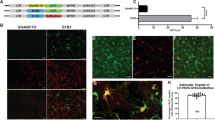
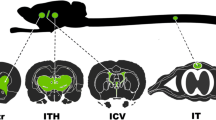
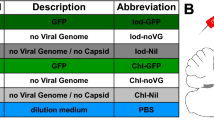
Abstract
The use of viral vectors for gene delivery into mammalian cells provides a new approach in the treatment of many human diseases. The first viral vector approved for human clinical trials was murine leukemia virus (MLV), which remains the most commonly used vector in clinical trials to date. However, the application of MLV vectors is limited since MLV requires cells to be actively dividing in order for transduction and therefore gene delivery to occur. This limitation precludes the use of MLV for delivering genes to the adult CNS, where very little cell division is occurring. However, we speculated that this inherent limitation of MLV may be overcome by utilizing the known mitogenic effect of growth factors on cells of the CNS. Specifically, an in vivo application of growth factor to the adult brain, if able to induce cell division, could enhance MLV-based gene transfer to the adult brain. We now show that an exogenous application of basic fibroblast growth factor induces cell division in vivo. Under these conditions, where cells of the adult brain are stimulated to divide, MLV-based gene transfer is significantly enhanced. This novel approach precludes any vector modifications and provides a simple and effective way of delivering genes to cells of the adult brain utilizing MLV-based retroviral vectors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rosenberg SA, Blaese RM, Anderson WF . The N2-TIL human gene transfer protocol Hum Gene Ther 1990 1: 73–92
ORDA report: Human Gene Therapy Protocols (12/1/98) Office of Recombinant DNA activities, NIH, Bethesda, MD, 1998
Anderson WF . Human gene therapy Nature 1998 392: (Suppl) 25–30
Vile RG, Russell SJ . Retroviruses as vectors Br Med Bull 1995 51: 12–30
Varmus H . Retroviruses Science 1988 240: 1427–1435
Frisch EF, Temin HM . Inhibition of viral DNA synthesis in stationary chicken embryo fibroblasts infected with avian retroviruses J Virol 1977 24: 461–469
Miller DG, Adam MA, Miller AD . Gene transfer by retrovirus vectors occurs only in cells that are actively replicating at the time of infection Molec Cell Biol 1990 10: 4239–4242
Ridet JL, Privat A . Gene therapy in the central nervous system: direct versus indirect gene delivery J Neurosci Res 1995 42: 287–293
Friedmann T . Gene therapy for neurological disorders Trends Genet 1994 10: 210–214
Vescovi AL, Reynolds BA, Fraser DD, Weiss S . βFGF regulates the proliferative state of unipotent (neuronal) and bipotent (neuronal/astroglial) EGF-generated CNS progenitor cells Neuron 1993 11: 951–966
Engel NJ, Wolswijk G . Oligodendrocyte-type-2 astrocyte (O-2A) progenitor cells derived from adult rat spinal cord: in vitro characteristic and response to PDGF, bFGF and NT-3 Glia 1996 16: 16–26
Noble M et al. Platelet-derived growth factor promotes division and motility and inhibits premature differentiation of the oligodendrocyte/type-2 astrocyte progenitor cell Nature 1988 333: 560–562
Richardson WD . A role for platelet-derived growth factor in normal gliogenesis in the central nervous system Cell 1988 53: 309–319
Ferry N et al. Retroviral-mediated gene transfer into hepatocytes in vivo Proc Nat Acad Sci USA 1991 88: 8377–8381
Bosch A et al. Proliferation induced by keratinocyte growth factor enhancers in vivo retroviral-mediated gene transfer to mouse hepatocytes J Clin Invest 1996 98: 2683–2687
Forbes SJ et al. Retroviral gene transfer to the liver in vivo during tri-iodothyronine induced hyperplasia Gene Therapy 1998 5: 552–555
Colello RJ, Clark L, King L . The cellular response of the adult mammalian CNS to growth factor application Soc Neurosci 1997 23: 1430 (Abstr.)
