Abstract
Ischemia, absence or loss of blood flow in organs always presents as a dual phenomenon: tissue oxygen deficit and CO2 excess (hypercapnia). Commonly hypoxic cell culture models kept CO2 at normal nonischemic values. We report a study of apoptosis in an in vitro model of renal hypoxia that mimics in vivo tissue gas atmosphere composition determined during experimental ischemia in rat kidney (low O2 plus high CO2). Renal tubular LLC-PK1 cell were transiently exposed to hypoxia, to hypercapnia or to both conditions (simulated ischemia). Exposure to simulated ischemic atmosphere, but not to low O2 or high CO2 alone, induced cell apoptosis in vitro. This suggests that ischemia-induced apoptosis in vivo would be dependent on the natural, joint action of hypoxia and hypercapnia. This should be taken into account in cell culture studies that would like to mimic in vivo ischemic conditions.
Similar content being viewed by others
Main
Apoptosis or programmed cell death may play a significant role in the pathogenesis of relevant ischemic diseases.1 This finding has been considered to be of great importance in pathology, including ischemic acute renal failure (ARF) due to tubular dysfunction;2, 3 partly, because, instead of rapid ischemic cell death by necrosis, attenuation of delayed cell death by apoptosis is potentially feasible, thus suggesting new approaches to therapy.4, 5
In vitro cell culture models of ischemic ARF have become a basic tool to gain an insight into the mechanisms triggering and regulating tubular cell apoptosis.3, 6 Overall, in vitro models mimic some of the features of tissue ischemia that may be linked to the regulation of cellular pathways toward apoptosis, such as ATP depletion.6 However, as in vivo tissues cannot be reproduced precisely, the results obtained should be transferred with caution to real-life physiological situations. Also, in in vitro models, one runs the risk of dismissing factors of physiological significance as regulatory stimuli influencing apoptosis. This is the case in the regulation of CO2.
When blood supply to an organ is blocked or impaired, leading to tissue hypoxia, low O2 is always accompanied by CO2 excess (hypercapnia).7 CO2 values increase as a result of bicarbonate buffering of anaerobic acid generation, coupled with CO2 retention due to impaired perfusion.7, 8 Hypercapnia influences a number of cell features that have been closely linked to the apoptotic process, such as mitochondrial metabolism and intracellular pH.9, 10 However, until now, cell culture models of renal ischemia/reperfusion (I/R) injury have not considered the possible dependence of apoptosis on ischemic hypercapnia. Moreover, models of renal cell hypoxia/reperfusion injury submit cells to hypoxia or chemical anoxia, but unlike physiological ischemia, in general retain, CO2 concentrations at normal nonischemic values during the cultured simulated ischemic period.11, 12 This implies that metabolically produced CO2 is being continuously washed (open bicarbonate/CO2 system), thus avoiding CO2 accumulation and its potential action.
In the present study, we hypothesized that, during in vitro cell culture models of ischemia, not only hypoxia but also high CO2 could influence cell apoptosis. We show here, using a renal tubular epithelial LLC-PK1 cell culture model of I/R, that a step toward well-established ischemic physiological conditions as represented to superimpose hypercapnia to hypoxic exposition, induces the appearance of apoptotic cell death features. This suggests that tissue CO2 levels may play a significant role in determining the mechanism of cell death associated to hypoxia.
Material and methods
In Vivo Study
Animals and protocol
The study on rats was conducted under the supervision of our institution's ethics commission and conformed to EU guidelines for the handling and care of laboratory animals. Briefly, 12 male Wistar rats weighing 250–300 g (Ifa Credo, Spain), six controls and six submitted to 30 min of unilateral left kidney ischemia were anesthetized with sodium pentobarbital (50 mg/kg). Polyethylene cannula (SPE-50, Clay Adams, Sparks, MD, USA) were inserted through the left carotid artery into the aorta for blood sampling. From arterial blood samples (0.15 ml) pre-ischemic pH, gas composition and plasma bicarbonate were determined (BMS3 MK2 Blood Microsystem and EML100 Electrolyte Metabolite Laboratory, Radiometer, Denmark). To induce 30 min kidney ischemia, laparotomy was performed and the left renal pedicle was dissected and occluded with a non-traumatic microvascular clamp.
