Abstract
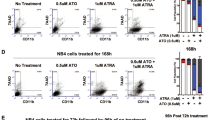
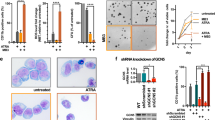
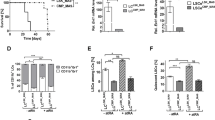
The pharmacological inhibitors of phosphoinositide 3-kinase (PI3K)/Akt pathway have been proposed in the treatment of leukemia based on their antiproliferative effects. However, several studies demonstrated the activation of PI3K in the nuclei of all-trans-retinoic acid (ATRA) – differentiated HL-60 cells, raising the possibility that PI3K/Akt-inhibitors may block antitumor properties of retinoids. The aim of the present study was to investigate the possible activation of nuclear Akt in ATRA-treated cells and to test the effects of Akt-inhibitors on ATRA-mediated differentiation. The Akt-activity was found to be increased in the nuclei and lysates of ATRA-differentiated HL-60 and NB4 cells. The down-modulation of the expression of Akt protein in HL-60 cells using siRNA reduces the CD11b expression in ATRA-treated cells. The treatment of both cell lines with the commercially available Akt inhibitors inhibited the growth of both control and ATRA-treated cells. Akt-inhibitors had no inhibitory effects on ATRA-mediated growth arrest and the expression of CD11b in HL-60 cells, but increased the percentage of control cells expressing CD11b. In contrast, the presence of Akt inhibitors reduced the expression of CD11b in ATRA-treated NB4 cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Datta SR, Brunet A, Greenberg ME . Cellular survival: a play in three Akts. Genes Dev 1999; 13: 2905–2927.
Hanada M, Feng J, Hemmings BA . Structure, regulation and function of PKB/AKT-a major therapeutic target. Biochim Biophys Acta 2004; 1697: 3–16.
Williams MR, Arthur JS, Balendran A, van der Kaay J, Poli V, Cohen P et al. The role of 3-phosphoinositide-dependent protein kinase 1 in activating AGC kinases defined in embryonic stem cells. Curr Biol 2000; 10: 439–448.
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM . Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 2005; 307: 1098–1101.
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 1997; 91: 231–241.
Cardone MH, Roy N, Stennicke HR, Salvesen GS, Franke TF, Stanbridge E et al. Regulation of cell death protease caspase-9 by phosphorylation. Science 1998; 282: 1318–1321.
Kim AH, Khursigara G, Sun X, Franke TF, Chao MV . Akt phosphorylates and negatively regulates apoptosis signal-regulating kinase 1. Mol Cell Biol 2001; 21: 893–901.
Biggs III WH, Meisenhelder J, Hunter T, Cavenee WK, Arden KC . Protein kinase B/Akt-mediated phosphorylation promotes nuclear exclusion of the winged helix transcription factor FKHR1. Proc Natl Acad Sci USA 1999; 96: 7421–7426.
Du K, Montminy M . CREB is a regulatory target for the protein kinase Akt/PKB. J Biol Chem 1998; 273: 32377–32379.
Shin I, Yakes FM, Rojo F, Shin NY, Bakin AV, Baselga J et al. PKB/Akt mediates cell-cycle progression by phosphorylation of p27(Kip1) at threonine 157 and modulation of its cellular localization. Nat Med 2002; 8: 1145–1152.
Nicholson KM, Anderson NG . The protein kinase B/Akt signalling pathway in human malignancy. Cell Signal 2002; 14: 381–395.
Neri LM, Marchisio M, Colamussi ML, Bertagnolo V . Monocytic differentiation of HL-60 cells is characterized by the nuclear translocation of phosphatidylinositol 3-kinase and of definite phosphatidylinositol-specific phospholipase C isoforms. Biochem Biophys Res Commun 1999; 259: 314–320.
Visnjic D, Crljen V, Curic J, Batinic D, Volinia S, Banfic H . The activation of nuclear phosphoinositide 3-kinase C2beta in all-trans-retinoic acid-differentiated HL-60 cells. FEBS Lett 2002; 529: 268–274.
