Abstract
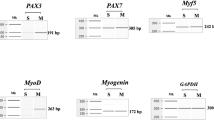
The limited proliferative capacity of DMD myoblasts severely limits their ability to be genetically modified and used for myoblast transplantation. Transformation by SV40 large T antigen (Tag) delays senescence of mouse and human myoblasts but fails to immortalize these cells. The cells ceased to proliferate and entered into crisis. Reconstitution of telomerase activity has been shown sufficient to enable different types of transformed cells to escape crisis. DMD myoblasts, previously transformed by Tag, were therefore infected with a telomerase retrovirus. The expression of telomerase was sufficient to allow DMD-Tag myoblasts to escape crisis. The telomerase-positive transformed myoblasts continued to divide for more than 55 doublings while Tag myoblasts stopped proliferating after 35 doublings. These cells are able to fuse and to differentiate normally. The average telomere length of these telomerase-positive DMD-Tag myoblasts seems to continue to elongate. Thus, transiently genetically modified myoblasts could constitute an important pool of DMD myoblasts for autologous transplantation in DMD patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Webster C, Blau HM . Accelerated age-related decline in replicative life-span of Duchenne muscular dystrophy myoblasts: implications for cell and gene therapy Somat Cell Mol Genet 1990 16: 557–565
Simon LV, Beauchamp JR, O'Hare M, Olsen I . Establishment of long-term myogenic cultures from patients with Duchenne muscular dystrophy by retroviral transduction of a temperature-sensitive SV40 large T antigen Exp Cell Res 1996 224: 264–271
Mouly V et al. SV40 large T antigen interferes with adult myosin heavy chain expression, but not with differentiation of human satellite cells Exp Cell Res 1996 225: 268–276
Counter CM et al. Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity EMBO J 1992 11: 1921–1929
Counter CM et al. Stabilization of short telomeres and telomerase activity accompany immortalization of Epstein–Barr virus-transformed human B lymphocytes J Virol 1994 68: 3410–3414
Shay JW, Wright WE . Quantitation of the frequency of immortalization of normal human diploid fibroblasts by SV40 large T-antigen Exp Cell Res 1989 184: 109–118
Ray FA, Kraemer PM . Iterative chromosome mutation and selection as a mechanism of complete transformation of human diploid fibroblasts by SV40 T antigen Carcinogenesis 1993 14: 1511–1516
Montalto MC, Ray FA . Telomerase activation during the linear evolution of human fibroblasts to tumorigenicity in nude mice (published erratum appears in Carcinogenesis 1997 Jun; 18(6): 1281) Carcinogenesis 1996 17: 2631–2634
Greider CW, Blackburn EH . Telomeres, telomerase and cancer Sci Am 1996 274: 92–97
Bodnar AG et al. Extension of life-span by introduction of telomerase into normal human cells Science 1998 279: 349–352
Seigneurin-Venin S et al. Transplantation in Scid mouse of normal and DMD myoblasts expressing the telomerase gene. (Submitted)
Counter CM et al. Dissociation among in vitro telomerase activity, telomere maintenance, and cellular immortalization Proc Natl Acad Sci USA 1998 95: 14723–14728
Zhu J, Wang H, Bishop JM, Blackburn EH . Telomerase extends the lifespan of virus-transformed human cells without net telomere lengthening (see comments) Proc Natl Acad Sci USA 1999 96: 3723–3728
Halvorsen TL, Leibowitz G, Levine F . Telomerase activity is sufficient to allow transformed cells to escape from crisis Mol Cell Biol 1999 19: 1864–1870
Miranda AF, Babiss LE, Fisher PB . Transformation of human skeletal muscle cells by simian virus 40 Proc Natl Acad Sci USA 1983 80: 6581–6585
Iujvidin S, Fuchs O, Nudel U, Yaffe D . SV40 immortalizes myogenic cells: DNA synthesis and mitosis in differentiating myotubes Differentiation 1990 43: 192–203
Moisset PA et al. Transplantation of large T antigen-immortalized myoblasts Gene Therapy (Submitted)
Hahn WC et al. Creation of human tumour cells with defined genetic elements Nature 1999 400: 464–468
Kim NW et al. Specific association of human telomerase activity with immortal cells and cancer Science 1994 266: 2011–2015
Allsopp RC et al. Telomere length predicts replicative capacity of human fibroblasts Proc Natl Acad Sci USA 1992 89: 10114–10118
Acknowledgements
We thank F Tardif, M Goulet and B Roy for technical assistance, Drs P Leboulch (Masschusetts Institute of Technology, Cambridge) and W Wright (Department of Cell Biology and Neuroscience, The University of Texas Southwestern Medical Center) for their generous gifts. This work was supported by the Association Française contre les Myopathies (AFM) and the Muscular Dystrophy Association (MDA) and The National Institute On Aging (Ago 1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Seigneurin-Venin, S., Bernard, V. & Tremblay, J. Telomerase allows the immortalization of T antigen-positive DMD myoblasts: a new source of cells for gene transfer application. Gene Ther 7, 619–623 (2000). https://doi.org/10.1038/sj.gt.3301132
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301132
Keywords
This article is cited by
-
Impaired telomerase activity hinders proliferation and in vitro transformation of Penaeus monodon lymphoid cells
Cytotechnology (2016)
-
RTEL1 and TERT polymorphisms are associated with astrocytoma risk in the Chinese Han population
Tumor Biology (2013)
-
Human telomerase reverse transcriptase and glucose-regulated protein 78 increase the life span of articular chondrocytes and their repair potential
BMC Musculoskeletal Disorders (2012)
-
CDK4 and cyclin D1 allow human myogenic cells to recapture growth property without compromising differentiation potential
Gene Therapy (2011)
-
Autologous Transplantation of Muscle Precursor Cells Modified with a Lentivirus for Muscular Dystrophy: Human Cells and Primate Models
Molecular Therapy (2007)