Microelectrode pH measurement and pCO2 calculation
Intrarenal pH was continuously measured, during the ischemic period, as previously described.13 Briefly, miniaturized ISEs probes were inserted into the renal cortex after anteperitoneal exposure of the kidney. The monitoring assembly used, based on Microsystems Technology, was provided by the Centro Nacional de Microelectrónica (CNM) institute. Electrodes were calibrated with saline solutions and precision buffers, and the time responses of these electrodes were under 5 s in all circumstances. Cortical pCO2 values were calculated from tissue pH measurements, assuming constant ischemic tissue bicarbonate during complete renal circulatory arrest, corresponding to preischemic arterial values. This is a common assumption in acid–base splanchnic physiology, and, although certainly arguable,8 it should be considered that more precise CO2 determinations would be needless for the purpose of our study. CO2 values found were expressed as partial pressures in SI units (kPa). As gas mixtures used are also commonly expressed in %, it may be useful to recall that 1% dry gas is equivalent to 0.95 kPa (at 37°C, 1 atm, saturated with water vapor). So, kPa units are roughly equivalent to percent gas mixture.
In Vitro Study
Cell culture
Proximal tubular porcine LLC-PK1 cells (obtained from Professor Dr Bernhard Brüne) were cultured in Dulbecco modified Eagle medium (DMEM) with low glucose, supplemented with 2 mM glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin and 10% fetal calf serum (Biochrom, Germany). Cells were transferred two times each week. Cells were kept in a humidified atmosphere of 5% CO2 in air at 37°C.
Exposure to hypoxia/hypercapnia/simulated ischemia
Cultures were exposed transiently (7 h) either to low oxygen and/or high CO2 gas atmospheres, followed by a return (48 h) to normal culture conditions (5% CO2, air balance). Cell plates were incubated at 37°C in an open chamber, in which humidified-thermally equilibrated gas mixtures of controlled composition were continuously flushed. Six experimental groups were investigated:
Group I — Control group: 5% CO2 in air
Group II. — Hypoxic group (Hyp+5%CO2): 0.5% O2–5% CO2, N2 balance.
Group III. — Hypoxic+hypercapnic group (Hyp+18%CO2): 0.5% O2–18% CO2, N2 balance.
Group IV. — Hypoxic+hypercapnic group (Hyp+30%CO2): 0.5% O2–30%, N2 balance.
Group V. — Hypercapnic group (CO2 (18%)): 20% O2—18% CO2, N2 balance.
Group VI. — Hypercapnic group (CO2 (30%)): 20% O2–30% CO2, N2 balance.
Final setting gas values±1% within chamber was always attained within a 15 min time period.
Ternary (O2, CO2, N2) precision gas mixtures from gas cylinders were used (AL Air Liquide España S.A.). Gas was conveyed to the environmental chamber through a valve controlled flowmeter system (Control Module 208-01, Instrumentation Laboratory Inc., Barcelona, Spain) and a humidifier (at 37°C). Oxygen and carbon dioxide partial pressures in the environmental chamber gas were continuously monitored by means of a flowthrough CO2 electrode (Orion Research, Inc., Beverly, MA, USA) and a galvanic O2 sensor (Engineered System & Designs, Newark, DE, USA), calibrated and frequently checked during the experiment with humidified precision gases. The experimental procedure also included a preliminary study to test if gas equilibrium was also attained inside cell plates placed in the environmental chamber. Continuous O2 measurement within cell plates, using an oxygen microelectrode, (Microelectrodes, Londonderry, Inc., NH, USA), showed that hypoxic values below 1% O2, were reached in less than 10 min when the initial gas flow was equal to or higher than 0.7 l/min−1. Therefore, gas flow was initially set to this value for experimental procedures.
Assessment of Apoptotic Events
Caspase-3-like activity
Caspase 3-like activity was determined by measuring the proteolytic cleavage of the specific substrate N-acetyl-Asp-Glu-Val-Asp-7-amino-4-methylcoumarin (DEVD-AMC; Biomol, Plymouth Meeting, PA, USA). Cells were lysed and sonicated in an assay buffer (100 mM HEPES, pH 7.5, 10% sucrose, 0.1% (w/v) CHAPS, 1 mM PMSF, 1 mM dithiothreitol). Cell supernatants (250 μg protein) were incubated in an assay buffer with 50 μM Ac-DEVD-AMC. The AMC released was quantified for 2 h at 37°C by fluoro-spectrophotometry using 380-nm excitation and 450-nm emission.
