Abstract
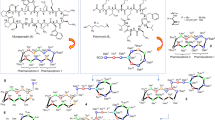
Further investigations towards novel glycopeptide/β-lactam heterodimers are reported. Employing a multivalent approach to drug discovery, vancomycin and cephalosporin synthons, 4, 2, 5 and 10, 18, 25 respectively, were chemically linked to yield heterodimer antibiotics. These novel compounds were designed to inhibit Grampositive bacterial cell wall biosynthesis by simultaneously targeting the principal cellular targets of both glycopeptides and β-lactams. The positional attachment of both the vancomycin and the cephalosporin central cores has been explored and the SAR is reported. This novel class of bifunctional antibiotics 28∼36 all displayed remarkable potency against a wide range of Gram-positive organisms, including methicillin-resistant Staphylococcus aureus (MRSA). A subset of compounds, 29, 31 and 35 demonstrated excellent bactericidal activity against MRSA (ATCC 33591) and 31 and 35 also exhibited superb in vivo efficacy in a mouse model of MRSA infection. As a result of this work compound 35 was selected as a clinical candidate, TD-1792.
Similar content being viewed by others
Article PDF
References
Baquero F . Gram-positive resistance: challenge for the development of new antibiotics. J Antimicrob Chemother 39 Suppl A: 1–6 ( 1997);
Neu HC . The crisis in antibiotic resistance. Science 257: 1064–1073 ( 1992)
Long DD, Aggen JB, Christensen BG, Judice JK, Hegde SS, Kaniga K, Krause KM, Linsell MS, Moran EJ, Pace JL . A multivalent approach to drug discovery for novel antibiotics. J Antibiot 61: 595–602 ( 2008)
Choi S-K . Synthetic Multivalent Molecules, Wiley & Sons, ISBN 0471563471 ( 2004)
Mammen M, Choi S-K, Whitesides GM . Polyvalent interactions in biological systems: implications for design and use of multivalent ligands and inhibitors. Angew Chem Int Ed 37: 2754–2794 ( 1998)
Wright D, Usher L . Multivalent binding in the design of bioactive compounds. Curr Org Chem 5: 1107–1131 ( 2001)
Griffin JH, Linsell MS, Nodwell MB, Chen Q, Pace JL, Quast KL, Krause KM, Farington L, Wu TX, Higgins DL, Jenkins TE, Christensen BG, Judice JK . Multivalent drug design. Synthesis and in vitro analysis of an array of vancomycin dimers. J Am Chem Soc 125: 6517–6531 ( 2003);
Nicolaou KC, Hughes R, Cho SY, Winssinger N, Labischinski H, Endermann R . 2001 Synthesis and biological evaluation of vancomycin dimers with potent activity against vancomycin-resistant bacteria: target-accelerated combinatorial synthesis. Chem Eur J 7: 3824–3843 ( 2001);
Nicolaou KC, Hughes R, Cho SY, Winssinger N, Smethurst C, Labischinski H, Endermann R . 2000 Target-accelerated combinatorial synthesis and discovery of highly potent antibiotics effective against vancomycin-resistant bacteria. Angew Chem Int Ed 39: 3823–3828 ( 2000);
Adamczyk M, Moore JA, Rege SD, Yu Z . Investigations into self-association of vancomycin covalent dimers using surface plasmon resonance technology. Bioorg Med Chem Lett 9: 2437–2440 ( 1999);
Rao J, Yan L, Lahiri J, Whitesides GM, Weis RM, Warren HS . Binding of a dimeric derivative of vancomycin to l-Lys-d-Ala-d-lactate in solution and at a surface. Chem Biol 6: 353–359 ( 1999);
Rao J, Yan L, Xu B, Whitesides GM . Using surface plasmon resonance to study the binding of vancomycin and its dimer to self-assembled monolayers presenting d-Ala-d-Ala. J Am Chem Soc 121: 2629–2630 ( 1999);
Rao J, Whitesides GM . Tight binding of a dimeric derivative of vancomycin with dimeric l-Lys-d-Ala-d-Ala. J Am Chem Soc 119: 10286–10290 ( 1997);
Sundram UN, Griffin JH, Nicas TI . Novel vancomycin dimers with activity against vancomycin-resistant enterococci. J Am Chem Soc 118: 13107–13190 ( 1996)
Rodrigues PCA, Scheuermann K, Stockmar C, Maier G, Fiebig HH, Unger C, Mulhaupt R, Kratz F . Synthesis and in vitro efficacy of acid-sensitive poly(ethylene glycol) paclitaxel conjugates. Biorg Med Chem Let 13: 355–360 ( 2003)
Portoghese PS . From models to molecules: Opioid receptor dimers, bivalent ligands, and selective opioid receptor probes. J Med Chem 44: 2259–2269 ( 2001)
Steinfeld T, Mammen M, Smith JAM, Wilson RD, Jasper JR . A novel multivalent ligand that bridges the allosteric and orthosteric binding sites of the M2 muscarinic receptor. Mol Pharm 72: 291–302 ( 2007)
Morphy R, Rankovic Z . Designed multiple ligands. J Med Chem 48: 6523–6543 ( 2005)
Malabarba A, Nicas TI, Thompson RC . Structural modifications of glycopeptide antibiotics. Med Res Rev 17: 69–137 ( 1997)
Sundram UM, Griffin JH . General and efficient method for the solution- and solid-phase synthesis of vancomycin carboxamide derivatives. J Org Chem 60: 1102–1103 ( 1995);
Shi Z, Griffin JH . Catalysis of carbamate hydrolysis by vancomycin and semisynthetic derivatives. J Am Chem Soc 115: 6482–6486 ( 1993)
Pavlov AY, Lazhki EI, Preobrazhenskaya MN . A new type of chemical modification of glycopeptides antibiotics: aminomethylated derivatives of eremomycin and their antibacterial activity. J Antibiot 50: 509–513 ( 1997)
Leadbetter MR, Adams SM, Bazzini B, Fatheree PR, Karr DE, Krause KM, Lam BT, Linsell MS, Nodwell MB, Pace JL, Quast K, Shaw J-P, Soriano E, Trapp SG, Villena JD, Wu TX, Christensen BG, Judice JK . Hydrophobic vancomycin derivatives with improved ADME properties: Discovery of telavancin (TD-6424). J Antibiot 57: 326–336 ( 2004)
Heck AJ, Bonnici PJ, Breukink E, Wills M . Modification and inhibition of vancomycin group antibiotics by formaldehyde and acetaldehyde. Chemistry 7: 910–916 ( 2001)
National Committee for Clinical Laboratory Standards. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved guideline, 5th ed., M7-A5. The Committee, Wayne, Penn. ( 2000)
Hegde SS, Reyes N, Wiens T, Vanasse N, Skinner R, McCullough J, Kaniga K, Pace J, Thomas R, Shaw JP, Obedencio G, Judice JK . Pharmacodynamics of telavancin (TD-6424), a novel bactericidal agent, against Gram-positive bacteria. Antimicrob Agents Chemother 48: 3043–3050 ( 2004)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Long, D., Aggen, J., Chinn, J. et al. Exploring the Positional Attachment of Glycopeptide/β-lactam Heterodimers. J Antibiot 61, 603–614 (2008). https://doi.org/10.1038/ja.2008.80
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2008.80
Keywords
This article is cited by
-
Targeting bactoprenol-coupled cell envelope precursors
Applied Microbiology and Biotechnology (2016)
-
Speculative strategies for new antibacterials: all roads should not lead to Rome
The Journal of Antibiotics (2013)