Pettmann B, Weibel M, Sensenbrenner M, Labourdette, G . Purification of two astroglial growth factors from bovine brain FEBS Lett 1985 189: 102–108
Ray J, Gage FH . Spinal cord neuroblasts proliferate in response to fibroblast growth factor J Neurosci 1994 14: 3548–3564
King LA et al. Growth factor enhanced retroviral gene transfer to the adult CNS Soc Neurosci 1999 131.19: 329 (Abstr.)
Gratzner HG . Monoclonal antibody to 5-bromo-and 5-iodeoxyuridine: a new reagent for detection of DNA replication Science 1982 218: 474–475
Miller MW, Nowakowski RS . Use of bromodeoxyuridine-immunohistochemistry to examine the proliferation, migration and time or origin of cells in the central nervous system Brain Res 1988 457: 44–52
Gomez-Pinilla F, Vu L, Cotman CW . Regulation of astrocyte proliferation by FGF-2 and heparan sulfate in vivo J Neurosci 1995 15: 2021–2029
Amat JA, Ishiguro H, Nakamura K, Norton WT . Phenotype diversity and kinetics of proliferating microglia and astrocytes following cortical stab wounds Glia 1996 16: 368–382
Mori K, Yamagami S, Kawakita Y . Thymidine metabolism and deoxyribonucleic acid synthesis in the developing rat brain J Neurochem 1970 17: 835–843
Dahl D, Bignami A . Immunogenic properties of the glial fibrillary acidic protein Brain Res 1976 116: 150–157
Bignami A, Dahl D . Localization of vimentin, the nonspecific intermediate filament protein in embryonal glia Dev Biol 1982 91: 286–295
Thomas WE . Brain macrophages: evaluation of microglia and their functions Brain Res Rev 1992 17: 61–74
Saranat HB, Nochlin D, Born DE . Neuronal nuclear antigen (NeuN): a marker of neuronal maturation in early human fetal nervous system Brain Dev 1998 20: 88–94
Trapnell BC, Gorziglia M . Gene therapy using adenoviral vectors Curr Opin Biotechnol 1994 5: 617–625
Dai Y et al. Cellular and humoral immune responses to adenoviral vectors containing factor IX gene: tolerization of factor IX and vector antigens allows for long-term expression Proc Natl Acad Sci USA 1995 92: 1401–1405
Byrnes AP, Rusby JE, Wood MJA, Charlton HM . Adenovirus gene transfer causes inflammation in the brain Neurosci 1995 66: 1015–1024
Kaplitt MG et al. Long-term gene expression and phenotypic correction using adeno-associated virus vectors in the mammalian brain Nat Genet 1994 8: 148–154
Kremer EJ, Perricaudet M . Adenovirus and adeno-associated virus mediated gene transfer Br Med Bull 1995 51: 31–44
Verma IM, Somia N . Gene therapy – promises, problems, and prospects Nature 1997 389: 239–242
Chen X, Schmidt MC, Goins WF, Glorioso J . Two herpes simplex virus type 1 latency-active promoters differ in their contributions to latency-associated transcript expression during lytic and latent infections J Virol 1995 12: 7899–7908
Karpati G, Lochmuller H, Nalbantoglu J, Durham H . The principles of gene therapy for the nervous system Trends Neurosci 1996 19: 49–54
Krisky DM et al. Deletion of multiple immediate–early genes from herpes simplex virus reduces cytotoxicity and permits long-term expression in neurons Gene Therapy 1998 5: 1593–1603
Lewis P, Hensel M, Emerman M . Human immunodeficiency virus infection of cells arrested in the cell cycle EMBO J 1992 11: 3053–3058 (erratum, 11: 4249)
Naldini L et al. In vivo gene delivery and stable transduction of non-dividing cells by a lentiform vector Science 1996 272: 263–267
Blomer U et al. Highly efficient and sustained gene transfer in adult neurons with a lentivirus vector J Virol 1997 71: 6641–6649
Mitrophanous K et al. Stable gene transfer to the nervous system using a non-primate lentiviral vector Gene Therapy 1999 6: 1808–1818
