Abstract
Objective:
Gene-targeted deletion of the voltage-gated potassium channel, Kv1.3, results in ‘super-smeller’ mice that have altered firing patterns of mitral cells in the olfactory bulb, modified axonal targeting to glomerular synaptic units, and behaviorally have an increased ability to detect and discriminate odors. Moreover, the Kv1.3-null mice weighed less than their wild-type counterparts, have modified ingestive behaviors, and are resistant to fat deposition following a moderately high-fat dietary regime. In this study, we investigate whether or not gene-targeted deletion of Kv1.3 (Shaker family member) can abrogate weight gain in a genetic model of obesity, the melanocortin-4 receptor-null mouse (MC4R-null).
Design:
Mice with double gene-targeted deletions of Kv1.3 and MC4R were generated by interbreeding Kv1.3 (Kv)- and MC4R-null mouse lines to homozygosity. Developmental weights, nose to anus length, fat pad weight, fasting serum chemistry, oxygen consumption, carbon dioxide respiration, locomotor activity and caloric intake were monitored in control, Kv-null, MC4R-null and Kv/MC4R-null mice. Physiological and metabolic profiles were acquired at postnatal day 60 (P60) in order to explore changes linked to body weight at the reported onset of obesity in the MC4R-null model.
Results:
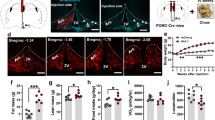
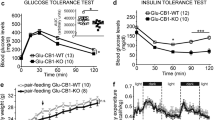
Gene-targeted deletion of Kv1.3 in MC4R-null mice reduces body weight by decreasing fat deposition and subsequent fasting leptin levels, without changing the overall growth, fasting blood glucose or serum insulin. Gene-targeted deletion of Kv1.3 in MC4R-null mice significantly extended lifespan and increased reproductive success. Basal or light-phase mass-specific metabolic rate and locomotor activity were not affected by genetic deletion of Kv1.3 in MC4R-null mice but dark-phase locomotor activity and mass-specific metabolism were significantly increased resulting in increased total energy expenditure.
Conclusions:
Gene-targeted deletion of Kv1.3 can reduce adiposity and total body weight in a genetic model of obesity by increasing both locomotor activity and mass-specific metabolism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Bean BP . The action potential in mammalian central neurons. Nat Rev Neurosci 2007; 8: 451–465.
Kaczmarek LK . Non-conducting functions of voltage-gated ion channels. Nat Rev Neurosci 2006; 7: 761–771.
Bowlby MR, Fadool DA, Holmes TC, Levitan IB . Modulation of the Kv1.3 potassium channel by receptor tyrosine kinases. J Gen Physiol 1997; 110: 601–610.
Fadool DA, Holmes TC, Berman K, Dagan D, Levitan IB . Tyrosine phosphorylation modulates current amplitude and kinetics of a neuronal voltage-gated potassium channel. J Neurophysiol 1997; 78: 1563–1573.
Fadool DA, Levitan IB . Modulation of olfactory bulb neuron potassium current by tyrosine phosphorylation. J Neurosci 1998; 18: 6126–6137.
Cayabyab FS, Khanna R, Jones OT, Schlichter LC . Suppression of the rat microglia Kv1.3 current by src-family tyrosine kinases and oxygen/glucose deprivation. Eur J Neurosci 2000; 12: 1949–1960.
Fadool DA, Tucker K, Phillips JJ, Simmen JA . Brain insulin receptor causes activity-dependent current suppression in the olfactory bulb through multiple phosphorylation of Kv1.3. J Neurophysiol 2000; 83: 2332–2348.
Tucker K, Fadool DA . Neurotrophin modulation of voltage-gated potassium channels in rat through TrkB receptors is time and sensory experience dependent. J Physiol 2002; 542 (Part 2): 413–429.
Cook KK, Fadool DA . Two adaptor proteins differentially modulate the phosphorylation and biophysics of Kv1.3 ion channel by SRC kinase. J Biol Chem 2002; 277: 13268–13280.
Marks DR, Fadool DA . Post-synaptic density perturbs insulin-induced Kv1.3 channel modulation via a clustering mechanism involving the SH3 domain. J Neurochem 2007; 103: 1608–1627.
