Abstract
Aims:
Lipoprotein lipase (LPL), a pivotal enzyme in lipoprotein metabolism, catalyzes the hydrolysis of triglycerides of very low-density lipoproteins and chylomicrons. Assuming that the variants in the promoter of the LPL gene may be associated with changes in lipid metabolism leading to obesity and type 2 diabetes, we examined the role of promoter variants (–T93G and –G53C) in the LPL gene in an urban South Indian population.
Methods:
The study subjects (619 type 2 diabetic and 731 normal glucose-tolerant (NGT) subjects) were chosen from the Chennai Urban Rural Epidemiology Study, an ongoing population-based study in southern India. The polymorphisms were genotyped using polymerase chain reaction-restriction-fragment length polymorphism (PCR-RFLP). Linkage disequilibrium (LD) was estimated from the estimates of haplotypic frequencies.
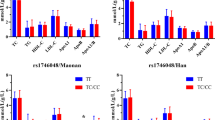
Results:
The two polymorphisms studied were not in LD. The –T93G was not associated with type 2 diabetes but was associated with obesity. 11.5% of the obese subjects (62/541) had the XG(TG+GG) genotype compared with 6.4% of the nonobese subjects (52/809; P=0.001). The odds ratio for obesity for the XG genotype was 1.766 (95% CI: 1.19–2.63, P=0.005). Subjects with XG genotype also had higher body mass index and waist circumference compared with those with TT genotype. With respect to G53C, subjects with the XC(GC+CC) genotype had 0.527 and 0.531 times lower risk for developing type 2 diabetes and obesity, respectively.
Conclusions:
Among Asian Indians, the –T93G SNP of the LPL gene is associated with obesity but not type 2 diabetes, whereas the –G53C SNP appears to be protective against both obesity and type 2 diabetes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Mohan V, Sharp PS, Cloke HR, Burrin JM, Schumer B, Kohner EM . Serum immunoreactive insulin responses to a glucose load in Asian Indian and European Type 2 (non-insulin-dependent) diabetic patients and control subjects. Diabetologia 1986; 29: 235–237.
Sharp PS, Mohan V, Levy JC, Mather HM, Kohner EM . Insulin resistance in patients of Asian Indian and European origin with non-insulin dependent diabetes. Horm Metab Res 1987; 19: 84–85.
Misra A, Vikram NK . Insulin resistance syndrome [metabolic syndrome] and Asian Indians. Curr Sci 2002; 83: 1483–1496.
Chandalia M, Abate N, Garg A, Stray-Gundersen J, Grundy SM . Relationship between generalized and upper body obesity to insulin resistance in Asian Indian men. J Clin Endocrinol Metab 1999; 84: 2329–2335.
Ramachandran A, Snehalatha C, Viswanathan V, Viswanathan M, Haffner SM . Risk of noninsulin dependent diabetes mellitus conferred by obesity and central adiposity in different ethnic groups: a comparative analysis between Asian Indians, Mexican Americans and Whites. Diabetes Res Clin Pract 1997; 36: 121–125.
Joshi SR . Metabolic Syndrome – Emerging clusters of the Indian phenotype. J Assoc Physicians India 2003; 51: 445–446.
McKeigue PM, Pierpoint T, Ferrie JE, Marmot MG . Relationship of glucose intolerance and hyperinsulinaemia to body fat pattern in south Asians and Europeans. Diabetologia 1992; 35: 785–791.
Pradeepa R, Mohan V . The changing scenario of the diabetes epidemic: implications for India. Indian J Med Res 2002; 116: 121–132.
Raji A, Seely EW, Arky RA, Siminson DC . Body fat distribution and insulin resistance in healthy Asian Indians and Caucasians. J Clin Endocrinol Metab 2001; 86: 5366–5371.
Valsamakis G, Chetty R, Anwar A, Bannerjee AK, Barneet A, Kumar S . Association of simple anthropometric measures of obesity with visceral fat and the metabolic syndrome in male Caucasian and IndoAsian subjects. Diabet Med 2004; 21: 1339–1345.
Yang W, Huang J, Yao C, Su S, Liu D, Ge D et al. Linkage and linkage disequilibrium analysis of the lipoprotein lipase gene with lipid profiles in Chinese hypertensive families. Clin Sci (Lond) 2005; 108: 137–142.
Ehrenborg E, Clee SM, Pimstone SN, Reymer PW, Benlian P, Hoogendijk CF et al. Ethnic variation and in vivo effects of the −93t-->g promoter variant in the lipoprotein lipase gene. Arterioscler Thromb Vasc Biol 1997; 17: 2672–2678.
Schoonjans K, Peinado-Onsurbe J, Lefebvre AM, Heyman RA, Briggs M, Deeb S et al. PPARalpha and PPARgamma activators direct a distinct tissue-specific transcriptional response via a PPRE in the lipoprotein lipase gene. EMBO J 1996; 15: 5336–5348.
Auwerx J, Schoonjans K, Fruchart JC, Staels B . Transcriptional control of triglyceride metabolism: fibrates and fatty acids change the expression of the LPL and apo C-III genes by activating the nuclear receptor PPAR. Atherosclerosis 1996; 124: 29–37.