Diphenylamine assay
DNA fragmentation was determined by a modification of the method reported by Ikeda et al.14 Briefly, cells were centrifuged, washed in PBS and harvested by centrifugation at 3000 r.p.m. for 5 min. Pellets were lysed by adding 400 μl of lysis buffer (10 mM Tris-HCl, 10 mM EDTA, pH 8.0) containing 0.5% Triton X-100. Samples were allowed to lyse for 45 min at 4°C prior to centrifugation (15 000 r.p.m., 20 min) to separate intact chromatin (pellet) from DNA fragments (supernatant). The supernatant was placed in a separate microfuge tube and both fraction volumes were equalled and then precipitated and homogenized in 400 μl of 1 N perchloric acid for 30 min at 4°C. Precipitates were sedimented at 15 000 r.p.m. for 20 min. The supernatant was discarded and pellets resuspended with 100 μl of 1 N perchloric acid and diphenylamine reagent.15 Samples were boiled for 5 min to hydrolyze DNA. The percentage of DNA fragmentation was defined as the ratio of the DNA content in the supernatant to the total DNA in the lysate.
DAPI staining
Nuclear changes such as chromatin condensation and fragmentation were analyzed by staining with DAPI, which stains nuclei (apoptotic or viable) blue under DAPI filter. Briefly, cells were fixed in 4% paraformaldehyde, washed with PBS, incubated with DAPI solution (0.2 μg/ml). for 5 min at room temperature. Cells were then rinsed twice in PBS, mounted and viewed by fluorescence microscopy.
TUNEL staining
Apoptosis was detected in situ by enzymatic labelling of DNA strand breaks using the Apotag Peroxidase In situ Apoptosis Detection Kit (Serologicals Corporation, CA, USA). Fixed cells were processed and incubated following the manufacturer's instructions. After that, samples were counterstained with hematoxylin, coverslipped and subsequent by visualized by light microscopy.
Western blot analysis
Cleavage of caspase 3 was detected in cell lysates by Western blot analysis. Following protein separation in 16% SDS-gel, proteins were transferred to nitrocellulose membranes, which were subsequently blocked with 5% nonfat dry milk in 0.06%. Tween-TBS for 1 h. Membranes were incubated with primary antibody (anti caspase-3 at 1:1000 dilution; Stressgen, Canada) overnight at 4°C. Membranes were washed five times with 0.06% Tween-TBS and then incubated for 1 h with a horseradish peroxidase conjugated anti-rabbit IgG antibody (dilution 1:5000) at room temperature. Membranes were washed five times with 0.06% Tween-TBS, followed by ECL-detection.
Statistical Analysis
Data are expressed as means±s.e.m. The means of different groups were compared using one-way ANOVA. Student–Newman–Keuls test was used to evaluate significant differences between groups. Significant differences were assumed when P<0.05.
Results
In Vivo pCO2
Figure 1 shows the evolution of cortical pCO2 values during 30 min ischemia.
Renal vascular occlusion induced an immediate rapid rise in tissue pCO2, reaching mean values over 15 kPa at 5 min of ischemia. A plateau phase, with a mean ischemic pCO2 of in the order of 30 kPa, was reached after approximately 15 min of ischemia.
Apoptotic Indicators
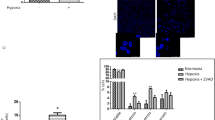
Figure 2 shows caspase-3 activity during the recovery period. Significant variations were only seen in samples exposed to ‘ischemic atmosphere’ (hypoxia and hypercapnia together), either corresponding to short (18% CO2) or long in vivo ischemia (30% CO2). The large increase in the activity corresponding to hypercapnia of 18% CO2 (Hyp+18%CO2) was note worthy. No changes were found in cultures submitted to low O2 or high CO2 exposition alone.
This enzymatic activity was concomitant with the protein profile as shown in the Western blot analysis of cleaved caspase 3 (Figure 3) corresponding to 17 kDa fragment. In the Hyp+18%CO2 and Hyp+30%CO2 groups, marked increases of cleaved caspase-3 expression were detected, but no increases were detected in the cultures exposed either to low O2 or to high CO2 independently.
Analysis of DNA fragmentation corroborated the previous results (Figure 4), showing that significant changes were restricted to groups with increases in caspase-3 activity.