Kues WA, Wunder F . Heterogeneous expression patterns of mammalian potassium channel genes in developing and adult rat brain. Eur J Neurosci 1992; 4: 1296–1308.
Xu J, Koni PA, Wang P, Li G, Kaczmarek L, Wu Y et al. The voltage-gated potassium channel Kv1.3 regulates energy homeostasis and body weight. Hum Mol Genet 2003; 12: 551–559.
Mourre C, Chernova MN, Martin-Eauclaire MF, Bessone R, Jacquet G, Gola M et al. Distribution in rat brain of binding sites of kaliotoxin, a blocker of Kv1.1 and Kv1.3 alpha-subunits. J Pharmacol Exp Ther 1999; 291: 943–952.
Decoursey TE, Chandy KG, Gupta S, Cahalan MD . Voltage-dependent ion channels in T-lymphocytes. J Neuroimmunol 1985; 10: 79–95.
Szabo I, Bock J, Jekle A, Soddemann M, Adams C, Lang F et al. A novel potassium channel in lymphocyte mitochondria. J Biol Chem 2005; 280: 12790–12798.
Norenberg W, Gebicke-Haerter PJ, Illes P . Voltage-dependent potassium channels in activated rat microglia. J Physiol (London) 1994; 475: 15–32.
Biju KC, Marks DR, Mast TG, Fadool DA . Deletion of voltage-gated channel affects glomerular refinement and odorant receptor expression in the mouse olfactory system. J Comp Neurol 2008; 506: 161–179.
Fadool DA, Tucker K, Perkins R, Fasciani G, Thompson RN, Parsons AD et al. Kv1.3 channel gene-targeted deletion produces ‘Super-Smeller Mice’ with altered glomeruli, interacting scaffolding proteins, and biophysics. Neuron 2004; 41: 389–404.
Xu J, Wang P, Li Y, Li G, Kaczmarek LK, Wu Y et al. The voltage-gated potassium channel Kv1.3 regulates peripheral insulin sensitivity. Proc Natl Acad Sci USA 2004; 101: 3112–3117.
Li Y, Wang P, Xu J, Desir GV . Voltage-gated potassium channel Kv1.3 regulates GLUT4 trafficking to the plasma membrane via a Ca2+-dependent mechanism. Am J Physiol Cell Physiol 2006; 290: C345–C351.
Li Y, Wang P, Xu J, Gorelick F, Yamazaki H, Andrews N et al. Regulation of insulin secretion and GLUT4 trafficking by the calcium sensor synaptotagmin VII. Biochem Biophys Res Commun 2007; 362: 658–664.
Butler AA . The melanocortin system and energy balance. Peptides 2006; 27: 281–290.
Berthoud HR, Sutton GM, Townsend RL, Patterson LM, Zheng H . Brainstem mechanisms integrating gut-derived satiety signals and descending forebrain information in the control of meal size. Physiol Behav 2006; 89: 517–524.
Huszar D, Lynch CA, Fairchild-Huntress V, Dunmore JH, Fang Q, Berkemeier LR et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 1997; 88: 131–141.
Weide K, Christ N, Moar KM, Arens J, Hinney A, Mercer JG et al. Hyperphagia, not hypometabolism, causes early onset obesity in melanocortin-4 receptor knockout mice. Physiol Genomics 2003; 13: 47–56.
Koni PA, Khanna R, Chang MC, Tang MD, Kaczmarek LK, Schlichter LC et al. Compensatory anion currents in Kv1.3 channel-deficient thymocytes. J Biol Chem 2003; 278: 39443–39451.
Balthasar N, Dalgaard LT, Lee CE, Yu J, Funahashi H, Williams T et al. Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell 2005; 123: 493–505.
Schioth HB, Watanobe H . Melanocortins and reproduction. Brain Res Rev 2002; 38: 340–350.
Rashotte ME, Basco PS, Henderson RP . Daily cycles in body temperature, metabolic rate, and substrate utilization in pigeons: influence of amount and timing of food consumption. Physiol Behav 1995; 57: 731–746.
Bartholomew GA, Vleck D, Vleck CM . Instantaneous measurements of oxygen consumption during preflight warm up and postflight cooling in sphinged and saturniid moths. J Exp Biol 1981; 90: 17–32.
Arch J, Hislop D, Wang S, Speakman J . Some mathematical and technical issues in the measurement and interpretation of open-circuit indirect calorimetry in small animals. Int J Obes 2006; 30: 1322–1331.