Sartippour MR, Renier G . Differential regulation of macrophage peroxisome proliferator-activated receptor expression by glucose: role of peroxisome proliferator-activated receptors in lipoprotein lipase gene expression. Arterioscler Thromb Vasc Biol 2000; 20: 104–110.
Michaud SE, Renier G . Direct regulatory effect of fatty acids on macrophage lipoprotein lipase: potential role of PPARs. Diabetes 2001; 50: 660–666.
Currie RA, Eckel RH . Characterization of a high affinity octamer transcription factor binding site in the human lipoprotein lipase promoter. Arch Biochem Biophys 1992; 298: 630–639.
Kastelein JJ, Groenemeyer BE, Hallman DM, Henderson H, Reymer PW, Gagne SE et al. The Asn9 variant of lipoprotein lipase is associated with the −93G promoter mutation and an increased risk of coronary artery disease. The Regress Study Group Clin Genet 1998; 53: 27–33.
Deepa M, Pradeepa R, Rema M, Anjana M, Deepa R, Shanthirani S et al. The Chennai Urban Rural Epidemiology Study (CURES) – Study design and Methodology (Urban Component) (CURES – 1). J Assoc Physicians India 2003; 51: 863–870.
The Asia Pacific perspective: Redefining obesity and its treatment. Regional office for the western Pacific of the World Health Organization World Health Organization, International association for the study of Obesity and International Obesity Task force. Health Communications Australia Pvt Limited 2000; 22–29.
Maniatis T, Fritsch EF, Sambrook J . Molecular Cloning, A Laboratory Manual. Cold Spring Harbor Laboratory: Plainview, NY, 1982; 149–151.
Yang WS, Nevin DN, Iwasaki L, Peng R, Brown BG, Brunzell JD et al. Regulatory mutations in the human lipoprotein lipase gene in patients with familial combined hyperlipidemia and coronary artery disease. J Lipid Res 1996; 37: 2627–2637.
Tanuma Y, Nakabayashi H, Esumi M, Endo H . A silencer element for the lipoprotein lipase gene promoter and cognate double- and single-stranded DNA-binding proteins. Mol Cell Biol 1995; 15: 517–523.
Hall S, Chu G, Miller G, Cruickshank K, Cooper JA, Humphries SE et al. A common mutation in the lipoprotein lipase gene promoter, −93T/G, is associated with lower plasma triglyceride levels and increased promoter activity in vitro. Arterioscler Thromb Vasc Biol 1997; 17: 1969–1976.
Staels B, Martin G, Martinez M, Albert C, Peinado-Onsurbe J, Saladin R et al. Expression and regulation of the lipoprotein lipase gene in human adrenal cortex. J Biol Chem 1996; 271: 17425–17432.
Sheng M, Dougan ST, McFadden G, Greenberg ME . Calcium and growth factor pathways of c-fos transcriptional activation require distinct upstream regulatory sequences. Mol Cell Biol 1988; 8: 2787–2796.
Enerback S, Ohlsson BG, Samuelsson L, Bjursell G . Characterization of the human lipoprotein lipase (LPL) promoter: evidence of two cis-regulatory regions, LP-alpha and LP-beta, of importance for the differentiation-linked induction of the LPL gene during adipogenesis. Mol Cell Biol 1992; 12: 4622–4633.
Basu A, Mukherjee N, Roy S, Sengupta S, Banerjee S, Chakraborty M et al. Ethnic India: a genomic view, with special reference to peopling and structure. Genome Res 2003; 13: 2277–2290.
Devlin B, Roeder K, Wasserman L . Genomic control, a new approach to genetic-based association studies. Theor Popul Biol 2001; 60: 155–166.
Radha V, Mohan V, Vidya R, Ashok Ayyappa K, Deepa R, Rasika AM . Association of lipoprotein lipase HindIII and Ser 447 Ter polymorphisms with dyslipidemia in Asian Indians. Am J Cardiol 2006; 97: 1337–1342.
Acknowledgements
The study was supported by a grant from Dr Reddy's Laboratories, Hyderabad. The Chennai Wellingdon Corporate Foundation supported the CURES field studies. This is the 28th paper from the CURES study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Radha, V., Vimaleswaran, K., Ayyappa, K. et al. Association of lipoprotein lipase gene polymorphisms with obesity and type 2 diabetes in an Asian Indian population. Int J Obes 31, 913–918 (2007). https://doi.org/10.1038/sj.ijo.0803547
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0803547
Keywords
This article is cited by
-
Association of NFκβ, TNFα, IL-6, IL-1β, and LPL Polymorphisms with Type 2 Diabetes Mellitus and Biochemical Parameters in a Mexican Population
Biochemical Genetics (2021)
-
Genomics of body fat distribution
Journal of Genetics (2021)
-
Genetics of obesity and its measures in India
Journal of Genetics (2018)
-
High fat diet modifies the association of lipoprotein lipase gene polymorphism with high density lipoprotein cholesterol in an Asian Indian population
Nutrition & Metabolism (2017)
-
Diabetes and Associated Complications in the South Asian Population
Current Cardiology Reports (2014)