Abstract
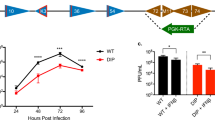
Replication-competent, attenuated mutants of herpes simplex virus type 1 (HSV-1) have been shown to be efficacious for tumor therapy. However, these studies did not address the consequences of prior exposure to HSV, as will be the case with many patients likely to receive this therapy. Two strains of mice, A/J and BALB/c, were infected with wild-type HSV-1 by intraperitoneal injection and the immune response was determined by plaque reduction assay for neutralizing antibody and ELISA for IgG and IgM. Syngeneic tumors, N18 neuroblastoma and CT26 colon carcinoma, were implanted subcutaneously in HSV-1 seropositive and naive A/J and BALB/c mice, respectively. Established tumors were subsequently treated intratumorally with a multi-mutated HSV-1, G207. G207 inhibited tumor growth to a similar extent whether the mice were seropositive or not. We next examined the effect of multiple intratumoral inoculations of a 10-fold lower dose of G207 on tumor growth. In the multiple treatment group (biweekly for 3 weeks), 75% of tumors were cured, whereas no cures were seen in the single treatment group. We conclude that HSV seropositivity should not deleteriously affect the efficacy of G207 tumor therapy, and multiple inoculations of virus should be considered for clinical evaluation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Whitley RJ . Herpes simplex viruses In: Fields BN, Knipe DM, Howley PM (eds) . Fields Virology, third edition, vol. 2 Lippincott-Raven: Philadelphia 1996 pp 2297–2342
Mester JC, Rouse BT . The mouse model and understanding immunity to herpes simplex virus Rev Infect Dis 1991 13: S935–S945
Meignier B, Longnecker R, Roizman B . In vivo behavior of genetically engineered herpes simplex viruses R7017 and R7020: construction and evaluation in rodents J Infect Dis 1988 158: 602–614
Burke RL . Contemporary approaches to vaccination against herpes simplex virus Curr Top Microbiol Immunol 1992 179: 137–158
McDermott MR et al. Immunity in the female genital tract after intravaginal vaccination of mice with an attenuated strain of herpes simplex virus type 2 J Virol 1984 51: 747–753
Halford WP, Veress LA, Gebhardt BM, Carr DJJ . Innate and acquired immunity to herpes simplex virus type 1 Virology 1997 236: 328–337
Walker J, Leib DA . Protection from primary infection and establishment of latency by vaccination with a herpes simplex virus type 1 recombinant deficient in the virion host shutoff (vhs) function Vaccine 1998 16: 1–5
Corey L, Reeves WC, Holmes KK . Cellular immune response in genital herpes simplex virus infection New Engl J Med 1978 299: 986–991
Ashley R, Benedetti J, Corey L . Humoral immune response to HSV-1 and HSV-2 viral proteins in patients with primary genital herpes J Med Virol 1985 17: 153–166
Whitley RJ, Kimberlin DW, Roizman B . Herpes simplex viruses Clin Infect Dis 1998 26: 541–553
Eberle R, Mou SW, Zaia JA . The immune response to herpes simplex virus: comparison of the specificity and relative titers of serum antibodies directed against viral polypeptides following primary herpes simplex virus type 1 infections J Med Virol 1985 16: 147–162
Buchman TG, Roizman B, Nahmias AJ . Demonstration of exogenous genital reinfection with herpes simplex virus type 2 by restriction endonuclease fingerprinting of viral DNA J Infect Dis 1979 140: 295–304
Lodmell DL, Niwa A, Hayashi K, Notkins AL . Prevention of cell-to-cell spread of herpes simplex virus by leukocytes J Exp Med 1973 137: 706–720
Lopez C, Arvin AM, Ashley R . Immunity to herpesvirus infections in humans. In: Roizman B, Whitley RJ, Lopez C (eds) . The Human Herpesviruses Raven: New York 1993 397–425
Mineta T et al. Attenuated multi-mutated herpes simplex virus-1 for the treatment of malignant gliomas Nature Med 1995 1: 938–943
Yazaki T, Manz HJ, Rabkin SD, Martuza RL . Treatment of human malignant meningiomas by G207, a replication-competent multimutated herpes simplex virus 1 Cancer Res 1995 55: 4752–4756
Toda M, Rabkin SD, Martuza RL . Treatment of human breast cancer in a brain metastatic model by G207, a replication-competent multimutated herpes simplex virus 1 Hum Gene Ther 1998 9: 2177–2185
Toda M, Rabkin SD, Kojima H, Martuza L . Herpes simplex virus as an in situ cancer vaccine for the induction of specific anti-tumor immunity Hum Gene Ther 1999 10: 385–393