Blaxter K . Energy Metabolism in Animals and Man. Cambridge University Press: Cambridge, UK, 1989.
Weir JB . New methods for calculating metabolic rate with special reference to protein metabolism. J Physiol (London) 1949; 109: 1–9.
Williams TD, Chambers JB, Henderson RP, Rashotte ME, Overton JM . Cardiovascular responses to caloric restriction and thermoneutrality in C57BL/6J mice. Am J Physiol Regul Integr Comp Physiol 2002; 282: R1459–R1467.
Williams TD, Chambers JB, May OL, Henderson RP, Rashotte ME, Overton JM . Concurrent reductions in blood pressure and metabolic rate during fasting in the unrestrained SHR. Am J Physiol Regul Integr Comp Physiol 2000; 278: R255–R262.
Marie L, Miura GI, Marsh DJ, Yagaloff K, Palmiter RD . A metabolic defect promotes obesity in mice lacking melanocortin-4 receptors. PNAS 2000; 97: 12339–12344.
Mutch DM, Clement K . Genetics of human obesity. Best Pract Res Clin Endocrinol Metab 2006; 20: 647–664.
Carroll L, Voisey J, Van Daal A . Mouse models of obesity. Clin Dermatol 2004; 22: 345–349.
Asarian L, Geary N . Modulation of appetite by gonadal steroid hormones. Philos Trans R Soc Lond B Biol Sci 2006; 361: 1251–1263.
Novak CM, Levine JA . Central neural and endocrine mechanisms of non-exercise activity thermogenesis and their potential impact on obesity. J Neuroendocrinol 2007; 19: 923–940.
Irani BG, Xiang Z, Moore MC, Mandel RJ, Haskell-Luevano C . Voluntary exercise delays monogenetic obesity and overcomes reproductive dysfunction of the melanocortin-4 receptor knockout mouse. Biochem Biophys Res Commun 2005; 326: 638–644.
Zheng H, Patterson LM, Phifer CB, Berthoud HR . Brain stem melanocortinergic modulation of meal size and identification of hypothalamic POMC projections. Am J Physiol Regul Integr Comp Physiol 2005; 289: R247–R258.
Harrison DE, Archer JR, Astle CM . Effects of food restriction on aging: separation of food intake and adiposity. Proc Natl Acad Sci USA 1984; 81: 1835–1838.
Bertrand HA, Lynd FT, Masoro EJ, Yu BP . Changes in adipose mass and cellularity through the adult life of rats fed ad libitum or a life-prolonging restricted diet. J Gerontol 1980; 35: 827–835.
McCay CM . Nutritional experiments on longevity. J Am Geriatr Soc 1958; 6: 171–181.
Bluher M, Kahn BB, Kahn CR . Extended longevity in mice lacking the insulin receptor in adipose tissue. Science 2003; 299: 572–574.
Huang AM, Jen CJ, Chen HF, Yu L, Kuo YM, Chen HI . Compulsive exercise acutely upregulates rat hippocampal brain-derived neurotrophic factor. J Neural Transm 2006; 113: 803–811.
Xu B, Goulding EH, Zang K, Cepoi D, Cone RD, Jones KR et al. Brain-derived neurotrophic factor regulates energy balance downstream of melanocortin-4 receptor. Nat Neurosci 2003; 6: 736–742.
Nicholson JR, Peter JC, Lecourt AC, Barde YA, Hofbauer KG . Melanocortin-4 receptor activation stimulates hypothalamic brain-derived neurotrophic factor release to regulate food intake, body temperature and cardiovascular function. J Neuroendocrinol 2007; 19: 974–982.
Ono M, Itakura Y, Nonomura T, Nakagawa T, Nakayama C, Taiji M et al. Intermittent administration of brain-derived neurotrophic factor ameliorates glucose metabolism in obese diabetic mice. Metabolism 2000; 49: 129–133.
Gray J, Yeo GSH, Cox JJ, Morton J, Adlam AL, Keogh JM et al. Hyperphagia, severe obesity, impaired cognitive function, and hyperactivity associated with functional loss of one copy of the brain-derived neurotrophic factor (BDNF) gene. Diabetes 2006; 55: 3366–3371.
