr-Abl is an important mechanism by which chronic myelogenous leukemia (CML) cells become resistant to Gleevec. The T315I mutation is clinically significant since CML cells harboring this mutation are insensitive to Gleevec and other Bcr-Abl-targeted drugs. Identification of new agents capable of effectively killing CML cells with T315I mutation would have important therapeutic implications in Gleevec-resistant CML. Here, we showed that β-phenylethyl isothiocyanate (PEITC), a natural compound found in vegetables, is effective in killing CML cells expressing T315I BCR-ABL. Treatment of leukemia cell lines harboring wild-type or mutant Bcr-Abl with 10 μM PEITC resulted in an elevated ROS stress and a redox-mediated degradation of the BCR-ABL protein, leading to massive death of the leukemia cells. Antioxidant NAC attenuated the PEITC-induced oxidative stress in CML cells and prevented the degradation of BCR-ABL, caspase-3 activation and cell death. We further showed that the ROS-induced degradation of BCR-ABL was mediated partially by caspase-3 and the proteasome pathway. The ability of PEITC to effectively kill T315I-positive CML cells was further confirmed using primary leukemia cells isolated from CML patients. Our results suggest that PEITC is a promising compound capable of killing Gleevec-resistant CML cells through a ROS-mediated mechanism and warrants further investigations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Faderl S, Talpaz M, Estrov Z, O'Brien S, Kurzrock R, Kantarjian HM . The biology of chronic myeloid leukemia. N Engl J Med 1999; 341: 164–172.
Ren R . Mechanisms of BCR-ABL in the pathogenesis of chronic myelogenous leukaemia. Nat Rev Cancer 2005; 5: 172–183.
Melo JV, Barnes DJ . Chronic myeloid leukaemia as a model of disease evolution in human cancer. Nat Rev Cancer 2007; 7: 441–453.
Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM et al. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 2001; 344: 1031–1037.
Kantarjian H, Sawyers C, Hochhaus A, Guilhot F, Schiffer C, Gambacorti-Passerini C et al. Hematologic and cytogenetic responses to imatinib mesylate in chronic myelogenous leukemia. N Engl J Med 2002; 346: 645–652.
Gorre ME, Mohammed M, Ellwood K, Hsu N, Paquette R, Rao PN et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 2001; 293: 876–880.
Shah NP, Nicoll JM, Nagar B, Gorre ME, Paquette RL, Kuriyan J et al. Multiple BCR-ABL kinase domain mutations confer polyclonal resistance to the tyrosine kinase inhibitor imatinib (STI571) in chronic phase and blast crisis chronic myeloid leukemia. Cancer Cell 2002; 2: 117–125.
Branford S, Rudzki Z, Walsh S, Parkinson I, Grigg A, Szer J et al. Detection of BCR-ABL mutations in patients with CML treated with imatinib is virtually always accompanied by clinical resistance, and mutations in the ATP phosphate-binding loop (P-loop) are associated with a poor prognosis. Blood 2003; 102: 276–283.
Yamamoto M, Kurosu T, Kakihana K, Mizuchi D, Miura O . The two major imatinib resistance mutations E255K and T315I enhance the activity of BCR/ABL fusion kinase. Biochem Biophys Res Commun 2004; 319: 1272–1275.
Talpaz M, Shah NP, Kantarjian H, Donato N, Nicoll J, Paquette R et al. Dasatinib in imatinib-resistant Philadelphia chromosome-positive leukemias. N Engl J Med 2006; 354: 2531–2541.
Cortes J, Rousselot P, Kim DW, Ritchie E, Hamerschlak N, Coutre S et al. Dasatinib induces complete hematologic and cytogenetic responses in patients with imatinib-resistant or -intolerant chronic myeloid leukemia in blast crisis. Blood 2007; 109: 3207–3213.
Bradeen HA, Eide CA, O'Hare T, Johnson KJ, Willis SG, Lee FY et al. Comparison of imatinib mesylate, dasatinib (BMS-354825), and nilotinib (AMN107) in an N-ethyl-N-nitrosourea (ENU)-based mutagenesis screen: high efficacy of drug combinations. Blood 2006; 108: 2332–2338.
Druker BJ . Circumventing resistance to kinase-inhibitor therapy. N Engl J Med 2006; 354: 2594–2596.
Quintas-Cardama A, Kantarjian H, Cortes J . Flying under the radar: the new wave of BCR-ABL inhibitors. Nat Rev Drug Discov 2007; 6: 834–848.
Sattler M, Verma S, Shrikhande G, Byrne CH, Pride YB, Winkler T et al. The BCR/ABL tyrosine kinase induces production of reactive oxygen species in hematopoietic cells. J Biol Chem 2000; 275: 24273–24278.
Kim JH, Chu SC, Gramlich JL, Pride YB, Babendreier E, Chauhan D et al. Activation of the PI3K/mTOR pathway by BCR-ABL contributes to increased production of reactive oxygen species. Blood 2005; 105: 1717–1723.
Koptyra M, Falinski R, Nowicki MO, Stoklosa T, Majsterek I, Nieborowska-Skorska M et al. T BCR/ABL kinase induces self-mutagenesis via reactive oxygen species to encode imatinib resistance. Blood 2006; 108: 319–327.
Trachootham D, Zhou Y, Zhang H, Demizu Y, Chen Z, Pelicano H et al. Selective killing of oncogenically transformed cells through a ROS-mediated mechanism by beta-phenylethyl isothiocyanate. Cancer Cell 2006; 10: 241–252.
Zhang Q, Tsukahara F, Maru Y . N-acetyl-cysteine enhances growth in BCR-ABL-transformed cells. Cancer Sci 2005; 96: 240–244.
