Abstract
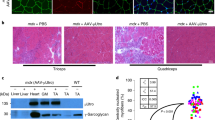
Ovine adenovirus (OAV) vectors represent a promising tool for human gene therapy since these vectors overcome the problem of pre-existing immunity against human adenovirus vectors. In this report we investigated the in vivo characteristics of this novel vector system with respect to its potential for gene transfer into skeletal muscle. We found that moderate doses of an OAV-derived vector expressing the human α1-antitrypsin gene (OAVhaat) infected skeletal muscle in mice very efficiently resulting in high serum hAAT levels. The infection was restricted to skeletal muscle, but gene expression was transient and vector DNA was rapidly cleared. Vector clearance was also observed with a vector that lacked the transgene. The loss of vector DNA was accompanied by a cellular immune response in the infected muscle but was not connected with detectable expression of early or late genes of the viral backbone as analyzed by RT-PCR. A very low dose of OAVhaat (3 × 107 infectious particles) was sufficient to produce reasonable amounts (>100 ng/ml) of serum hAAT, and this was accompanied by a weak immune response to the vector. Under these conditions, a second intramuscular injection of the same recombinant OAV vector was successful. Our study expands the known tissue tropism of OAV-derived vectors in vivo and points to the possible utility of the vector for muscle gene transfer and vaccination.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Barr E, Leiden JM . Systemic delivery of recombinant proteins by genetically modified myoblasts Science 1991 254: 1507–1509
Dai Y, Roman M, Naviaux RK, Verma IM . Gene therapy via primary myoblasts: long-term expression of factor IX protein following transplantation in vivo Proc Natl Acad Sci USA 1992 89: 10892–10895
Wolff JA et al. Long-term persistence of plasmid DNA and foreign gene expression in mouse muscle Hum Mol Genet 1992 1: 363–369
Tripathy SK et al. Long-term expression of erythropoietin in the systemic circulation of mice after intramuscular injection of a plasmid DNA vector Proc Natl Acad Sci USA 1996 93: 10876–10880
Hartikka J et al. An improved plasmid DNA expression vector for direct injection into skeletal muscle Hum Gene Ther 1996 7: 1205–1217
Dai Y et al. Cellular and humoral immune response to adenoviral vectors containing factor IX gene: tolerization of factor IX and vector antigens allows for long-term expression Proc Natl Acad Sci USA 1995 92: 1401–1405
Svensson EC et al. Long-term erythropoietin expression in rodents and non-human primates following intramuscular injection of a replication-defective adenoviral vector Hum Gene Ther 1997 8: 1797–1806
Clemens PR et al. In vivo muscle gene transfer of full-length dystrophin with an adenoviral vector that lacks all viral genes Gene Therapy 1996 3: 965–972
Kumar-Singh R, Chamberlain JS . Encapsidated adenovirus minichromosomes allow delivery and expression of a 14kb dystrophin cDNA to muscle cells Hum Mol Genet 1996 5: 913–921
Chen H-H et al. Persistence in muscle of an adenoviral vector that lacks all viral genes Proc Natl Acad Sci USA 1997 94: 1645–1650
Herzog RW et al. Stable gene transfer and expression of human blood coagulation factor IX after intramuscular injection of recombinant adeno-associated virus Proc Natl Acad Sci USA 1997 94: 5804–5809
Kessler PD et al. Gene delivery to skeletal muscle results in sustained expression and systemic delivery of a therapeutic protein Proc Natl Acad Sci USA 1996 93: 14082–14087
Snyder RO et al. Efficient and stable adeno-associated virus-mediated transduction in the skeletal muscle of adult immunocompetent mice Hum Gene Ther 1997 8: 1891–1900
Monahan PE et al. Direct intramuscular injection with recombinant AAV vectors results in sustained expression in a dog model of hemophilia Gene Therapy 1998 5: 40–49
Ascadi G et al. A differential efficiency of adenovirus-mediated in vivo gene transfer into skeletal muscle cells of different maturity Hum Mol Genet 1994 3: 579–584
Ascadi G et al. Cultured human myoblasts and myotubes show markedly different transducibility by replication-defective adenovirus recombinants Gene Therapy 1994 1: 338–340
Nalbantoglu J, Pari G, Karpati G, Holland PC . Expression of the primary coxsackie and adenovirus receptor is downregulated during skeletal muscle maturation and limits the efficacy of adenovirus-mediated gene delivery to muscle cells Hum Gene Ther 1999 10: 1009–1019
Horwitz MS . Adenoviruses. In: Fields BN, Knipe DM, Howley PM (eds) Fields Virology Lippincott^Raven: Philadelphia, New York 1996 pp 2149–2197
Chirmule N et al. Immune response to adenovirus and adeno-associated virus in humans Gene Therapy 1999 6: 1574–1583
Flomenberg P, Piaskowski V, Truitt RL, Casper JT . Characterization of human T cell responses to adenovirus J Infect Dis 1995 171: 1090–1096
Hofmann C et al. Ovine adenovirus vectors overcome preexisting humoral immunity against human adenoviruses in vivo J Virol 1999 73: 6930–6936
Löser P et al. Ovine adenovirus vectors promote efficient gene delivery in vivo Gene Ther Mol Biol 1999 4: 33–43
Boyle DB et al. Characterisation of Australian ovine adenovirus isolates Vet Microbiol 1994 41: 281–291
Vrati S, Boyle DB, Kockerhans R, Both GW . Sequence of ovine adenovirus 100k hexon assembly, 33k, pVIII and fiber genes: early region E3 is not in the expected location Virology 1995 209: 400–408
