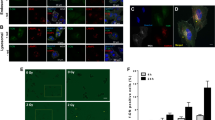
Abstract
Atm, the gene mutated in ataxia-telangiectasia (AT) patients, is an essential component of the signal transduction pathway that responds to DNA damage due to ionizing radiation (IR). We attenuated ATM protein expression in human glioblastoma cells by expressing antisense RNA to a functional domain of the atm gene. While ATM expression decreased, constitutive expression of p53 and p21 increased. Irradiated ATM-attenuated cells failed to induce p53, demonstrated radioresistant DNA synthesis, and increased radiosensitivity. Antisense-ATM gene therapy in conjunction with radiation therapy may provide a novel strategy for the treatment of cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Taylor AM et al. Ataxia telangiectasia: a human mutation with abnormal radiation sensitivity Nature 1975 258: 427–429
Gatti RA et al. Ataxia-telangiectasia: an interdisciplinary approach to pathogenesis Medicine 1991 70: 99–117
Beamish H, Lavin MF . Radiosensitivity in ataxia-telangiectasia: anomalies in radiation-induced cell cycle delay Int J Radiat Biol 1994 65: 175–184
Kastan MB et al. A mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is defective in ataxia-telangiectasia Cell 1992 71: 587–597
Painter RB, Young BR . Radiosensitivity in ataxia-telangiectasia: a new explanation Proc Natl Acad Sci USA 1980 77: 7315–7317
Savitsky K et al. A single ataxia telangiectasia gene with a product similar to PI-3 kinase (see comments) Science 1995 268: 1749–1753
Morgan SE et al. Fragments of ATM which have dominant-negative or complementing activity Molec Cell Biol 1997 17: 2020–2029
Barlow C et al. Atm-deficient mice: a paradigm of ataxia telangiectasia Cell 1996 86: 159–171
Xu Y, Baltimore D . Dual roles of ATM in the cellular response to radiation and in cell growth control (see comments) Genes Dev 1996 10: 2401–2410
Khanna KK et al. Nature of G1/S cell cycle checkpoint defect in ataxia-telangiectasia Oncogene 1995 11: 609–618
Banin S et al. Enhanced phosphorylation of p53 by ATM in response to DNA damage Science 1998 281: 1674–1677
Canman CE et al. Activation of the ATM kinase by ionizing radiation and phosphorylation of p53 Science 1998 281: 1677–1679
Westphal CH et al. Loss of atm radiosensitizes multiple p53 null tissues Cancer Res 1998 58: 5637–5639
Herzog KH et al. Requirement for Atm in ionizing radiation-induced cell death in the developing central nervous system Science 1998 280: 1089–1091
Barlow C et al. Atm selectively regulates distinct p53-dependent cell-cycle checkpoint and apoptotic pathways Nat Genet 1997 17: 453–456
Young BR, Painter RB . Radioresistant DNA synthesis and human genetic diseases Hum Genet 1989 82: 113–117
Haas-Kogan DA et al. p53-dependent G1 arrest and p53-independent apoptosis influence the radiobiologic response of glioblastoma Int J Radiat Oncol Biol Phys 1996 36: 95–103
Yount GL et al. Cell cycle synchrony unmasks the influence of p53 function on radiosensitivity of human glioblastoma cells Cancer Res 1996 56: 500–506
Kuerbitz SJ, Plunkett BS, Walsh WV, Kastan MB . Wild-type p53 is a cell cycle checkpoint determinant following irradiation Proc Natl Acad Sci USA 1992 89: 7491–7495
Kastan MB et al. Participation of p53 protein in the cellular response to DNA damage Cancer Res 1991 51: 6304–6311
Levine AJ . p53, the cellular gatekeeper for growth and division Cell 1997 88: 323–331
Waterman MJ, Stavridi ES, Waterman JL, Halazonetis TD . ATM-dependent activation of p53 involves dephosphorylation and association with 14–3-3 proteins Nat Genet 1998 19: 175–178
Xu Y et al. Involvement of p53 and p21 in cellular defects and tumorigenesis in Atm−/− mice Mol Cell Biol 1998 18: 4385–4390
Gosink EC, Chong MJ, McKinnon PJ . Ataxia telangiectasia mutated deficiency affects astrocyte growth but not radiosensitivity Cancer Res 1999 59: 5294–5298
West CM, Davidson SE, Roberts SA, Hunter RD . Intrinsic radiosensitivity and prediction of patient response to radiotherapy for carcinoma of the cervix Br J Cancer 1993 68: 819–823
West CM, Davidson SE, Roberts SA, Hunter, RD . The independence of intrinsic radiosensitivity as a prognostic factor for patient response to radiotherapy of carcinoma of the cervix Br J Cancer 1997 76: 1184–1190
Kelland LR, Steel, GG . Recovery from radiation damage in human squamous carcinoma of the cervix Int J Radiat Biol 1989 55: 119–127
Taghian A et al. Intrinsic radiation sensitivity may not be the major determinant of the poor clinical outcome of glioblastoma multiforme Int J Radiat Oncol Biol Phys 1993 25: 243–249
Kim JH, Kim SH, Brown SL, Freytag SO . Selective enhancement by an antiviral agent of the radiation-induced cell killing of human glioma cells transduced with HSV-tk gene Cancer Res 1994 54: 6053–6056
Rogulski KR, Kim JH, Kim SH, Freytag SO . Glioma cells transduced with an Escherichia coli CD/HSV-1 TK fusion gene exhibit enhanced metabolic suicide and radiosensitivity Hum Gene Ther 1997 8: 73–85
Hallahan DE et al. Spatial and temporal control of gene therapy using ionizing radiation Nature Med 1995 1: 786–791
Dranoff G et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte–macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity Proc Natl Acad Sci USA 1993 90: 3539–3543
Ponten J, Macintyre EH . Long term culture of normal and neoplastic human glia Acta Pathol Microbiol Scand 1968 74: 465–486
Harlow E, Lane D (eds) . Immunoblotting. In: Antibodies: A Laboratory Manual Cold Spring Harbor Laboratory Press: Plainview, NY 1988 pp471–510
Acknowledgements
We thank Zoya Niazova and Zuoheng Fan for technical assistance, TK Pandita for providing the pGEM-ATM14 plasmid, J Roy Chowdhury and R Kucheralapati for helpful discussions and reviewing the manuscript. This work was supported by NIH grant (NS 34746 to TKP) and by a pilot project grant from the Department of Radiation Oncology, Montefiore Medical Center, Bronx, NY.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Guha, C., Guha, U., Tribius, S. et al. Antisense ATM gene therapy: a strategy to increase the radiosensitivity of human tumors. Gene Ther 7, 852–858 (2000). https://doi.org/10.1038/sj.gt.3301174
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301174