With DAPI staining (Figure 5), viable cells that represent the large part of the total population showed homogeneous staining of their nuclei. In contrast, apoptotic cells showed irregular staining as a result of chromatin condensation and nuclear fragmentation (Figure 5b, c and e). This irregular staining was detected mainly in Hyp+18%CO2 (Figure 5b) and Hyp+30%CO2 (Figure 5c) groups.
Representative DAPI staining showing homogeneous staining of the nucleus in viable cells and irregular staining with small and highlighted bodies as a result of chromatin condensation and nuclear fragmentation of the following groups. Control (a), Hyp+18%CO2 (b), Hyp+30%CO2 (c), CO2 (18%) (d), CO2 (30%) (e). Magnification × 40.
In order to support DAPI staining and to discriminate better the apoptotic morphology, we also used TUNEL reaction (Figure 6) as one of the most commonly used techniques to detect apoptosis. With this technique, positive apoptotic cells showed a dark brown stain together with loss of the typical cell morphology. These cells were again detected mainly in Hyp+18%CO2 (Figure 6b) and Hyp+30%CO2 (Figure 6c) groups.
Discussion
Ischemia due to circulatory failure in organs has been defined as dual defects of O2 deficits and CO2 excesses, and it is likely that both factors impair tissue and organ function.7 Hypercapnia modifies different cell issues linked to the apoptotic process, such as cytosolic pH and osmolality.16 Taking these premises together, it could be expected that hypercapnia might play a shared role with hypoxia on ischemia-mediated cell death by apoptosis. However, the possible dependence of apoptosis on physiological hypercapnia in in vitro culture models of I/R injury has not been considered.
The current study reports as a new finding that, in a renal cell culture model of I/R, in which we reproduced tissue O2 and CO2 gas tensions found during experimental ischemia in vivo, the appearance of apoptotic signals is dependent not only on hypoxic but also on hypercapnic conditions.
In Vitro vs In Vivo ‘Ischemic’ Gas Atmosphere
Ischemic and critical renal oxygen tensions have been well established.17, 18, 19 Taking into account these studies, we used a 0.5% O2 gas atmosphere to keep the cell culture hypoxic below critical and in the range of oxygen pressures measured during acute renal arterial occlusion in experimental animals. We avoided the use of anoxia and, also, of commonly used gas mixtures over 1% O2 for hypoxic cell culture; the use of anoxic gas induce the expression of cell transcription factors different from physiological hypoxic conditions.20 By contrast, hypoxic O2 gas mixtures over 1% may not be suited to reproduce in vivo ischemia conditions, as lower cortical oxygen pressures have been measured during acute renal arterial occlusion in experimental animals.19
Contrary to other organs, intrarenal CO2 studies during ischemia are limited and discordant. To extend the in vivo frame of reference to set up in vitro simulated ischemic CO2 conditions, we determined intrarenal CO2 values during 30 min of experimental ischemia in rat kidney (Figure 1). This period of complete renal ischemia is based on previous studies that showed the presence of renal apoptotic signals.21
From the above, we decided to simulate ischemia in vitro by means of two different hypoxic–hypercapnic gas mixtures. The first mixture containing 18% CO2 represents values found during early ischemia (5 min). The second with 30% CO2 replicates mean stationary CO2 values found during prolonged ischemia (30 min). Both 5 and 30 min of arterial clamping in the rat kidney have been reported to induce the presence of apoptotic signals after 24 and 48 h of reperfusion.21
Apoptotic LLC-PK1 Cell Features
Caspase-3 activation seems to be a defining feature of apoptotic LLC-PK1 cell death.22, 23 We found here large caspase-3 activity increases after exposure to hypercapnic hypoxia. Changes were not found in cultures submitted either to hypoxia or to hypercapnia independently (Figure 2). Western blot analysis of cleaved caspase 3 confirmed enzymatic activity assays (Figure 3). Additionally, analysis of DNA fragmentation showed the same tendency (Figure 4). All these results were supported by morphological criteria of apoptosis, as cells showing nuclear apoptotic morphology revealed by DAPI staining (Figure 5) and by positive TUNEL staining (Figure 6). Although we were unable to find any apoptotic signal in cells exposed to low O2 alone, other similar hypoxic studies have shown apoptotic features in LLC-PK1 cells.12, 24, 25 Disagreement with our finding may be due to the different model used, either chemically induced ATP or hypoxic gas exposure in our model. Interestingly, the work from Meldrum et al26 supports our data. This model simulates tubular kidney ischemic conditions by immersing the monolayer cells in mineral oil. Then the oxygen is restricted but probably also CO2 washout could be prevented, generating hypercapnic hypoxia. Nevertheless, this work does not discern whether there is a specific CO2 effect on apoptosis.