Chou J, Kern ER, Whitley RJ, Roizman B . Mapping of herpes simplex virus-1 neurovirulence to gamma 34.5, a gene nonessential for growth in culture Science 1990 250: 1262–1266
Goldstein DJ, Weller SK . Herpes simplex virus type 1-induced ribonucleotide reductase activity is dispensible for virus growth and DNA synthesis: isolation and characterization of an ICP6 1acZ insertion mutant J Virol 1988 62: 196–205
Nguyen LH, Knipe DM, Finberg RW . Replication-defective mutants of herpes simplex virus (HSV) induce cellular immunity and protect against lethal HSV infection J Virol 1992 66: 7067–7072
Morrison LA, Knipe DM . Contributions of antibody and T cell subsets to protection elicited by immunization with a replication-defective mutant of herpes simplex virus type 1 Virology 1997 239: 315–326
Morrison LA, Knipe DM . Immunization with replication-defective mutants of herpes simplex virus type 1: sites of immune intervention in pathogenesis of challenge virus infection J Virol 1994 68: 689–696
Knoblich A, Gortz J, Harle-Grupp V, Falke D . Kinetics and genetics of herpes simplex virus-induced antibody formation in mice Infect Immun 1983 39: 15–23
Kahlon J, Lakeman FD, Ackermann M, Whitley RJ . Human antibody response to herpes simplex virus-specific polypeptides after primary and recurrent infection J Clin Microbiol 1986 23: 725–730
Zawatzky R, Hilfenhaus J, Marcucci F, Kirchner H . Experimental infection of inbred mice with herpes simplex virus type 1. I. Investigation of humoral and cellular immunity and of interferon induction J Gen Virol 1981 53: 31–38
Lopez C . Genetics of natural resistance to herpesvirus infections in mice Nature 1975 258: 152–153
Fearon ER et al. Induction in a murine tumor of immunogenic tumor variants by transfection with a foreign gene Cancer Res 1988 48: 2975–2980
Plautz GE et al. Immunotherapy of malignancy by in vivo gene transfer into tumors Proc Natl Acad Sci USA 1993 90: 4645–4649
Toda M, Martuza RL, Kojima H, Rabkin SD . In situ cancer vaccination: a defective IL-12 defective vector/replication-competent herpes simplex virus vector combination induces local and systemic antitumor activity J Immunol 1998 160: 4457–4464
Dai Y et al. Cellular and humoral immune response to adenoviral vectors containing factor IX gene: tolerization of factor IX and vector antigens allows for long-term expression Proc Natl Acad Sci USA 1995 92: 1401–1405
Pelner L, Fowler GA, Hauts HC . Effects of concurrent infections and their toxins on the course of leukemia Act Med Scand 1958 338 (Suppl.): 1–47
Bluming AZ, Ziegler JL . Regression of Burkitt’s lymphoma in association with measles infection Lancet 1971 2: 105–106
Lindenmann J, Klein P . Viral oncolysis: increased immunogenicity of host cell antigen associated with influenza virus J Exp Med 1967 126: 93–108
Sinkovics J, Horvath J . New developments in the virus therapy of cancer: a historical review Intervirology 1993 36: 193–214
Kapoor AK, Nash AA, Wildy P . Pathogenesis of herpes simplex virus in B cell suppressed mice: the relative roles of cell-mediated and humoral immunity J Gen Virol 1982 61: 127–131
Kapoor AK et al. Pathogenesis of herpes simplex virus in congenitally athymic mice: the relative roles of cell-mediated and humoral immunity J Gen Virol 1982 60: 225–233
Mitchell BM, Stevens JG . Neuroinvasive properties of herpes simplex virus type 1 glycoprotein variants are controlled by the immune response J Immunol 1996 156: 246–255
Shimeld C, Hill TJ, Blyth WA, Easty DL . Passive immunization protects the mouse eye from damage after herpes simplex virus infection by limiting spread of virus in the nervous system J Gen Virol 1990 71: 681–687
Lopez C, O’Reilly RJ . Cell-mediated immune responses in recurrent herpesvirus infections. I. Lymphocyte proliferation assay J Immunol 1977 118: 895–902
O’Reilly RJ, Chibbaro A, Anger E, Lopez C . Cell-mediated immune responses in patients with recurrent Herpes Simplex infections. II. Infection-associated deficiency of lymphokine production in patients with recurrent herpes labialis or herpes progenitalis J Immunol 1977 118: 1095–1102
Zweerink HJ, Stanton LW . Immune response to herpes simplex virus infections: virus-specific antibodies in sera from patients with recurrent facial infections Infect Immun 1981 31: 624–630