Marchisio M, Bertagnolo V, Colamussi ML, Capitani S, Neri LM . Phosphatidylinositol 3-kinase in HL-60 nuclei is bound to the nuclear matrix and increases during granulocytic differentiation. Biochem Biophys Res Commun 1998; 253: 346–351.
Bertagnolo V, Brugnoli F, Marchisio M, Celeghini C, Carini C, Capitani S . Association of PI 3-K with tyrosine phosphorylated Vav is essential for its activity in neutrophil-like maturation of myeloid cells. Cell Signal 2004; 16: 423–433.
Bertagnolo V, Neri LM, Marchisio M, Mischiati C, Capitani S . Phosphoinositide 3-kinase activity is essential for all-trans-retinoic acid-induced granulocytic differentiation of HL-60 cells. Cancer Res 1999; 59: 542–546.
Bertolaso L, Gibellini D, Secchiero P, Previati M, Falgione D, Visani G et al. Accumulation of catalytically active PKC-zeta into the nucleus of HL-60 cell line plays a key role in the induction of granulocytic differentiation mediated by all-trans retinoic acid. Br J Haematol 1998; 100: 541–549.
Cappellini A, Tabellini G, Zweyer M, Bortul R, Tazzani PL, Billi AM et al. The phosphoinositide 3-kinase/Akt pathway regulates cell cycle progression of HL60 human leukemia cells through cytoplasmic relocalization of the cyclin-dependent kinase inhibitor p27Kip1 and control of cyclin D1 expression. Leukemia 2003; 17: 2157–2167.
Neri LM, Borgatti P, Tazzari PL, Bortul R, Cappellini A, Tabellini G et al. The phosphoinositide 3-kinase/AKT1 pathway involvement in drug and all-trans-retinoic acid resistance of leukemia cells. Mol Cancer Res 2003; 1: 234–246.
Martelli AM, Tazzari PL, Tabellini G, Bortul R, Billi AM, Manzoli L et al. A new selective AKT pharmacological inhibitor reduces resistance to chemotherapeutic drugs, TRAIL, all-trans-retinoic acid, and ionizing radiation of human leukemia cells. Leukemia 2003; 17: 1794–1805.
Tabellini G, Cappellini A, Tazzari PL, Fala F, Billi AM, Manzoli L et al. Phosphoinositide 3-kinase/Akt involvement in arsenic trioxide resistance of human leukemia cells. J Cell Physiol 2005; 202: 623–634.
Visnjic D, Batinic D, Banfic H . Arachidonic acid mediates interferon-gamma-induced sphingomyelin hydrolysis and monocytic marker expression in HL-60 cell line. Blood 1997; 89: 81–91.
Visnjic D, Curic J, Crljen V, Batinic D, Volinia S, Banfic H . Nuclear phosphoinositide 3-kinase C2beta activation during G2/M phase of the cell cycle in HL-60 cells. Biochim Biophys Acta 2003; 1631: 61–71.
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P et al. Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J 1996; 15: 6541–6551.
Rassidakis GZ, Feretzaki M, Atwell C, Grammatikakis I, Lin Q, Lai R et al. Inhibition of Akt increases p27Kip1 levels and induces cell cycle arrest in anaplastic large cell lymphoma. Blood 2005; 105: 827–829.
Tabellini G, Tazzari PL, Bortul R, Billi AM, Conte R, Manzoli L et al. Novel 2′-substituted, 3′-deoxy-phosphatidyl-myo-inositol analogues reduce drug resistance in human leukaemia cell lines with an activated phosphoinositide 3-kinase/Akt pathway. Br J Haematol 2004; 126: 574–582.
Yen A, Roberson MS, Varvayanis S, Lee AT . Retinoic acid induced mitogen-activated protein (MAP)/extracellular signal-regulated kinase (ERK) kinase-dependent MAP kinase activation needed to elicit HL-60 cell differentiation and growth arrest. Cancer Res 1998; 58: 3163–3172.
Lanotte M, Martin-Thouvenin V, Najman S, Balerini P, Valensi F, Berger R . NB4, a maturation inducible cell line with t(15;17) marker isolated from a human acute promyelocytic leukemia (M3). Blood 1991; 77: 1080–1086.