Basis of CO2 Action
Unlike O2 deprivation, the hypercapnic influence on apoptosis has barely been considered. The question of which are the factors and the key steps in the apoptotic pathway that mediate the proapoptotic CO2 effect opens multiple approaches for further research. High CO2 influence on apoptosis could be indirect, mediated by factors such as intracellular hydrogen ion, or direct on cell metabolic pathways.
As an indirect mechanism, it should be mentioned that a lowering of intracellular pH-mediated CO2 increase may be of special significance in LLC-PK1 cell cultures, as it has been shown that in this cell line, intrinsic intracellular buffering (buffering power vs CO2 changes) is the lowest reported for any cell.27
It is important to point out that the CO2 acidifying effect differs essentially from extracellular acidification by the addition of exogenous acid, leading to different experimental models. As CO2 diffuses easily through cell membranes, its pH influence would be immediate in all intracellular compartments, whereas intracellular acidification through extracellular acid load entails the contribution of cell membrane acid–base equivalent exchangers.
As direct actions (not pH mediated) of hypercapnia on cell metabolism, it was found that changing the environmental CO2 concentration had a direct influence on kidney cell glucose metabolism and on mitochondrial ATPase activity in cortical renal tissue, whereas medullar structures contained cells that did not respond to CO2 changes.28, 29 Changes in glycolytic ATP synthesis and mitochondrial ATPase activity have been shown to influence renal cell apoptosis.30, 9
In summary, the essential new finding of this study is that CO2 levels play a significant role in determining apoptosis during in vitro ischemia. Therefore, not only hypoxia but also hypercapnia must be considered when in vitro studies are used to try to reproduce in vivo ischemia.
References
Thompson CB . Apoptosis in the pathogenesis and treatment of disease. Science 1995;267:1456–1462.
Edelstein CL, Ling H, Schrier RW . The nature of renal cell injury. Kidney Int 1997;51:1341–1351.
Ueda N, Kaushal GP, Shah SV . Apoptotic mechanisms in acute renal failure. Am J Med 2000;108:403–415.
Bonegio R, Lieberthal W . Role of apoptosis in the pathogenesis of acute renal failure. Curr Opin Nephrol Hypertens 2002;11:301–308.
Padanilam BJ . Cell death induced by acute renal injury: a perspective on the contributions of apoptosis and necrosis. Am J Physiol 2003;284:F608–F627.
Molitoris BA, Weingberg JM, Venkatachalam MA, et al. Acute renal failure. II. Experimental models of acute renal failure: imperfect but indispensable. Am J Physiol 2000;278:F1–F12.
Johnson BA, Weil MH . Redefining ischemia due to circulatory failure as dual defects of oxygen deficits and carbon dioxide excesses. Crit Care Med 1991;19:1432–1438.
Tønnesen TI . Biological basis for CO2 as detector of ischemia. Acta Anaesthesiol Scand 1997;41:659–669.
Michea L, Combs C, Andrews P, et al. Mitochondrial dysfunction is an early even in high-NaCl-induced apoptosis of IMCD3 cells. Am J Physiol 2002;282:F981–F990.
Liu D, Martino G, Thangaraju M, et al. Caspase-8-mediated intracellular acidification precedes mitochondrial dysfunction in somatostatin-induced apoptosis. J Biol Chem 2000;275:9244–9250.
Allen J, Winterford C, Axelsen . Effects of hypoxia on morphological and biochemical characteristics of renal epithelial cell and tubule cultures. Renal Failure 1992;14:453–460.
Hagar H, Ueda N, Shah SV . Endonuclease induced DNA damage and cell death in chemical hypoxic injury to LLC-PK1 cells. Kidney Int 1996;49:355–361.
Sola A, Palacios L, López-Martí J, et al. Multiparametric monitoring of ischemia-reperfusion in rat kidney: effect of ischemic preconditioning. Transplantation 2003;75:744–749.
Ikeda H, Suzuki Y, Suzuki M, et al. Apoptosis is a major mode of cell death caused by ischaemia and ischaemia/reperfusion injury to the rat intestinal epithelium. Gut 1998;42:530–537.
