Abstract
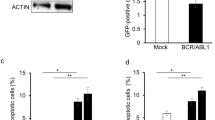
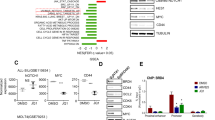
BCR-ABL fusion protein, a t(9;22) translocation product is indispensable for generation, maintenance and progression of chronic myeloid leukemia. RNA interference is an approach to silence gene at post-transcriptional level. We show that dsRNA targeted against the translocation region leads to more than 90% inhibition of BCR-ABL mRNA and protein expression levels using K562 as a model. Lack of BCR-ABL leads to cell cycle arrest in G1 phase as observed by decrease in cyclin D1 and increase in p21 and p27 cdk inhibitors mRNA. Apoptosis resistance imparted by BCR-ABL is lost in these cells in caspase-dependent or independent manner. Decrease in Bcl-XL is observed along with decrease in mitochondrial membrane integrity. Transient removal of BCR-ABL expression has a profound effect on proliferation and clonogenic capacity also confirmed by long-term silencing using lentiviral vectors. Interestingly, low level of BCR-ABL message leads to enhanced erythroid differentiation and reduced expression of megakaryocytic markers. Importantly, in six CML patient samples studied, silencing BCR-ABL in the lineage depleted enriched stem cell population leads to a decrease in colony-forming capacity. Thus, long-term silencing of BCR-ABL might prove to be a promising alternative approach in CML patients especially for those who do not respond to any other drug treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rowley J . Nonrandom chromosomal abnormalities in hematologic disorders of man. PNAS 1975; 72: 152–156.
Fialkow PJ, Jacobson RJ, Papayannopoulou T . Chronic myelocytic leukemia. Clonal origin in a stem cell common to the granulocyte, erythrocyte, platelet and monocyte/macrophage. Am J Med 1977; 63: 125.
Cortez D, Reuther G, Pendergast AM . The BCR-ABL tyrosine kinase activates mitogenic signaling pathways and stimulates G1-to-S phase transition in hematopoietic cells. Oncogene 1997; 15: 2333–2342.
Jena N, Deng M, Sicinska E, Sicinski P, Daley GQ . Critical role for cyclin D2 in BCR/ABL-induced proliferation of hematopoietic cells. Cancer Research 2002; 62: 535–541.
Gesbert F, Sellers WR, Signoretti S, Loda M, Griffin JD . BCR/ABL regulates expression of the cyclin-dependent kinase inhibitor p27Kip1 through the phosphatidylinositol 3-kinase/AKT pathway. J Biol Chem 2000; 275: 39223–39230.
Nimmanapalli R, Fuino L, Bali P, Gasparetto M, Glozak M, Tao J et al. Histone deacetylase inhibitor LAQ824 both lowers expression and promotes proteasomal degradation of BCR-ABL and induces apoptosis of Imatinib mesylate-sensitive or -refractory chronic myelogenous leukemia-blast crisis cells. Cancer Res 2003; 63: 5126–5135.
Shet AS, Jahagirdar BN, Verfaillie CM . Chronic myelogenous leukemia: mechanisms underlying disease progression. Leukemia 2002; 16: 1402–1411.
Dubrez L, Eymin B, Sordet O, Droin N, Turhan AG, Solary E . BCR-ABL delays apoptosis upstream of procaspase-3 activation. Blood 1998; 91: 2415–2422.
Amarante-Mendes GP, Kim CN, Liu L, Huang Y, Perkins CL, Green DR et al. BCR-ABL exerts its antiapoptotic effect against diverse apoptotic stimuli through blockage of mitochondrial release of cytochrome C and activation of caspase-3. Blood 1998; 91: 1700–1705.
Perkins C, Kim C, Fang G, Bhalla K . Arsenic induces apoptosis of multidrug-resistant human myeloid leukemia cells that express BCR-ABL or overexpress MDR, MRP, Bcl-2, or Bcl-xL. Blood 2000; 95: 1014–1022.
Yu C, Rahmani M, Almenara J, Subler M, Krystal G, Conrad D et al. Histone deactylase inhibitors promote STI571-mediated apoptosis in STI571-sensitive and -resistant BCR/ABL+ human myeloid leukemia cells. Cancer Res 2003; 63: 2118–2126.
Yu C, Rahmani M, Conrad D, Subler M, Dent P, Grant S . The proteasome inhibitor bortezomib interacts synergistically with histone deacetylase inhibitors to induce apoptosis in BCR-ABL+ cells sentitive and resistant to STI571. Blood 2003; 102: 3765–3774.
Okada M, Adachi S, Imai T, Watanabe K, Toyokuni S, Ueno M et al. A novel mechanism for imatinib mesylate-induced cell death of BCR-ABL+ human leukemic cells: caspase-independent, necrosis-like programmed cell death mediated by serine protease activity. Blood 2004; 103: 2299–2307.
Konopka J, Watanabe S, Witte O . An alteration of the human c-abl protein in K562 leukemia cells unmasks associated tyrosine kinase activity. Cell 1984; 37: 1035–1042.
Lugo T, Pendergast A, Muller A, Witte O . Tyrosine kinase activity and transformation potency of bcr-abl oncogene products. Science 1990; 247: 1079–1082.
Wong S, Witte O . The BCR-ABL story: bench to bedside and back. Annu Rev Immunol 2004; 22: 247–306.
