Abstract
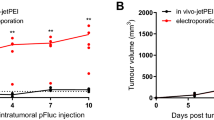
Nitric oxide (NO•) has many characteristics including cytotoxicity, radiosensitization and anti-angiogenesis, which make it an attractive molecule for use in cancer therapy. We have investigated the use of iNOS gene transfer, driven by both a constitutive (CMV) and X-ray inducible (WAF1) promoter, for generating high concentrations of NO• within tumour cells. We have combined this treatment with radiation to exploit the radiosensitizing properties of this molecule. Transfection of murine RIF-1 tumour cells in vitro with the iNOS constructs resulted in increased iNOS protein levels. Under hypoxic conditions cells were radiosensitized by delivery of both constructs so that these treatments effectively eliminated the radioresistance observed under hypoxic conditions. In vivo transfer of the CMV/iNOS construct by direct tumour injection resulted in a delay (4.2 days) in tumour growth compared with untreated controls. This was equivalent to the effect of 20 Gy X-rays alone. Combination of CMV/iNOS gene transfer with 20 Gy X-rays resulted in a dramatic 19.8 day growth delay compared with controls. Tumours treated with the CMV/iNOS showed large areas of necrosis and abundant apoptosis. We believe that iNOS gene transfer has the potential to be a highly effective treatment in combination with radiotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Moncada S., Palmer R.M., Higgs E.A. . Nitric oxide: physiology, pathophysiology, and pharmacology Pharmacol Rev 1991 43: 109 109
Maeda H., Akaike T. . Nitric oxide and oxygen radicals in infection, inflammation, and cancer Biochemistry (Mosc) 1998 63: 854 854
Brennan P.A. . The actions and interactions of nitric oxide in tumours Eur J Surg Oncol 2000 5: 434 434
Doi X. et al. Excessive production of nitric oxide in rat solid tumor and its implication in rapid tumor growth Cancer 1996 77: 1598 1598
Kennovin G.D., Hirst D.G., Stratford M.R.L., Flitney F.W. . Inducible nitric oxide synthase is expressed in tumour associated vasculature: inhibition retards tumour growth in vivo Moncada S, Feelish M, Busse R, Higgs EA (eds); The Biology of Nitric Oxide, Immunology and Inflammation Portland Press Proceedings 1995 pp 473–479
MacMicking J., Xie Q.W., Nathan C. . Nitric oxide and macrophage function Annu Rev Immunol 1997 15: 323 323
Peretti M., Szabo C., Thiemermann C. . Effect of interleukin-4 and interleukin-10 on leukocytes migration and nitric oxide production in the mouse Br J Pharmacol 1995 116: 2251 2251
Mitchell J.B. et al. Hypoxic mammalian cell radiosensitization by nitric oxide Cancer Res 1993 53: 5845 5845
Janssens M.Y. et al. Activation of inducible nitric oxide synthase results in nitric oxide-mediated radiosensitization of hypoxic EMT-6 tumor cells Cancer Res 1998 58: 5646 5646
Kurimoto M. et al. Growth inhibition and radiosensitization of cultured glioma cells by nitric oxide generating agents J Neurooncol 1999 42: 35 35
Griffin R.J., Makepeace C.M., Hur W.J., Song C.W. . Radiosensitization of hypoxic cells in vitro by nitric oxide Int J Radiat Oncol Biol Phys 1996 36: 377 377
Soler M.N. et al. Gene therapy of rat medullary thyroid cancer by naked nitric oxide synthase II DNA injection J Gene Med 2000 2: 344 344
Juang S.H. et al. Use of retroviral vectors encoding murine inducible nitric oxide synthase gene to suppress tumorigenicity and cancer metastasis of murine melanoma Cancer Biother Radiopharm 1997 12: 167 167
Kuroki M. et al. Specific targeting strategies of cancer gene therapy using a single-chain variable fragment (scFv) with a high affinity for CEA Anticancer Res 2000 20: 4067 4067
Juang S.H. et al. Suppression of tumorigenicity and metastasis of human renal carcinoma cells by infection with retroviral vectors harboring the murine inducible nitric oxide synthase gene Hum Gene Ther 1989 9: 845 845
Xie K. et al. Direct correlation between expression of endogenous inducible nitric oxide synthase and regression of M5076 reticulum cell sarcoma hepatic metastases in mice treated with liposomes containing lipopeptide CGP 31362 Cancer Res 1995 55: 3123 3123
El-Deiry W.S. et al. WAF1, a potential mediator of p53 tumour suppression Cell 1995 75: 817 817
Worthington J. et al. Modification of vascular tone using iNOS under the control of a radiation-inducible promoter Gene Therapy 2000 7: 1126 1126
Vaupel P.W. . The influence of tumor blood flow and microenvironmental factors on the efficacy of radiation, drugs and localized hyperthermia Klin Padiatr 1997 209: 243 243