Burton K . A study of the conditions and mechanism of diphenylamine reaction for the colorimetric estimation of deoxyribonucleic acid. Biochem J 1956;62:315–323.
Segal MS, Beem E . Effect of pH, ionic charge, and osmolality on cytochrome c-mediated caspase-3 activity. Am J Physiol 2001;281:C1196–C1204.
Deetjen P . Normal and critical oxygen supply of the kidney. In: (eds) Oxygen Transport in Blood and Tissue, Lübbers D-W, Luft UC, Thews G and Witzleb E Georg Thieme Verlag: Stuttgart, 1968, pp 212–226.
Leichtweiis HP, Lübbers HW, Weiss CH . The oxygen supply of rat kidney, measurement of intrarenal pO2 . Pflügers Arch 1969;309:328–349.
Nelimarkka O, Niinikoski J . Oxygen and carbon dioxide tensions in the canine kidney during arterial occlusion and hemorrhagic hypotension. Surg Gynecol Obstet 1984;158:27–32.
Gassmann M, Wenger RH . HIF-1, a mediator of the molecular response to hypoxia. News Physiol Sci 1997;12:214–218.
Schumer M, Colombel MC, Sawczuc IS, et al. Morphologic, biological and molecular evidence of apoptosis during the reperfusion phase after brief periods of renal ischemia. Am J Pathol 1992;140:831–838.
Kaushal GP, Kaushal V, Hong X, et al. Role and regulation of activation of caspases in cisplatin-induced injury to renal tubular epithelial cells. Kidney Int 2001;60:1726–1736.
Zhan Y, van de Water B, Wang Y, et al. The role of caspase-3 and bcl-2 in chemically-induced apoptosis but not necrosis of renal epithelial cells. Oncogene 1999;18:6505–6512.
Wiegele G, Brandis M, Zimmerhackl LB . Apoptosis and necrosis during ischaemia in renal tubular cells (LLC-PK1 and MDCK). Nephrol Dial Transplant 1998;13:1158–1167.
Ueda N, Kaushal GP, Hong X, et al. Role of enhanced ceramide generation in DNA damage and cell death in chemical hypoxic injury to LLC-PK1 cells. Kidney Int 1998;54:399–406.
Meldrum KK, Meldrum DR, Hile K, et al. A novel model of ischemia in renal tubular cells which closely parallels in vivo injury. J Surg Res 2001;99:288–293.
Chaillet JR, Amsler K, Boron WF . Optical measurements of intracellular pH in single LLC-PK1 cells: demonstration of Cl-HCO3 . Proc Natl Acad Sci USA 1986;83:522–526.
Hastings AB, Fanestil DD . Effect of CO2 concentration on glucose-C14 metabolism by rabbit kidney cortex and medulla, in vitro. Biochem Z 1963;338:276–282.
Longmore WJ, Hastings AB, Harrison E . The effect of environmental pH and CO2 concentration on mitochondrial ATPase activity of rabbit kidney cortex and medulla. Hvalrådets Skr 1965;48:178–184.
Lieberthal W, Menza SA, Levine JS . Graded ATP depletion can cause necrosis or apoptosis of cultured mouse proximal tubular cells. Am J Physiol 1998;274:F315–F327.
Acknowledgements
We specially thank the Biomedical Applications Group from Centro Nacional de Microelectrònica, M. Ángeles Muñoz for her excellent technical support and Robyn Rycroft for grammatical assessment. This work was supported by EU Grant QLK6-CT-2000-00064, SAF 2000/3090-CE0057 and FISS 01/1691.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hotter, G., Palacios, L. & Sola, A. Low O2 and high CO2 in LLC-PK1 cells culture mimics renal ischemia-induced apoptosis. Lab Invest 84, 213–220 (2004). https://doi.org/10.1038/labinvest.3700026
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.3700026
Keywords
This article is cited by
-
A Review of the Role of the Partial Pressure of Carbon Dioxide in Mechanically Loaded Tissues: The Canary in the Cage Singing in Tune with the Pressure Ulcer Mantra
Annals of Biomedical Engineering (2015)
-
Ischemia’s Proliferative and Morphological Effects: Analyzing the Roles of Hypoxia, Hypercapnia, and Glucose
Cellular and Molecular Bioengineering (2010)
-
Investigating the Role of Ischemia vs. Elevated Hydrostatic Pressure Associated with Acute Obstructive Uropathy
Annals of Biomedical Engineering (2009)