Bueno-da-Silva AEB, Brumatti G, Russo FO, Green DR, Amarante-Mendes GP . BCR-ABL-mediated resistance to apoptosis is independent of constant tyrosine-kinase activity. Cell Death Differ 2003; 10: 592–598.
Graham SM, Jorgensen HG, Allan E, Pearson C, Alcorn MJ, Richmond L et al. Primitive, quiescent, Philadelphia-positive stem cells from patients with chronic myeloid leukemia are insensitive to STI571 in vitro. Blood 2002; 99: 319–325.
Donato NJ, Wu JY, Stapley J, Gallick G, Lin H, Arlinghaus R et al. BCR-ABL independence and LYN kinase overexpression in chronic myelogenous leukemia cells selected for resistance to STI571. Blood 2003; 101: 690–698.
Wohlbold L, van der Kuip H, Miething C, Vornlocher H-P, Knabbe C, Duyster J et al. Inhibition of bcr-abl gene expression by small interfering RNA sensitizes for imatinib mesylate (STI571). Blood 2003; 102: 2236–2239.
Scherr M, Battmer K, Winkler T, Heidenreich O, Ganser A, Eder M . Specific inhibition of bcr-abl gene expression by small interfering RNA. Blood 2002; 101: 1566–1569.
Wilda M, Fuchs U, Woessmann W, Borkhardt A . Killing of leukemic cells with a BCR/ABL fusion gene by RNA interference (RNAi). Oncogene 2002; 21: 5716–5724.
Li M-J, McMahon R, Snyder D, Yee J-Y, Rossi J . Specific killing of Ph+ chronic myeloid leukemia cells by a lentiviral vector-delivered anti-bcr/abl small hairpin RNA. Oligonucleotides 2003; 13: 401–409.
Scherr M, Battmer K, Schultheis B, Ganser A, Eder M . Stable RNA interference (RNAi) as an option for anti-bcr-abl therapy. Gene Therapy 2005; 12: 12–21.
Demaison C, Parsley K, Brouns G, Scherr M, Battmer K, Kinnon C et al. High-level transduction and gene expression in hematopoietic repopulating cells using a human immunodeficiency [correction of imunodeficiency] virus type 1-based lentiviral vector containing an internal spleen focus forming virus promoter. Hum Gene Ther 2002; 13: 803–813.
Richmond LJAM, Pearson C, Cameron G, Thomas T, Eaves CJ, Eaves AC et al. CML leukapheresis products can be enriched for CD34+ cells and simultaneously depleted of CD15+ cells using a simple Ab cocktail. Cytotherapy 2002; 4: 407–413.
Kawano T, Horiguchi-Yamada J, Saito S, Iwase S, Furukawa Y, Kano Y et al. Ectopic cyclin D1 expression blocks STI571-induced erythroid differentiation of K562 cells. Leuk Res 2004; 28: 623–629.
Elmaagacli AH, Beelen DW, Opalka B, Seeber S, Schaefer UW . The amount of BCR-ABL fusion transcripts detected by the real-time quantitative polymerase chain reaction method in patients with Philadelphia chromosome positive chronic myeloid leukemia correlates with the disease stage. Ann Hematol 2000; 79: 424–431.
Keeshan K, Mills KI, Cotter TG, McKenna SL . Elevated BCR-ABL expression levels are sufficient for a haemetopoietic cell line to acquire a drug-resistance phenotype. Leukemia 2001; 15: 1823–1833.
Moravcova J, Zmekova V, Klamova H, Voglova J, Faber E, Michalova K et al. Differences and similarities in kinetics of BCR-ABL transcript levels in CML patients treated with Imatinib mesylate for chronic or accelerated disease phase. Leukemia Res 2004; 28: 415–419.
Bedi AZB, Collector MI, Barber JP, Zicha MS, Sharkis SJ, Jones RJ . BCR-ABL gene rearrangement and expression of primitive hematopoietic progenitors in chronic myeloid leukemia. Blood 1993; 81: 2892–2902.
Acknowledgements
We thank the FACS facility at CR-UK, London for their excellent support. We also thank Dr Anne Galy and Daniel Compagno for providing the self-inactivating lentiviral and pH1 shuttle vectors. JR would like to thank Rajani Kanth Vangala and family for their continued support and encouragement.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by funding from Cancer Research UK
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu).
Supplementary information
Rights and permissions
About this article
Cite this article
Rangatia, J., Bonnet, D. Transient or long-term silencing of BCR-ABL alone induces cell cycle and proliferation arrest, apoptosis and differentiation. Leukemia 20, 68–76 (2006). https://doi.org/10.1038/sj.leu.2403999
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403999
Keywords
This article is cited by
-
BCR-ABL induces tyrosine phosphorylation of YAP leading to expression of Survivin and Cyclin D1 in chronic myeloid leukemia cells
International Journal of Hematology (2019)
-
Curcumin targets multiple enzymes involved in the ROS metabolic pathway to suppress tumor cell growth
Scientific Reports (2018)
-
Progress in RNAi-mediated Molecular Therapy of Acute and Chronic Myeloid Leukemia
Molecular Therapy - Nucleic Acids (2015)
-
Cleavage of BCR–ABL transcripts at the T315I point mutation by DNAzyme promotes apoptotic cell death in imatinib-resistant BCR–ABL leukemic cells
Leukemia (2013)
-
The Blk pathway functions as a tumor suppressor in chronic myeloid leukemia stem cells
Nature Genetics (2012)