Dachs G., Tozer G.M. . Hypoxia modulated gene expression: angiogenesis, metastasis and therapeutic exploitation Eur J Cancer 2000 36: 1649 1649
Tozer G.M. et al. The comparative effects of the NOS inhibitor Nomega-nitro-L-arginine, and the haemoxygenase inhibitor, zinc protoporphyrin IX, on tumour blood flow Int J Radiat Oncol Biol Phys 1998 42: 849 849
Tamir S., Burney S., Tannenbaum S.R. . DNA damage by nitric oxide Chem Res Toxicol 1996 9: 821 821
DeRojas-Walker T. et al. Nitric oxide induces oxidative damage in addition to deaminationin macrophage DNA Chem Res Toxicol 1995 8: 473 473
Wink D.A. et al. The cytotoxicity of nitroxyl: possible implications for the the pathophysiological role of NO Arch Biochem Biophy 1998 351: 66 66
Li H., Forstermann U. . Nitric oxide in the pathogenesis of vascular disease J Pathol 2000 190: 244 244
Burney S. et al. A mechanism analysis of nitric oxide-induced cellular toxicity Nitric Oxide 1997 1: 130 130
Schimmel M., Bauer G. . Selective apoptosis induction in transformed fibroblasts by B cell lines: involvement of reactive oxygen and nitrogen species Int J Oncol 2001 19: 299 299
Sakkoula E., Haralabopoulos G., Andriopoulou P., Magoudakis M.E. . Evidence that nitric oxide is an endogenous anti-angiogenic mediator Br J Pharmacol 1994 111: 894 894
Barba D. et al. Development of anti-tumour immunity following thymidine kinase-mediated killing of experimental brain tumours Proc Natl Acad Sci USA 1994 91: 4348 4348
Caruso M. et al. Regression of established macroscopic liver metastases after in situ transduction of a suicide gene Proc Natl Acad Sci USA 1993 90: 7024 7024
Hibbs J.B., Taintor R.R., Vavrin Z., Rachlin E.M. . Nitric oxide: a cytotoxic activated macrophage effector molecule Biochem Biophys Res Commun 1988 157: 87 87
Xie K. et al. Destruction of bystander cells by tumor cells transfected with inducible nitric oxide (NO) synthase gene J Natl Cancer Inst 1997 89: 421 421
Palmer R.M., Rees D.D., Ashton D.S., Moncada S. . L-arginine is the physiological precursor for the formation of NO in endothelium-dependent relaxation Biochem Biophys Res Commun 1988 153: 1251 1251
Vodovotz Y. et al. Induction of nitric oxide production in infiltrating leukocytes following in vivo irradiation of tumour-bearing mice Radiat Oncol Invest 1999 7: 86 86
Douple E.B., Green C.J., Simic M.G. . Potentiation of cellular radioensitivity by nitroprusside and vitamin B12 Int J Radiat Oncol Biol Phys 1980 6: 1545 1545
Verovski V.N. et al. Intrinsic radiosensitivity of human pancreatic tumour cells and the radiosensitising potency of the nitric oxide donor sodium nitroprusside Br J Cancer 1996 74: 1734 1734
Brown J.M. . Exploiting the hypoxic cell: mechanisms and therapeutic strategies Mol Med Today 2000 6: 157 157
Khare P.D. et al. Specifically targeted killing of carcinoembryonic antigen (CEA)-expressing cells by a retroviral vector displaying single-chain variable fragmented antibody to CEA and carrying the gene for inducible nitric oxide synthase Cancer Res 2001 61: 370 370
Twentyman P.R. et al. A new mouse tumour model system (RIF-1) for comparison of end-point studies J Natl Cancer Inst 1980 64: 595 595
Green L.C. et al. Analysis of nitrate, nitrite and [15N] nitrate in biological fluids Anal Biochem 1982 126: 131 131
Acknowledgements
This work was supported by a grant from the Cancer Research Campaign, UK. We would also like to thank Drs Stephanie McKeown and Steve Everett for their helpful discussions.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Worthington, J., Robson, T., O'Keeffe, M. et al. Tumour cell radiosensitization using constitutive (CMV) and radiation inducible (WAF1) promoters to drive the iNOS gene: a novel suicide gene therapy. Gene Ther 9, 263–269 (2002). https://doi.org/10.1038/sj.gt.3301609
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301609
Keywords
This article is cited by
-
Evaluation of p21 promoter for interleukin 12 radiation induced transcriptional targeting in a mouse tumor model
Molecular Cancer (2013)
-
The radiation-inducible pE9 promoter driving inducible nitric oxide synthase radiosensitizes hypoxic tumour cells to radiation
Gene Therapy (2008)
-
p21(WAF1)-mediated transcriptional targeting of inducible nitric oxide synthase gene therapy sensitizes tumours to fractionated radiotherapy
Gene Therapy (2007)
-
Cancer gene therapy: Combination with radiation therapy and the role of bystander cell killing in the anti-tumor effect
Pathology & Oncology Research (2006)
-
Evaluation of a synthetic CArG promoter for nitric oxide synthase gene therapy of cancer
Gene Therapy (2005)