Vrati S, Brookes DE, Boyle DB, Both GW . Nucleotide sequence of ovine adenovirus tripartite leader sequence and homologous of IVa2, DNA polymerase and terminal proteins Gene 1996 177: 35–41
Vrati S et al. Unique genome arrangement of an ovine adenovirus: identification of new proteins and proteinase cleavage sites Virology 1996 220: 186–199
Khatri A, Both GW . Identification of transcripts and promoter regions of ovine adenovirus OAV287 Virology 1998 245: 128–141
Venktesh A, Watt F, Xu ZZ, Both GW . Ovine adenovirus (OAV287) lacks a virus-associated RNA gene J Gen Virol 1998 79: 509–516
Harrach B et al. Close phylogenetic relationship between egg drop syndrome virus, bovine adenovirus serotype 7, and ovine adenovirus strain 287 Virology 1997 229: 302–306
Harrach B, Benkö M . Phylogenetic analysis of adenovirus sequences. Proof of the necessity of establishing a third genus in the adenoviridae family. In: Wold WSM (ed) Adenovirus Methods and Protocols Humana Press, Totowa 1999 309–339
Barr D et al. Strain related variations in adenovirally mediated transgene expression from mouse hepatocytes in vivo: comparisons between immunocompetent and immunodeficient inbred strains Gene Therapy 1995 2: 151–155
Michou AI et al. Adenovirus-mediated gene transfer: influence of transgene, mouse strain and type of immune response on persistence of transgene expression Gene Therapy 1997 4: 473–482
Morral N et al. Immune response to reporter proteins and high viral dose limit duration of expression with adenoviral vectors: comparison of E2a wildtype and E2a deleted vectors Hum Gene Ther 1997 8: 1275–1286
Schowalter DB et al. Implication of interfering antibody formation and apoptosis as two different mechanisms leading to variable duration of adenovirus-mediated transgene expression in immune-competent mice J Virol 1999 73: 4755–4766
Vilquin J-T et al. FK506 immunosuppression to control the immune reactions triggered by first-generation adenovirus-mediated gene transfer Hum Gene Ther 1995 6: 1391–1401
Yang Y, Haecker SE, Su Q, Wilson JM . Immunology of gene therapy with adenoviral vectors in mouse skeletal muscle Hum Mol Genet 1996 5: 1703–1712
Yang Y et al. Cellular immunity to viral antigens limits E1-deleted adenoviruses for gene therapy Proc Natl Acad Sci USA 1994 91: 4407–4411
Yang Y et al. Inactivation of E2a in recombinant adenoviruses improves the prospect for gene therapy in cystic fibrosis Nat Genet 1994 7: 362–369
Yang Y, Su Q, Wilson JM . Role of viral antigens in destructive cellular immune responses to adenovirus vector-transduced cells in mouse lungs J Virol 1996 70: 7209–7212
van Deutekom JCT et al. Extended tropism of an adenoviral vector does not circumvent the maturation-dependent transducibility of mouse skeletal muscle J Gene Med 1999 1: 393–399
Kremer EJ, Boutin S, Chillon M, Danos O . Canine adenovirus vectors: an alternative for adenovirus-mediated gene transfer J Virol 2000 74: 505–512
Brann T et al. Adenoviral vector-mediated expression of physiologic levels of human factor VIII in non-human primates Hum Gene Ther 1999 10: 2099–3011
Connelly S et al. Sustained expression of therapeutic levels of human factor VIII in mice Blood 1996 87: 4671–4677
Rothel JS et al. Sequential nucleic acid and recombinant adenovirus vaccination induces host-protective immune responses against Taenia ovis infection in sheep Parasite Immunol 1997 19: 221–227
Tripathy SK, Black HB, Goldwasser E, Leiden JM . Immune response to transgene-encoded proteins limit the stability of gene expression after injection of replication-defective adenovirus vectors Nature Med 1996 2: 545–550
Kafri T et al. Cellular immune response to adenoviral vector infected cells does not require de novo viral gene expression: implications for gene therapy Proc Natl Acad Sci USA 1998 95: 11377–11382
Jooss K, Yang Y, Fisher KJ, Wilson JM . Transduction of dendritic cells by DNA viral vectors directs the immune response to transgene products in muscle fibers J Virol 1998 72: 4212–4223
Pye D . Cell lines for growth of sheep viruses Austr Vet J 1989 66: 231–232
Sandig V et al. HBV-derived promoters direct liver-specific expression of an adenovirally transduced LDL receptor gene Gene Therapy 1996 3: 1002–1009
Cichon G, Strauss M . Transient immunosuppression with 15-deoxyspergualin prolongs reporter gene expression and reduces humoral immune response after adenoviral gene transfer Gene Therapy 1998 5: 85–90
Acknowledgements
We wish to thank Ulrike Schneeweiss and Gabi N'Diaye for excellent technical assistance and Gary S Jennings for critical reading of the manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Löser, P., Hillgenberg, M., Arnold, W. et al. Ovine adenovirus vectors mediate efficient gene transfer to skeletal muscle. Gene Ther 7, 1491–1498 (2000). https://doi.org/10.1038/sj.gt.3301260
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301260
Keywords
This article is cited by
-
Chick embryo lethal orphan virus can be polymer-coated and retargeted to infect mammalian cells
Gene Therapy (2006)
-
Gene therapy progress and prospects: adenoviral vectors
Gene Therapy (2003)
-
Gene Therapy Progress and Prospects: Alpha-1 antitrypsin
Gene Therapy (2003)
-
Viral vectors for gene therapy: the art of turning infectious agents into vehicles of therapeutics
Nature Medicine (2001)