Wang C, Bomberg E, Billington C, Levine A, Kotz CM . Brain-derived neurotrophic factor in the hypothalamic paraventricular nucleus increases energy expenditure by elevating metabolic rate. Am J Physiol Regul Integr Comp Physiol 2007; 293: R992–1002.
Wang C, Bomberg E, Billington C, Levine A, Kotz CM . Brain-derived neurotrophic factor in the hypothalamic paraventricular nucleus reduces energy intake. Am J Physiol Regul Integr Comp Physiol 2007; 293: R1003–R1012.
Wang C, Bomberg E, Levine A, Billington C, Kotz CM . Brain-derived neurotrophic factor in the ventromedial nucleus of the hypothalamus reduces energy intake. Am J Physiol Regul Integr Comp Physiol 2007; 293: R1037–R1045.
Yamanaka M, Tsuchida A, Nakagawa T, Nonomura T, Ono-Kishino M, Sugaru E et al. Brain-derived neurotrophic factor enhances glucose utilization in peripheral tissues of diabetic mice. Diabetes Obes Metab 2007; 9: 59–64.
Kernie SG, Liebl DJ, Parada LF . BDNF regulates eating behavior and locomotor activity in mice. EMBO J 2000; 19: 1290–1300.
Fox EA, Byerly MS . A mechanism underlying mature-onset obesity: evidence from the hyperphagic phenotype of brain-derived neurotrophic factor mutants. Am J Physiol Regul Integr Comp Physiol 2004; 286: R994–1004.
Unger TJ, Calderon GA, Bradley LC, Sena-Esteves M, Rios M . Selective deletion of Bdnf in the ventromedial and dorsomedial hypothalamus of adult mice results in hyperphagic behavior and obesity. J Neurosci 2007; 27: 14265–14274.
Monteggia LM, Luikart B, Barrot M, Theobold D, Malkovska I, Nef S et al. Brain-derived neurotrophic factor conditional knockouts show gender differences in depression-related behaviors. Biol Psychiatry 2007; 61: 187–197.
Zorner B, Wolfer DP, Brandis D, Kretz O, Zacher C, Madani R et al. Forebrain-specific trkB-receptor knockout mice: behaviorally more hyperactive than ‘depressive’. Biol Psychiatry 2003; 54: 972–982.
Acknowledgements
We thank Dr Leonard Kaczmarek and Dr Richard Flavell of Yale University School of Medicine, New Haven, CT, for donation of the Kv1.3-null mice and Dr Joel Elmquist of the University of Texas, Southwestern Medical Center, Dallas, TX, for the generous gift of the MC4R-null mice. A special thanks to Thomas Mast, Michelina Messina, Jeffery Godbey, Michael Henderson and Robert Daly for mouse maintenance and technical help. This work was supported by NIH grants DC03387 and T32 DC00044 (NIDCD), the Robinson Foundation (Tallahassee Memorial Hospital) and a CRC planning grant (FSU).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on International Journal of Obesity website (http://www.nature.com/ijo)
Supplementary information
Rights and permissions
About this article
Cite this article
Tucker, K., Overton, J. & Fadool, D. Kv1.3 gene-targeted deletion alters longevity and reduces adiposity by increasing locomotion and metabolism in melanocortin-4 receptor-null mice. Int J Obes 32, 1222–1232 (2008). https://doi.org/10.1038/ijo.2008.77
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2008.77
Keywords
This article is cited by
-
Loss of odor-induced c-Fos expression of juxtaglomerular activity following maintenance of mice on fatty diets
Journal of Bioenergetics and Biomembranes (2019)
-
Caveolar targeting links Kv1.3 with the insulin-dependent adipocyte physiology
Cellular and Molecular Life Sciences (2018)
-
Kv1.3 contains an alternative C-terminal ER exit motif and is recruited into COPII vesicles by Sec24a
BMC Biochemistry (2015)
-
Structure–Activity Relationship of a Highly Selective Peptidyl Inhibitor of Kv1.3 Voltage-Gated K+-Channel from Scorpion (B. sindicus) Venom
International Journal of Peptide Research and Therapeutics (2014)
-
Kv1.3 inhibitors have differential effects on glucose uptake and AMPK activity in skeletal muscle cell lines and mouse ex vivo skeletal muscle
The Journal of Physiological Sciences (2014)