Konig H, Hartel N, Schultheis B, Schatz M, Lorentz C, Melo JV et al. Enhanced Bcr-Abl-specific antileukemic activity of arsenic trioxide (Trisenox) through glutathione-depletion in imatinib-resistant cells. Haematologica 2007; 92: 838–841.
Beran M, Pisa P, O'Brien S, Kurzrock R, Siciliano M, Cork A et al. Biological properties and growth in SCID mice of a new myelogenous leukemia cell line (KBM-5) derived from chronic myelogenous leukemia cells in the blastic phase. Cancer Res 1993; 53: 3603–3610.
Wetzler M, Talpaz M, Van Etten RA, Hirsh-Ginsberg C, Beran M, Kurzrock R . Subcellular localization of Bcr, Abl, and Bcr-Abl proteins in normal and leukemic cells and correlation of expression with myeloid differentiation. J Clin Invest 1993; 92: 1925–1939.
Ricci C, Scappini B, Divoky V, Gatto S, Onida F, Verstovsek S et al. Mutation in the ATP-binding pocket of the ABL kinase domain in an STI571-resistant BCR/ABL-positive cell line. Cancer Res 2002; 62: 5995–5998.
Deng M, Daley GQ . Expression of interferon consensus sequence binding protein induces potent immunity against BCR/ABL-induced leukemia. Blood 2001; 97: 3491–3497.
Shah NP, Tran C, Lee FY, Chen P, Norris D, Sawyers CL . Overriding imatinib resistance with a novel ABL kinase inhibitor. Science 2004; 305: 399–401.
Samanta AK, Lin H, Sun T, Kantarjian H, Arlinghaus RB . Janus kinase 2: a critical target in chronic myelogenous leukemia. Cancer Res 2006; 66: 6468–6472.
Zhou Y, Achanta G, Pelicano H, Gandhi V, Plunkett W, Huang P . Action of (E)-2′-deoxy-2′-(fluoromethylene)cytidine on DNA metabolism: incorporation, excision, and cellular response. Mol Pharmacol 2002; 61: 222–229.
Zhou Y, Hileman EO, Plunkett W, Keating MJ, Huang P . Free radical stress in chronic lymphocytic leukemia cells and its role in cellular sensitivity to ROS-generating anticancer agents. Blood 2003; 101: 4098–4104.
Xu RH, Pelicano H, Zhang H, Giles FJ, Keating MJ, Huang P . Synergistic effect of targeting mTOR by rapamycin and depleting ATP by inhibition of glycolysis in lymphoma and leukemia cells. Leukemia 2005; 19: 2153–2158.
Liebes L, Conaway CC, Hochster H, Mendoza S, Hecht SS, Crowell J et al. High-performance liquid chromatography-based determination of total isothiocyanate levels in human plasma: application to studies with 2-phenethyl isothiocyanate. Anal Biochem 2001; 291: 279–289.
Wu SJ, Ng LT, Lin CC . Effects of antioxidants and caspase-3 inhibitor on the phenylethyl isothiocyanate-induced apoptotic signaling pathways in human PLC/PRF/5 cells. Eur J Pharmacol 2005; 518: 96–106.
Bang JH, Han ES, Lim I, Lee CS . Differential response of MG132 cytotoxicity against small cell lung cancer cells to changes in cellular GSH contents. Biochem Pharmacol 2004; 68: 659–666.
Di Bacco AM, Cotter TG . p53 expression in K562 cells is associated with caspase-mediated cleavage of c-ABL and BCR-ABL protein kinases. Br J Haematol 2002; 117: 588–597.
Barila D, Rufini A, Condo I, Ventura N, Dorey K, Superti-Furga G et al. Caspase-dependent cleavage of c-Abl contributes to apoptosis. Mol Cell Biol 2003; 23: 2790–2799.
Machuy N, Rajalingam K, Rudel T . Requirement of caspase-mediated cleavage of c-Abl during stress-induced apoptosis. Cell Death Differ 2004; 11: 290–300.
Podar K, Raab MS, Tonon G, Sattler M, Barila D, Zhang J et al. Up-regulation of c-Jun inhibits proliferation and induces apoptosis via caspase-triggered c-Abl cleavage in human multiple myeloma. Cancer Res 2007; 67: 1680–1688.
Ling X, Wang Y, Dietrich MF, Andreeff M, Arlinghaus RB . Vaccination with leukemia cells expressing cell-surface-associated GM-CSF blocks leukemia induction in immunocompetent mice. Oncogene 2006; 25: 4483–4490.
Acknowledgements
This work was supported in part by Grants CA085563, CA100428, CA109041 and CA16672 from the National Institutes of Health. DT is a recipient of a scholarship from Anandamahidol Foundation under the royal patronage of His Majesty the King of Thailand.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H., Trachootham, D., Lu, W. et al. Effective killing of Gleevec-resistant CML cells with T315I mutation by a natural compound PEITC through redox-mediated mechanism. Leukemia 22, 1191–1199 (2008). https://doi.org/10.1038/leu.2008.74
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2008.74
Keywords
This article is cited by
-
Modulation of energy metabolism to overcome drug resistance in chronic myeloid leukemia cells through induction of autophagy
Cell Death Discovery (2022)
-
Oxidative resistance of leukemic stem cells and oxidative damage to hematopoietic stem cells under pro-oxidative therapy
Cell Death & Disease (2020)
-
Reactive oxygen species in haematopoiesis: leukaemic cells take a walk on the wild side
Journal of Experimental & Clinical Cancer Research (2018)
-
Dual effect of oxidative stress on leukemia cancer induction and treatment
Journal of Experimental & Clinical Cancer Research (2014)
-
No evident dose-response relationship between cellular ROS level and its cytotoxicity – a paradoxical issue in ROS-based cancer therapy
Scientific Reports (2014)