Abstract
Objective:
To examine the effects of ghrelin on appetite and energy expenditure in lean, obese and postgastrectomy subjects.
Design:
A randomized, double-blind, placebo-controlled study.
Patients:
Nine lean subjects (mean body mass index (BMI) 23.5±3 kg/m2) and nine morbidly obese subjects (mean BMI 51.4±10 kg/m2) and eight postgastrectomy subjects (mean BMI 22.4±1.0 kg/m2).
Interventions:
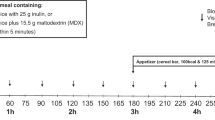
Subjects were infused with either intravenous ghrelin (5 pmol kg−1 min−1) or saline over 270 min. They were given a fixed energy breakfast followed by a free buffet lunch towards the end of the infusion.
Main outcome measures:
Visual analogue scales were used to record hunger and energy expenditure was measured by indirect calorimetry.
Results:
Ghrelin increased energy intake at the buffet lunch in lean subjects (a 41% increase, P<0.01) and obese subjects (35% increase, P=0.04) but not in postgastrectomy subjects. Lean subjects showed a characteristic preprandial rise and postprandial fall in hunger scores, which was exaggerated by ghrelin infusion. Obese subjects showed little variation in hunger scores, but a ‘lean-type’ pattern was restored when given exogenous ghrelin. Ghrelin had no effect on resting metabolic rate but did increase respiratory quotient (RQ) in obese subjects. Ghrelin also increased RQ variability over time in all three groups (ANOVA, P<0.001).
Conclusions:
Hunger scores are abnormal in the obese, perhaps because of impaired ghrelin secretion. The effect of ghrelin in restoring normal hunger profiles in the obese suggests causality, confirming an important role in eating behaviour. Ghrelin also increases RQ in obese humans and increased RQ variability in all groups. This suggests that ghrelin regulates substrate utilization and may promote metabolic flexibility.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K . Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999; 402: 656–660.
Druce MR, Wren AM, Park AJ, Milton JE, Patterson M, Frost G et al. Ghrelin increases food intake in obese as well as lean subjects. Int J Obes (Lond) 2005; 29: 1130–1136.
Wren AM, Seal LJ, Cohen MA, Brynes AE, Frost GS, Murphy KG et al. Ghrelin enhances appetite and increases food intake in humans. J Clin Endocrinol Metab 2001; 86: 5992.
Wren AM, Small CJ, Abbott CR, Dhillo WS, Seal LJ, Cohen MA et al. Ghrelin causes hyperphagia and obesity in rats. Diabetes 2001; 50: 2540–2547.
Cummings DE, Purnell JQ, Frayo RS, Schmidova K, Wisse BE, Weigle DS . A preprandial rise in plasma ghrelin levels suggests a role in meal initiation in humans. Diabetes 2001; 50: 1714–1719.
Cummings DE, Frayo RS, Marmonier C, Aubert R, Chapelot D . Plasma ghrelin levels and hunger scores in humans initiating meals voluntarily without time- and food-related cues. Am J Physiol Endocrinol Metab 2004; 287: E297–E304.
Tschop M, Smiley DL, Heiman ML . Ghrelin induces adiposity in rodents. Nature 2000; 407: 908–913.
English PJ, Ghatei MA, Malik IA, Bloom SR, Wilding JP . Food fails to suppress ghrelin levels in obese humans. J Clin Endocrinol Metab 2002; 87: 2984.
De JL, Bray GA . The thermic effect of food and obesity: a critical review. Obes Res 1997; 5: 622–631.
Date Y, Kojima M, Hosoda H, Sawaguchi A, Mondal MS, Suganuma T et al. Ghrelin, a novel growth hormone-releasing acylated peptide, is synthesized in a distinct endocrine cell type in the gastrointestinal tracts of rats and humans. Endocrinology 2000; 141: 4255–4261.
Dornonville de la CC, Bjorkqvist M, Sandvik AK, Bakke I, Zhao CM, Chen D et al. A-like cells in the rat stomach contain ghrelin and do not operate under gastrin control. Regul Pept 2001; 99: 141–150.
Le Roux CW, Neary NM, Halsey TJ, Small CJ, Martinez-Isla AM, Ghatei MA et al. Ghrelin does not stimulate food intake in patients with surgical procedures involving vagotomy. J Clin Endocrinol Metab 2005; 90: 4521–4524.
Neary NM, Small CJ, Wren AM, Lee JL, Druce MR, Palmieri C et al. Ghrelin increases energy intake in cancer patients with impaired appetite: acute, randomized, placebo-controlled trial. J Clin Endocrinol Metab 2004; 89: 2832–2836.
Karlsson J, Persson LO, Sjostrom L, Sullivan M . Psychometric properties and factor structure of the Three-Factor Eating Questionnaire (TFEQ) in obese men and women. Results from the Swedish Obese Subjects (SOS) study. Int J Obes Relat Metab Disord 2000; 24: 1715–1725.
Barkeling B, King NA, Naslund E, Blundell JE . Characterization of obese individuals who claim to detect no relationship between their eating pattern and sensations of hunger or fullness. Int J Obes (Lond) 2007; 31: 435–439.
Tschop M, Weyer C, Tataranni PA, Devanarayan V, Ravussin E, Heiman ML . Circulating ghrelin levels are decreased in human obesity. Diabetes 2001; 50: 707–709.
le Roux CW, Batterham RL, Aylwin SJ, Patterson M, Borg CM, Wynne KJ et al. Attenuated peptide YY release in obese subjects is associated with reduced satiety. Endocrinology 2006; 147: 3–8.