Bukovsky AA, Song JP, Naldini L . Interaction of human immunodeficiency virus-derived vectors with wild-type virus in transduced cells J Virol 1999 73: 7087–7092
Poeschla EM, Wong-Staal F, Looney DJ . Efficient transduction of non-dividing cells by feline immunodeficiency virus lentiviral vectors Nature Med 1998 4: 354–357
Dull T et al. A third generation lentivirus vector with a conditional packaging system J Virol 1998 72: 8463–8471
Zufferey R et al. Self-inactivating lentivirus vector for safe and efficient in vivo gene delivery J Virol 1998 72: 9873–9880
Bartz SR, Vodicka MA . Production of high-titre human immunodeficiency virus type 1 pseudotyped with vesicular stomatitis virus glycoprotein Methods 1997 12: 337–342
Snyder EY, Senut M . The use of nonneuronal cells for gene delivery Neurobiol Dis 1997 4: 69–102
Gage FH, Kawaja MD, Fisher LJ . Genetically modified cells: applications for intracerebral grafting Trends Neurosci 1991 14: 328–333
Calvo JL, Carbonell AL, Boya J . Co-expression of glial fibrillary protein and vimentin in reactive astrocytes following brain injury in rats Brain Res 1991 566: 333–336
Morrison RS, De Vellis J . Growth of purified astrocytes in a chemically defined medium Proc Natl Acad Sci USA 1981 78: 7205–7209
Menon VK, Landerholm TE . Intralesion injection of basic fibroblast growth factor alters glial reactivity to neural trauma Exp Neurol 1994 129: 142–154
Mocchetti I, Wrathall JR . Neurotrophic factors in the central nervous system trauma J Neurotrauma 1995 12: 853–870
Anchan RM et al. EGF and TGF-alpha stimulate retinal neuroepithelial cell proliferation in vitro Neuron 1991 6: 923–936
Kilpatrick TJ, Bartlett PF . Cloned multipotential precursors from the mouse cerebrum require FGF-2, whereas glial restricted precursors are stimulated with either FGF-2 or EGF J Neurosci 1995 15: 3653–3661
Lundberg C, Horellou P, Mallet J, Bjorklund A . Generation of dopa-producing astrocytes by retroviral transduction of the human tyrosine hydroxylase gene: in vitro characterization and in vivo effects of the rat Parkinson model Exp Neurol 1996 139: 39–53
Kirik D, Rosenblad C, Bjorkland A . Characterization of behavioral and neurodegenerative changes following partial lesions of the nigrostriatal dopamine system induced by intrastriatal 6-hydroxydopamine in the rat Exp Neurol 1998 152: 259–277
Rapraeger ASC, Krufka A, Olwin BB . Requirement of heparan sulfate for bFGF-mediated fibroblast growth and myoblast differentiation Science 1991 252: 1705–1708
Yayon A et al. Cell surface, heparin-like molecules are required for binding of basic fibroblast growth factor to its high affinity receptor Cell 1991 64: 841–848
Rogers AW . Techniques in Autoradiography Elsevier/North-Holland Biomedical Press: Amsterdam 1967
Soneoka Y et al. A transient three-plasmid expression system for the production of high titer retroviral vectors Nucleic Acids Res 1995 23: 628–633
Kim VN, Mitrophanous K, Kingsman SM, Kingsman AJ . Minimal requirement for lentivirus vector based on human immunodeficiency virus type 1 J Virol 1998 72: 811–816
Acknowledgements
This study was supported by grants from the Jeffress Trust Foundation and Oxford BioMedica.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
King, L., Mitrophanous, K., Clark, L. et al. Growth factor enhanced retroviral gene transfer to the adult central nervous system. Gene Ther 7, 1103–1111 (2000). https://doi.org/10.1038/sj.gt.3301198
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301198
Keywords
This article is cited by
-
Viruses exploit growth factor mechanisms to achieve augmented pathogenicity and promote tumorigenesis
Archives of Microbiology (2024)