Posavad CM, Koelle DM, Corey L . High frequency of CD8+ cytotoxic T-lymphocyte precursors specific for herpes simplex viruses in persons with genital herpes J Virol 1996 70: 8165–8168
Engler H, Zawatzky R, Kirchner H, Armerding D . Experimental infection of inbred mice with herpes simplex virus. IV. Comparison of interferon production and natural killer cell activity in susceptible and resistant adult mice Arch Virol 1982 74: 239–247
Brenner GJ, Cohen N, Moynihan JA . Similar immune response to nonlethal infection with herpes simplex virus-1 in sensitive (BALB/c) and resistant (C57BL/6) strains of mice Cell Immunol 1994 157: 510–524
Herrlinger U et al. Pre-existing herpes simplex virus 1 (HSV-1) immunity decreases, but does not abolish, gene transfer to experimental brain tumors by a HSV-1 vector Gene Therapy 1998 5: 809–819
Lopez C et al. Correlation between low natural killing of fibroblasts infected with herpes simplex virus type 1 and susceptibility to herpesvirus infections J Infect Dis 1983 147: 1030–1035
Brchm MA, Bonneau RH, Knipe DM, Tevethia SS . Immunization with a replication-deficient mutant of herpes simplex virus type 1 (HSV-1) induces a CD8+ cytotoxic T-lymphocyte response and confers a level of protection comparable to that of wild-type HSV-1 J Virol 1997 71: 3534–3544
Gathery-Segard H et al. Immune response to recombinant capsid proteins of adenovirus in humans: antifiber and anti-penton base antibodies have a synergistic effect on neutralizing activity J Virol 1998 72: 2388–2397
Juillard V et al. Long-term humoral and cellular immunity induced by a single immunization with replication-defective adenovirus recombinant vector Eur J Immunol 1995 25: 3467–3473
Yang Y, Li Q, Ertl HCJ, Wilson JM . Cellular and humoral immune responses to viral antigens create barriers to lung-directed gene therapy with recombinant adenoviruses J Virol 1995 69: 2004–2015
Zabner J et al. Repeat administration of an adenovirus vector encoding cystic fibrosis transmembrane conductance regulator ot the nasal epithelium of patients with cystic fibrosis J Clin Invest 1996 97: 1504–1511
Brattain MG et al. Establishment of mouse colonic carcinoma cell lines with different metastatic properties Cancer Res 1980 40: 2142–2146
Corbett TH, Griswold DPJ, Peckham JC, Schnabel FMJ . Tumor induction relationships in development of transplantable cancers of the colon in mice for chemotherapy assays, with a note on carcinogen structure Cancer Res 1975 35: 2434–2439
Hanfelt JJ . Statistical approaches to experimental design and data analysis of in vivo studies Breast Cancer Res 1997 46: 279–302
Acknowledgements
We thank Anu Iyer for technical assistance, Paul Johnson for preparation of column purified G207, Dr Joseph T Newsome and the Georgetown University Research Resource Facility staff for assistance with the animals, and Anastasia Ivanova for biostatistical help. This study was supported in part by grants from the National Institutes of Health (NS32677) and Neuro Vir, Inc. Samuel D Rabkin and Robert L Martuza are consultants to Neuro Vir, Inc. which has a license from Georgetown University to commercialize G207.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chahlavi, A., Rabkin, S., Todo, T. et al. Effect of prior exposure to herpes simplex virus 1 on viral vector-mediated tumor therapy in immunocompetent mice. Gene Ther 6, 1751–1758 (1999). https://doi.org/10.1038/sj.gt.3301003
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301003
Keywords
This article is cited by
-
Intratumoral oncolytic herpes virus G47∆ for residual or recurrent glioblastoma: a phase 2 trial
Nature Medicine (2022)
-
A phase I/II study of triple-mutated oncolytic herpes virus G47∆ in patients with progressive glioblastoma
Nature Communications (2022)
-
Anti-viral Immunity in the Tumor Microenvironment: Implications for the Rational Design of Herpes Simplex Virus Type 1 Oncolytic Virotherapy
Current Clinical Microbiology Reports (2019)
-
Pediatric cancer gone viral. Part I: strategies for utilizing oncolytic herpes simplex virus-1 in children
Molecular Therapy - Oncolytics (2015)
-
A Phase 1 Trial of Oncolytic HSV-1, G207, Given in Combination With Radiation for Recurrent GBM Demonstrates Safety and Radiographic Responses
Molecular Therapy (2014)