Abstract
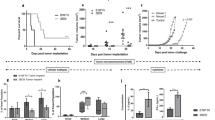
Most neoplasms do not induce antitumor immune responses that can control tumor growth. Tumor associated antigens (TAAs) are insufficiently immunogenic. A vaccine that augments the immunogenic properties of TAAs could be of importance in the treatment of cancer patients. In an animal model, we prepared a vaccine by transfection of highly antigenic allogeneic mouse fibroblasts (LM; H-2k) with DNA from B16 mouse melanoma cells. We then tested the transfected cells’ immunogenic properties in C57BL/6 mice, syngeneic with the melanoma (H-2b). We hypothesized that the immunogenic properties of ‘weak’ TAAs formed by the neoplasm would be enhanced if they were expressed by highly antigenic cells. The results indicated that mice with melanoma treated by immunization with the DNA-transfected fibroblasts survived significantly longer than mice in various control groups. To investigate the contribution of MHC determinants expressed by the transfected cells to their immunogenic properties, we compared the antimelanoma responses in mice immunized with transfected cells that expressed allogeneic or syngeneic class I determinants. The results indicated that the immunogenic properties of the DNA-transfected cells were enhanced if the cells expressed allogeneic MHC determinants. The antimelanoma responses of greatest magnitude, however, mediated predominantly by CD8+ T cells, were in mice immunized with transfected fibroblasts that expressed both syngeneic and allogeneic class I determinants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Boon T et al. Tumor antigens recognized by T lymphocytes Ann Rev Immunol 1994 12: 337–365
Boon T . Tumor antigens recognized by cytolytic T lymphocytes: present perspectives for specific immunotherapy Int J Cancer 1993 54: 177–180
Boon T . Toward a genetic analysis of tumor rejection antigens Adv Cancer Res 1992 58: 177–210
Storkus WJ et al. Identification of human melanoma peptides recognized by class I restricted tumors infiltrating T lymphocytes J Immunol 1993 151: 3719–3727
Boel P et al. BAGE: a new gene encoding an antigen recognized on human melanomas by cytolytic T lymphocytes Immunity 1995 2: 167–175
Van den Eynde B et al. A new family of genes coding for an antigen recognized by autologous cytolytic T lymphocytes on a human melanoma J Exp Med 1995 182: 689–698
De Smet C et al. Involvement of two Ets binding sites in the transcriptional activation of the MAGE-1 gene Immunogenetics 1995 42: 282–290
van der Bruggen P et al. A peptide encoded by human gene MAGE-3 and presented by HLA-A2 induces cytolytic T lymphocytes that recognize tumor cells expressing MAGE-3 Eur J Immunol 1994 24: 3038–3043
Traversari C et al. A nonapeptide encoded by human gene Mage-2 is recognized on HLA-A1 by cytolytic T lymphocytes directed against tumor antigen MZ2-E J Exp Med 1992 176: 1453–1457
Brichard V et al. The tyrosinase gene codes for an antigen recognized by autologous cytolytic lymphocytes on HLA-A2 melanomas J Exp Med 1993 178: 489–495
van den Eynde B et al. A new family of genes coding for an antigen recognized by autologous cytolytic T lymphocytes on a human melanoma J Exp Med 1995 182: 689–698
Bakker ABH et al. Melanocyte lineage-specific antigen gp100 is recognized by melanoma derived tumor infiltrating lymphocytes J Exp Med 1994 179: 1005–1009
Kawakami Y et al. Identification of the immunodominant peptides of the MART-1 human melanoma antigen recognized by the majority of HLA-A2-restricted tumor infiltrating lymphocytes J Exp Med 1994 180: 347–352
Wooster RA et al. Instability of short tandem repeats (microsatellites) in human cancers Nat Genet 1994 6: 152–156
Merlo AM et al. Frequent microsatellite instability in primary small lung cancer Cancer Res 1994 54: 2098–2101
Orth K et al. Genetic instability in human ovarian cancer cell lines Proc Natl Acad Sci USA 1994 91: 9495–9499
Parsons R et al. Hypermutability and mismatch repair deficiency in RER+ tumor cells Cell 1993 75: 1227–1236
Shibata D et al. Genomic instability in repeated sequences is an early somatic event in colorectal tumorigeneis that persists after transformation Nat Genetics 1994 6: 273–281
Cohen EP, de Zoeten EF, Schatzman M . DNA vaccines as cancer treatment Am Sci 1999 87: 328–335
Kim TS, Cohen EP . Interleukin-2-secreting mouse fibroblasts transfected with genomic DNA from murine melanoma cells prolong the survival of mice with melanoma Cancer Res 1994 54: 2531–2535
Sun T, Kim TS, Waltz MR, Cohen EP . Interleukin-2-secreting mouse fibroblasts transfected with genomic DNA from murine neoplasms induce tumor-specific immune responses that prolong the lives of tumor-bearing mice Cancer Gene Ther 1995 2: 183–190
Sun T, de Zoeten EF, Carr-Brendel V, Cohen EP . Immunization with interleukin-2-secreting allogeneic cells transfected with DNA from mouse melanoma cells induces immune responses that prolong the lives of mice with melanoma Cancer Gene Ther 1998 5: 110–118
Wigler M et al. DNA-mediated transfer of the adenine phosphoribosyltransferase locus into mammalian cells Proc Natl Acad Sci USA 1979 76: 1373–1376