Zhu QS, Robinson LJ, Roginskaya V, Corey SJ . G-CSF-induced tyrosine phosphorylation of Gab2 is Lyn kinase dependent and associated with enhanced Akt and differentiative, not proliferative, responses. Blood 2004; 103: 3305–3312.
Fujita T, Azuma Y, Fukuyama R, Hattori Y, Yoshida C, Koida M et al. Runx2 induces osteoblast and chondrocyte differentiation and enhances their migration by coupling with PI3K-Akt signaling. J Cell Biol 2004; 166: 85–95.
Sugatani T, Hruska KA . Akt1/Akt2 and mammalian target of rapamycin/Bim play critical roles in osteoclast differentiation and survival, respectively, whereas Akt is dispensable for cell survival in isolated osteoclast precursors. J Biol Chem 2005; 280: 3583–3589.
Lopez-Carballo G, Moreno L, Masia S, Perez P, Barettino D . Activation of the phosphatidylinositol 3-kinase/Akt signaling pathway by retinoic acid is required for neural differentiation of SH-SY5Y human neuroblastoma cells. J Biol Chem 2002; 277: 25297–25304.
Vandromme M, Rochat A, Meier R, Carnac G, Besser D, Hemmings BA et al. Protein kinase B beta/Akt2 plays a specific role in muscle differentiation. J Biol Chem 2001; 276: 8173–8179.
Lal L, Li Y, Smith J, Sassano A, Uddin S, Parmar S et al. Activation of the p70 S6 kinase by all-trans-retinoic acid in acute promyelocytic leukemia cells. Blood 2005; 105: 1669–1677.
Kambhampati S, Verma A, Li Y, Parmar S, Sassano A, Platanias LC . Signalling pathways activated by all-trans-retinoic acid in acute promyelocytic leukemia cells. Leuk Lymphoma 2004; 45: 2175–2185.
Bertagnolo V, Brugnoli F, Mischiati C, Sereni A, Bavelloni A, Carini C et al. Vav promotes differentiation of human tumoral myeloid precursors. Exp Cell Res 2005; 306: 56–63.
Hu Y, Qiao L, Wang S, Rong SB, Meuillet EJ, Berggren M et al. 3-(Hydroxymethyl)-bearing phosphatidylinositol ether lipid analogues and carbonate surrogates block PI3-K, Akt, and cancer cell growth. J Med Chem 2000; 43: 3045–3051.
Kozikowski AP, Sun H, Brognard J, Dennis PA . Novel PI analogues selectively block activation of the pro-survival serine/threonine kinase Akt. J Am Chem Soc 2003; 125: 1144–1145.
Secchiero P, Gonelli A, Mirandola P, Melloni E, Zamai L, Celeghini C et al. Tumor necrosis factor-related apoptosis-inducing ligand induces monocytic maturation of leukemic and normal myeloid precursors through a caspase-dependent pathway. Blood 2002; 100: 2421–2429.
Acknowledgements
We thank Jane Leitch, Nick Leslie and Peter Downes for valuable help and advice in performing Akt kinase assays. This work was supported by the Ministry for Science of the Republic of Croatia (to DV and HB), and by the Fogarty International Research Collaboration Award (to HB).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matkovic, K., Brugnoli, F., Bertagnolo, V. et al. The role of the nuclear Akt activation and Akt inhibitors in all-trans-retinoic acid-differentiated HL-60 cells. Leukemia 20, 941–951 (2006). https://doi.org/10.1038/sj.leu.2404204
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404204
Keywords
This article is cited by
-
Transcriptional drug repositioning and cheminformatics approach for differentiation therapy of leukaemia cells
Scientific Reports (2021)
-
Salt-inducible kinase inhibition sensitizes human acute myeloid leukemia cells to all-trans retinoic acid-induced differentiation
International Journal of Hematology (2021)
-
Over-expression of a retinol dehydrogenase (SRP35/DHRS7C) in skeletal muscle activates mTORC2, enhances glucose metabolism and muscle performance
Scientific Reports (2018)
-
5-Aminoimidazole-4-carboxamide ribonucleoside-induced autophagy flux during differentiation of monocytic leukemia cells
Cell Death Discovery (2017)
-
DNMT3A R882 mutants interact with polycomb proteins to block haematopoietic stem and leukaemic cell differentiation
Nature Communications (2016)