Batterham RL, Cohen MA, Ellis SM, Le Roux CW, Withers DJ, Frost GS et al. Inhibition of food intake in obese subjects by peptide YY3-36. N Engl J Med 2003; 349: 941–948.
Verdich C, Toubro S, Buemann B, Lysgard MJ, Juul HJ, Astrup A . The role of postprandial releases of insulin and incretin hormones in meal-induced satiety--effect of obesity and weight reduction. Int J Obes Relat Metab Disord 2001; 25: 1206–1214.
Jeon TY, Lee S, Kim HH, Kim YJ, Son HC, Kim DH et al. Changes in plasma ghrelin concentration immediately after gastrectomy in patients with early gastric cancer. J Clin Endocrinol Metab 2004; 89: 5392–5396.
Pekic S, Pesko P, Djurovic M, Miljic D, Doknic M, Glodic J et al. Plasma ghrelin levels of gastrectomized and vagotomized patients are not affected by glucose administration. Clin Endocrinol (Oxf) 2006; 64: 684–688.
Damjanovic SS, Lalic NM, Pesko PM, Petakov MS, Jotic A, Miljic D et al. Acute effects of ghrelin on insulin secretion and glucose disposal rate in gastrectomized patients. J Clin Endocrinol Metab 2006; 91: 2574–2581.
Popovic V, Miljic D, Pekic S, Pesko P, Djurovic M, Doknic M et al. Low plasma ghrelin level in gastrectomized patients is accompanied by enhanced sensitivity to the ghrelin-induced growth hormone release. J Clin Endocrinol Metab 2005; 90: 2187–2191.
Buhl K, Schlag P, Herfarth C . Quality of life and functional results following different types of resection for gastric carcinoma. Eur J Surg Oncol 1990; 16: 404–409.
Bozzetti F, Ravera E, Cozzaglio L, Dossena G, Agradi E, Bonfanti G et al. Comparison of nutritional status after total or subtotal gastrectomy. Nutrition 1990; 6: 371–375.
Takachi K, Doki Y, Ishikawa O, Miyashiro I, Sasaki Y, Ohigashi H et al. Postoperative ghrelin levels and delayed recovery from body weight loss after distal or total gastrectomy. J Surg Res 2006; 130: 1–7.
Date Y, Murakami N, Toshinai K, Matsukura S, Niijima A, Matsuo H et al. The role of the gastric afferent vagal nerve in ghrelin-induced feeding and growth hormone secretion in rats. Gastroenterology 2002; 123: 1120–1128.
Arnold M, Mura A, Langhans W, Geary N . Gut vagal afferents are not necessary for the eating-stimulatory effect of intraperitoneally injected ghrelin in the rat. J Neurosci 2006; 26: 11052–11060.
Bergh C, Sjostedt S, Hellers G, Zandian M, Sodersten P . Meal size, satiety and cholecystokinin in gastrectomized humans. Physiol Behav 2003; 78: 143–147.
Olsson U, Bergbom I, Bosaeus I . Patients’ experiences of their intake of food and fluid following gastrectomy due to tumor. Gastroenterol Nurs 2002; 25: 146–153.
Wortley KE, Anderson KD, Garcia K, Murray JD, Malinova L, Liu R et al. Genetic deletion of ghrelin does not decrease food intake but influences metabolic fuel preference. Proc Natl Acad Sci USA 2004; 101: 8227–8232.
Labayen I, Diez N, Parra D, Gonzalez A, Martinez JA . Basal and postprandial substrate oxidation rates in obese women receiving two test meals with different protein content. Clin Nutr 2004; 23: 571–578.
Longo KA, Charoenthongtrakul S, Giuliana DJ, Govek EK, McDonagh T, Qi Y et al. Improved insulin sensitivity and metabolic flexibility in ghrelin receptor knockout mice. Regul Pept 2008; 150: 55–61.
Broglio F, Gottero C, Prodam F, Gauna C, Muccioli G, Papotti M et al. Non-acylated ghrelin counteracts the metabolic but not the neuroendocrine response to acylated ghrelin in humans. J Clin Endocrinol Metab 2004; 89: 3062–3065.
Neary NM, Druce MR, Small CJ, Bloom SR . Acylated ghrelin stimulates food intake in the fed and fasted states but desacylated ghrelin has no effect. Gut 2006; 55: 135.
Acknowledgements
We thank Emma Boyland and David Kerrigan for their contributions to the study, as well as all of the volunteers who took part. This work was supported by an International Endocrine Research Prize from Pharmacia awarded to JP and PJE.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
None.
Rights and permissions
About this article
Cite this article
Huda, M., Dovey, T., Wong, S. et al. Ghrelin restores ‘lean-type’ hunger and energy expenditure profiles in morbidly obese subjects but has no effect on postgastrectomy subjects. Int J Obes 33, 317–325 (2009). https://doi.org/10.1038/ijo.2008.270
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2008.270
Keywords
This article is cited by
-
Identification, expression analysis, and functional characterization of ghrelin and its receptors in spotted sea bass (Lateolabrax maculatus)
Marine Life Science & Technology (2020)
-
Two ghrelin receptor agonists for adults with malnutrition: a systematic review and meta-analysis
Nutrition Journal (2016)
-
Bariatric Surgery and Effects on Calcium and Bone Metabolism
Clinical Reviews in Bone and Mineral Metabolism (2014)