Wigler M, Pellicer A, Silverstein S, Axel R . Biochemical transfer of single-copy eucaryotic genes using total cellular DNA as donor Cell 1978 14: 725–731
Mendersohn C et al. Transformation of a human poliovirus receptor gene into mouse cells Proc Natl Acad Sci USA 1986 83: 7845–7851
Barraclough R et al. Use of DNA transfer in the induction of metastasis in experimental mammary systems Biochem Soc Symp 1998 63: 273–294
Chen H et al. Isolation of and effector for metastasis-inducing DNAs from a human metastatic carcinoma cell line Oncogene 1997 14: 1581–1588
Hsu C, Kavathas P, Herzenberg LA . Cell surface antigens expressed on L cells transfected with whole DNA from non expressing and expressing cells Nature 1984 312: 68–69
Kavathas P, Herzenberg LA . Stable transformation of mouse LM cells (a transformed fibroblast cell line) for human membrane T cell differentiation antigens HLA and B2 microglobulin: selection by fluorescence activated cell sorting Proc Natl Acad Sci USA 1983 80: 524–528
Shih C, Padhy LC, Murray M, Weinberg RA . Transforming genes of carcinomas and neuroblastomas induced into mouse fibroblasts Nature 1981 290: 261–264
Shih C, Weinberg RA . Isolation of a transforming sequence from a human bladder carcinoma cell line Cell 1982 29: 161–169
Townsend SE, Allison JP . Tumor rejection after direct co stimulation of CD8+ T cells by B7-transfected melanoma cells Science 1993 259: 368–370
Allison JP, Hurwitz AA, Leach DR . Manipulation of costimulatory signals to enhance antitumor T cell responses Curr Opin Immunol 1995 7: 682–686
Gillis S, Ferm MM, Ou W, Smith KA . T cell growth factor: parameters of production and quantitative microassay for activity J Immunol 1978 120: 2027–2032
de Zoeten EF, Carr-Brendel V, Cohen EP . Resistance to melanoma in mice immunized with semi-allogeneic fibroblasts transfected with genomic DNA from melanoma cells J Immunol 1998 160: 2915–2922
de Zoeten E et al. Immunity to breast cancer in mice immunized with semi-allogeneic fibroblasts transfected with DNA from breast cancer cells J Immunol 1999 162: 6934–6941
Hui KM, Sim TF, Foo TT . Oei A-A. Tumor rejection mediated by transfection with allogeneic class I histocompatibility gene J Immunol 1989 143: 3835–3843
Ostrand-Rosenberg S, Thakur A, Clements V . Rejection of mouse sarcoma cells after transfection of MHC class II genes J Immunol 1990 144: 4068–4071
Fearon ER et al. Induction in a murine tumor of immunogenic tumor variants by transfection with a foreign gene Cancer Res 1988 48: 2975–2980
Gattoni-Celli S et al. Partial suppression of anchorage-dependent growth and tumorigenicity in immunodeficient mice by transfection of the H-2 class I gene H-2Ld into a human colon cancer cell line (HCT) Proc Natl Acad Sci USA 1988 85: 8543–8547
Fearon ER et al. Interleukin-2 production by tumor cells by-passes T helper function in the generation of an anti tumor response Cell 1990 60: 397–403
Connor J et al. Regression of bladder tumors in mice treated with interleukin-2 gene modified tumor cells J Exp Med 1993 177: 1127–1134
Hammerling GJ et al. Manipulation of metastasis and tumor growth by transfection wth histocompatibility class I genes J Immunogen 1986 13: 151–157
Tahara H et al. Fibroblasts genetically engineered to secrete IL-12 can suppress tumor growth and induce anti tumor immunity to murine melanoma in vivo Cancer Res 1994 54: 182–189
Conroy RM . A carcinoembryonic antigen polynucleotide vaccine has in vivo tumor activity Gene Therapy 1995 2: 59–65
Pertmer TM et al. Gene gun based nucleic acid immunization. Elucidation of humoral and cytotoxic T lymphocyte responses following epidermal delivery of nanogram quantities of DNA Vaccine 1995 13: 1427–1430
Wolff JA et al. Direct gene transfer into mouse muscle in vivo Science 1990 247: 1465–1468
Wolff JA et al. Direct gene transfer into mouse muscle in vivo Cohen EP, Kim TS. Neoplastic cells that express low levels of MHC class I determinants escape host immunity. In: Collins MK (ed.). Seminars in Cancer Biology, vol. 5. Academic Press: London, 1994, pp 419-428.
deVries TJ et al. Heterologous expression of immunotherapy candidate proteins gp100, MART-1 and tyrosinase in melanoma cell lines and in human melanocytic lesions Cancer Res 1997 57: 3223–3229
Pardoll DM . Cancer vaccines Nat Med 1998 4: 525–531
Acknowledgements
This work was supported by United States Army Medical Research and Material Command, grant No. DAMD 17-96-1-6178.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Zoeten, E., Markovic, D. & Cohen, E. An optimum anti-melanoma response in mice immunized with fibroblasts transfected with DNA from mouse melanoma cells requires the expression of both syngeneic and allogeneic MHC-determinants. Gene Ther 9, 1163–1172 (2002). https://doi.org/10.1038/sj.gt.3301765
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301765
Keywords
This article is cited by
-
Intratumoral injection of IL-secreting syngeneic/allogeneic fibroblasts transfected with DNA from breast cancer cells prolongs the survival of mice with intracerebral breast cancer
Cancer Gene Therapy (2005)
-
Immunity to breast cancer in mice immunized with fibroblasts transfected with a cDNA expression library derived from small numbers of breast cancer cells
Cancer Gene Therapy (2005)